Abstract
Background: Multimorbidity is a prevalent risk factor for COVID-19–related complications and death. We sought to evaluate the association of homologous booster vaccination using BNT162b2 (Pfizer-BioNTech) or CoronaVac (Sinovac) with COVID-19–related deaths among people with multimorbidity during the initial Omicron wave of the COVID-19 pandemic.
Methods: Using routine clinical records from public health care facilities in Hong Kong, we conducted a territory-wide retrospective cohort study comparing people aged 18 years or older with 2 or more chronic conditions who received a homologous booster (third) dose with those who received only 2 doses, between Nov. 11, 2021, and Mar. 31, 2022. The primary outcome was death related to COVID-19.
Results: We included 120 724 BNT162b2 recipients (including 87 289 who received a booster), followed for a median of 34 (interquartile range [IQR] 20–63) days and 127 318 CoronaVac recipients (including 94 977 who received a booster), followed for a median of 38 (IQR 22–77) days. Among BNT162b2 recipients, booster-vaccinated people had fewer COVID-19–related deaths than those who received 2 doses (5 v. 34, incidence rate 1.3 v. 23.4 per million person-days, weighted incidence rate ratio [IRR] 0.05, 95% confidence interval [CI] 0.02–0.16). We observed similar results among recipients of CoronaVac booster vaccination compared with those who received only 2 doses (26 v. 88, incidence rate 5.3 v. 53.1 per million person-days, weighted IRR 0.08, 95% CI 0.05–0.12).
Interpretation: Among people with multimorbidity, booster vaccination with BNT162b2 or CoronaVac was associated with reductions of more than 90% in COVID-19–related mortality rates compared with only 2 doses. These results highlight the crucial role of booster vaccination for protecting vulnerable populations as the COVID-19 pandemic continues to evolve.
Compared with the general population, people living with multimorbidity are disproportionately burdened by the ongoing COVID-19 pandemic.1 Research shows a higher risk of SARS-CoV-2 infection2 and death related to COVID-19 among those with multimorbidity. 3,4 The global roll-out of SARS-CoV-2 vaccines, therefore, has rightfully prioritized people with underlying chronic conditions and multimorbidity.5 By late 2021, most eligible people had received at least 2 doses of the vaccines in many jurisdictions.6 Amid the emergence of new SARS-CoV-2 variants, however, people with multimorbidity may further benefit from booster vaccination, given an established tolerable safety profile of the vaccines in this particular population.7,8 Some studies have shown the effectiveness of certain vaccines against infection with new variants in the general population, such as the mRNA vaccines.9,10 However, the effectiveness of SARS-CoV-2 booster vaccination has not been well explored in people living with multimorbidity.
Despite an absence of evident local transmission of SARS-CoV-2 in Hong Kong from mid to late 2021,11 the city reported the world’s highest COVID-19–related mortality rate in proportion to population size amid the Omicron (BA.2) variant epidemic, which started in late December 2021.12 As of April 2022, more than 9000 deaths in Hong Kong were related to COVID-19, of a population of 7.5 million.11 Booster vaccination with 2 of the most widely used vaccines worldwide, namely the BNT162b2 mRNA vaccine (Fosun-BioNTech, equivalent to Pfizer-BioNTech outside China) and the CoronaVac inactivated whole-virus vaccine (Sinovac), has been available to eligible Hong Kong residents since Nov. 11, 2021. Older people, health care professionals and other priority groups were allowed to receive the booster vaccination first, before it was extended to all other adults on Jan. 1, 2022. More than 3 million people received the booster vaccination within the first 4 months of 2022.13
We sought to evaluate the effectiveness of a homologous booster dose of these vaccines in lowering the risk of COVID-19–related death among people with multimorbidity using a territory-wide electronic health record database in Hong Kong amid the Omicron wave of the pandemic.
Methods
Data sources
We identified SARS-CoV-2 vaccine recipients with multimorbidity from routine health care records provided by the Hospital Authority of Hong Kong, linked with population-based vaccination records at the Department of Health. The Hospital Authority serves as the sole provider of public inpatient services and is a major provider of outpatient services in Hong Kong, with a comprehensive electronic health record system for facilitation of clinical management. Further details are available in Appendix 1, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221068/tab-related-content.
Study design
We used a retrospective cohort study design to compare the risk of COVID-19–related deaths among adults with multimorbidity who had received a homologous booster dose against those who had received only 2 doses. We identified adults aged 18 years or older with diagnoses of 2 or more chronic conditions (of a widely adopted list of 30 conditions; Appendix 1, Supplementary Table 1) who had received a booster dose between Nov. 11, 2021 (the date of the official roll-out of booster vaccination), and Mar. 31, 2022, of the same SARS-CoV-2 vaccine they had received for the first 2 doses (BNT162b2 or CoronaVac).14,15 Both vaccines were made freely available to all residents of Hong Kong during the study period. Dozens of community vaccination centres that provided either vaccine were set up in geographically convenient public facilities for a wider reach to the community. Given its easier storage, the CoronaVac vaccine was made available in private clinics as well. The supply of both vaccines has been more than adequate since their roll-out in early 2021 (Feb. 23 for CoronaVac and Mar. 6 for BNT162b2). In general, Hong Kong residents were encouraged to receive the booster vaccine at least 180 days after their second dose. However, they were allowed to receive the booster vaccine from 90 days after the second dose for reasons such as having an underlying, immunocompromising condition. Residents could receive a booster dose less than 90 days after their second dose only with medical advice to do so, or in other exceptional cases.
We considered adults with multimorbidity (deceased or alive by the end of data availability) who had received their second vaccine dose at least 180 days before Mar. 31, 2022, but had not received the booster dose as the comparison cohort (i.e., the 2-dose group). We conducted random matching by age and sex so that a randomly chosen, booster-vaccinated individual was mapped to each 2-dose individual of the same age and sex; the date of booster vaccination served as the pseudo-index date for the 2-dose group. We adopted the 180-day interval as a criterion to ensure each age- and sex-matched 2-dose individual was assigned a pseudo-index date well after the date of their second dose. We built such a comparison cohort for each of the 2 vaccine types. We excluded people who had received heterologous vaccines and those who had received a second booster (fourth) dose from the main analysis. Hence, we included 2 study cohorts in the main analysis, namely the BNT162b2 cohort and the CoronaVac cohort.
We followed individuals from the index date (booster vaccination) or pseudo-index date until the outcome of interest, death related to COVID-19; we censored individuals upon death unrelated to COVID-19 or at the end of data availability (Mar. 31, 2022), whichever came first. We defined a death related to COVID-19 as one that was unrelated to injury or poisoning (International Classification of Diseases, 10th Revision code S00-T88), with a positive test result from a polymerase chain reaction test for SARS-CoV-2 within 28 days before death.16 We excluded people who received 2 doses but died before the pseudo-index date and people (2 doses or 3 doses) who developed multimorbidity only after the index (or pseudo-index) date.
We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement checklist to guide transparent reporting of this study.17
Statistical analysis
We used propensity score–based, inverse probability of treatment, weighted Poisson regression, stratified by vaccine type (BNT162b2 v. CoronaVac) to estimate the weighted incidence rate ratio (IRR) of death related to COVID-19, comparing those who received the booster dose and those who received only 2 doses. Covariates for weighting included age, sex, time from second dose to index or pseudo-index date (d), presence of each of the 30 conditions used to define multimorbidity, as well as a range of chronic medications within 1 year before the index or pseudo-index date (see Appendix 1, Supplementary Table 2). Details regarding subgroup, secondary and sensitivity analyses are provided in the Appendix 1.
All statistical tests were 2-sided, and we considered p values less than 0.05 to be statistically significant. We conducted statistical analysis using R version 4.0.3 (www.R-project.org). We used the svyjskm package to generate a weighted Kaplan–Meier cumulative incidence plot. Two investigators (V.K.C.Y. and X.Y.) conducted the statistical analyses independently for quality assurance.
Ethics approval
As only anonymized secondary data analyses were involved, no informed consent was required. This research is approved by the Hospital Authority Central Institutional Review Board (CIRB-2021–005–4) and the Department of Health Ethics Committee (LM171/2021).
Results
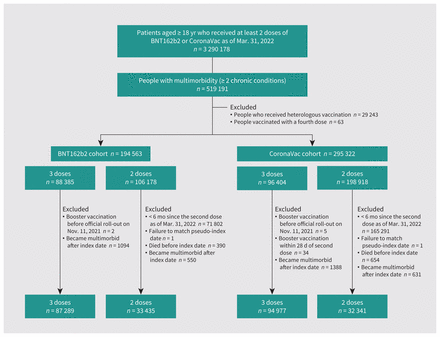
We identified 3 290 178 people aged 18 years or older who had received at least 2 doses of SARS-CoV-2 vaccines as of Mar. 31, 2022, in the database, of which 519 191 had 2 or more of the 30 chronic conditions. After further removing ineligible participants by our exclusion criteria, we included 120 724 BNT162b2 recipients (including 87 289 who received a booster) and 127 318 CoronaVac recipients (including 94 977 who received a booster) in the analyses (Figure 1). The median follow-up time was 34 (interquartile range [IQR] 20–63) days for BNT162b2 recipients and 38 (IQR 22–77) days for CoronaVac recipients.
Flow chart showing cohort selection.
Cohort characteristics
Table 1 and Table 2 show the BNT162b2 and CoronaVac cohort characteristics, respectively, before and after applying inverse probability of treatment weighting. People who received the booster dose were slightly older than those who received only 2 doses (64.9 v. 61.2 yr among BNT162b2 recipients; 67.9 v. 65.0 yr among CoronaVac recipients); the CoronaVac recipients were slightly older than the BNT162b2 recipients. The ratio of males to females was similar between groups, with slightly more males included. As of the index or pseudo-index date, about 20–30 more days had passed since the second dose vaccination among those who had received the booster than among those who received only 2 doses (Appendix 1, Supplementary Figure 1). For all 4 groups, the most prevalent chronic condition was hypertension (> 80%) followed by diabetes (> 60%), severe constipation (> 10%), chronic kidney disease (about 10%) and chronic pain (about 10%). More than 60% of participants had used calcium-channel blockers and about 60% had used lipid-lowering agents in the past year, followed by renin–angiotensin-system agents (about 50%) and antidiabetic medications (> 40%). After weighting, all characteristics had a standardized mean difference lower than 0.1, indicating a good balance between the booster and 2-dose groups for both vaccine cohorts.
Cohort characteristics before and after inverse probability of treatment weighting for recipients of BNT162b2 vaccine (booster v. 2 doses)
Cohort characteristics before and after inverse probability of treatment weighting for recipients of CoronaVac vaccine (booster v. 2 doses)
COVID-19–related deaths
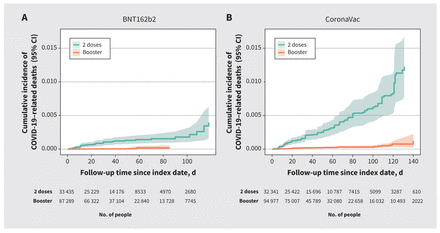
The weighted cumulative incidence of COVID-19–related deaths across the follow-up period was higher among people who were vaccinated with 2 doses than among those who received a booster (Figure 2). CoronaVac recipients had a higher rate of mortality related to COVID-19 than BNT162b2 recipients. Appendix 1, Supplementary Figure 2 shows the unweighted cumulative incidence plots.
Weighted cumulative incidence of COVID-19–related deaths after (A) BNT162b2 or (B) CoronaVac 2-dose or booster vaccination, with 95% confidence intervals (CIs) represented by the shaded area. The index date is operationalized as the date of booster vaccination or the matched pseudoindex date for those who received 2 doses of vaccine.
In total, 39 BNT162b2 recipients (including 5 who received a booster) and 114 CoronaVac recipients (including 26 who received a booster) died in relation to COVID-19, constituting an incidence rate of 0.7 per 100 000 person-days for BNT162b2 and and 1.7 per 100 000 person-days for CoronaVac. Weighted analysis estimated that a booster dose was associated with a reduced risk of COVID-19–related death among both BNT162b2 recipients (IRR 0.05, 95% confidence interval [CI] 0.02–0.16) and CoronaVac recipients (IRR 0.08, 95% CI 0.05–0.12) (Table 3).
COVID-19–related deaths among people who received either 2 doses or booster vaccination, by vaccine type (BNT162b2 or CoronaVac)
Results were similar in a weighted subgroup analysis including only those aged 60 years or older (BNT162b2: IRR 0.06, 95% CI 0.02–0.16; CoronaVac: IRR 0.07, 95% CI 0.04–0.11) (Appendix 1, Supplementary Table 3). Interaction between booster vaccination and age was not significant for either vaccine in the multivariable Poisson regression analysis (BNT162b2: p = 0.9; CoronaVac: p = 0.7) (Appendix 1, Supplementary Table 4).
In an analysis combining the vaccine cohorts, 2 doses of BNT162b2 (IRR 0.49, 95% CI 0.31–0.78), booster vaccination with CoronaVac (IRR 0.03, 95% CI 0.01–0.08) and booster vaccination with BNT162b2 (IRR 0.07, 95% CI 0.04–0.11) were all associated with a lower risk of COVID-19–related death, compared with 2 doses of CoronaVac (Appendix 1, Supplementary Table 5). In an analysis of people who received a heterologous booster vaccine, no COVID-19–related deaths were recorded for those vaccinated with a BNT162b2-BNT162b2-CoronaVac series, and a reduced incidence of COVID-19–related deaths (IRR 0.02, 95% CI 0.00–0.06) was observed for those vaccinated with a CoronaVac-CoronaVac-BNT162b2 series, compared with 2 doses of CoronaVac (Appendix 1, Supplementary Table 6). In a weighted analysis similar to the primary analysis, the incidence of SARS-CoV-2 infection was also reduced with booster vaccination compared with 2-dose vaccination for both BNT162b2 (IRR 0.32, 95% CI 0.30–0.34) and CoronaVac (IRR 0.31, 95% CI 0.29–0.33) (Appendix 1, Supplementary Table 7).
Sensitivity analyses (including one in which each comorbidity was removed from the analysis; others in which the definition of COVID-19–related mortality required a positive polymerase chain reaction test for SARS-CoV-2 within 7, 14 or 28 days before death; one in which the index date was set 14 days after booster vaccination, with adjustment for potential competing risks from non-COVID-19–related deaths; and one with exclusion of people who developed COVID-19 before receiving their second vaccine dose showed consistent results with the main analysis (Appendix 1, Supplementary Tables 8–13).
Interpretation
We found a substantially reduced risk of COVID-19–related death in adults with multimorbidity who received a homologous booster dose of BNT162b2, an mRNA vaccine, or CoronaVac, an inactivated whole-virus vaccine. These results support the effectiveness of booster doses of vaccines of 2 different technological platforms in lowering mortality among those with multimorbidity amid the Omicron epidemic. Subgroup and sensitivity analyses showed similar results, supporting the robustness of our results. As the data on SARS-CoV-2 vaccination records used for this study was provided by the sole operator of vaccine roll-out in Hong Kong, with a unified recording system, and with linked clinical records provided by a territory-wide public health care provider, our data should be highly reliable and representative. Numerous previous high-impact pharmacovigilance studies have been generated from this database.8,18–25
Since the roll-out of the BNT162b2 vaccine, observational studies worldwide have consistently shown its effectiveness against infection, severe disease and death.9,10,26–29 More recent research on the booster dose has suggested substantial extra protection, in addition to that conferred by the first and second doses.27,28,30,31 An Israeli cohort study of people aged 50 years or older reported a 90% reduced hazard of COVID-19–related death associated with the booster dose of BNT162b2 compared with only 2 doses.28 In another cohort study of people aged 12 years or older, the reduced risk of COVID-19–related death was estimated at 81%.32 Our estimates of 95% reduced risk among those with BNT162b2 booster vaccination are possibly the highest risk reductions reported to date, plausibly because of the selection of a high-risk population with multimorbidity, although the adopted age was as low as 18 years. Another potential explanation for the high effectiveness observed in our cohort may be the waning of protection conferred by the first and second doses, given that the average number of days since the second dose well exceeded 180 days in both the weighted and unweighted cohorts; this extended period may have incurred a reduction of effectiveness by more than 20–30 percentage points, compared with 14 days after vaccination, according to a systematic review.33,34 Certain underlying conditions and old age in this population may also accelerate this waning protection.35
Given its much wider use in developing countries than in developed countries, CoronaVac’s effectiveness has not been investigated as extensively as BNT162b2 beyond clinical trials, wherein vaccination (including booster vaccination) generally showed good efficacy against infection, severe disease and death.29,36–39 In the cross-platform comparison, we showed that 2 doses of CoronaVac confer lower protection than 2 doses of BNT162b2, which agrees with the existing efficacy data, but 3 doses of either CoronaVac or BNT162b2 offered similar protection. Our large postmarketing cohort study thereby supports the effectiveness of a booster dose of CoronaVac against death related to COVID-19. Our findings should inform public health policies in countries that are considering the roll-out of booster doses of CoronaVac, especially for the aging population and in populations with a high prevalence of multimorbidity.
Limitations
Similar to other pharmacoepidemiologic studies, residual confounding may be present as randomization was not possible. Specifically, people with multimorbidity who chose to receive a booster dose earlier than others may be better educated, more health aware and more proactive in health-seeking behaviours. These people may therefore be better at self-care to minimize the risk of SARS-CoV-2 infection and, thus, the analysis may overestimate the effectiveness of the booster dose. Nevertheless, people in the comparison cohort had already received 2 doses (rather than including a mix of vaccinated and unvaccinated people), and such an overestimation, if any, should be minimal and may not affect the results substantially. The outcome of this study was operationalized as death with COVID-19 rather than death from COVID-19 because the adoption of the latter requires a thorough causality assessment for each outcome event, and it was not feasible, given the limited information in the database of electronic health records for such an assessment and the large number of events. The list of conditions for multimorbidity was far from exhaustive and may not be perfectly applicable to Hong Kong, but research has suggested that prevalence tends to converge at the inclusion of 12 or more prevalent conditions.40 Moreover, the sensitivity analysis showed no substantial effects of the change of multimorbidity components on the findings. Further research should investigate the impact of an increased burden of multimorbidity on vaccine effectiveness. Although we adjusted for the presence of chronic conditions, we did not determine or adjust for their severity. We did not have a sufficient sample size to evaluate the effectiveness of heterologous booster doses across platforms, which future research with adequate data should investigate.41 People who received 2 doses and who were discouraged from receiving a booster dose owing to a recent SARS-CoV-2 infection may have had increased immunity during the follow-up period. Nevertheless, this should bias the result only toward the null hypothesis and not affect our conclusion. The 2-dose vaccinated cohort may have been infected with SARS-CoV-2 and acquired a certain degree of immunity. We did not adjust for this possibility as we took an intention-to-treat perspective. We did not have data on the exact variant type for each individual in the database. Nevertheless, the observation period coincides with the Omicron BA.2 outbreak, which has been widely reported.12 We can reasonably generalize the findings and conclusions to Omicron BA.2. The population in Hong Kong is predominantly ethnic Chinese.42 Replication of the analysis should be conducted to test for generalizability to other populations.
Conclusion
The mass roll-out of the booster dose of SARS-CoV-2 vaccines in Hong Kong in November 2021 coincided with the arrival of the Omicron variant in late 2021. Our findings suggest that this timely, massive public health measure has plausibly played a pivotal role in lowering the mortality rate amid the epidemic, especially among people living with multimorbidity. They also highlight the potential benefit from booster vaccination, specifically in vulnerable populations living with multimorbidity, and support the recent focus on older people and those with chronic conditions for future booster doses of SARS-CoV-2 vaccines beyond the first booster.43
Acknowledgements
The authors thank the Hospital Authority and the Department of Health for the generous provision of data for this study, and Lisa Lam for language proofread of the manuscript.
Footnotes
Competing interests: Francisco Tsz Tsun Lai has been supported by the RGC Postdoctoral Fellowship under the Hong Kong Research Grants Council and has received research grants from the Food and Health Bureau of the Government of the Hong Kong Special Administrative Region. Tiantian Ma reports consulting fees from Laboratory of Data Discovery for Health. Celine Sze Ling Chui has received grants from the Food and Health Bureau of the Hong Kong Government, Hong Kong Research Grant Council, Hong Kong Innovation and Technology Commission, Pfizer, IQVIA and Amgen, as well as personal fees from PrimeVigilance. Xue Li has received research grants from the Research Grants Council Early Career Scheme, the Food and Health Bureau of the Government of the Hong Kong Special Administrative Region; research and educational grants from Janssen and Pfizer; internal funding from the University of Hong Kong; and consulting fees from Merck, Sharp and Dohme, and Pfizer. Eric Yuk Fai Wan has received research grants from the Food and Health Bureau of the Government of the Hong Kong Special Administrative Region, and the Hong Kong Research Grants Council. Carlos King Ho Wong reports funding from the EuroQoL Group Research Foundation, the Hong Kong Research Grants Council, and the Hong Kong Health and Medical Research Fund. Ian Chi Kei Wong receives research funding from Amgen, Bristol Myers Squibb, Pfizer, Janssen, Bayer, GSK, Novartis, the Hong Kong Research Grants Council, the Food and Health Bureau of the Government of the Hong Kong Special Administrative Region, the National Institute for Health Research in England, the European Commission and the National Health and Medical Research Council in Australia He also reports speaker fees from Janssen and Medice, and a role as an independent non-executive director of Jacobson Medical in Hong Kong. Esther Wai Yin Chan reports grants from the Research Grants Council of Hong Kong, the Research Fund Secretariat of the Food and Health Bureau of Hong Kong, the National Natural Science Fund of China, the Wellcome Trust, Bayer, Bristol Myers Squibb, Pfizer, Janssen, Amgen, Takeda and the Narcotics Division of the Security Bureau of Hong Kong. She also reports an honorarium from the Hong Kong Hospital Authority. All competing interests are outside the submitted work. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Francisco Lai, Vincent Yan, Ian Wong and Esther Chan contributed to the conception of the work. All authors designed the study. Vincent Yan and Xuxiao Ye contributed to the acquisition and analysis of the data, and all authors interpreted the data. Francisco Lai drafted the manuscript. All of the authors revised the manuscript critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work. Francisco Lai and Vincent Yan are co–first authors with equal contributions. Ian Wong and Esther Chan share senior authorship.
Funding: This work was funded by a research grant from the Food and Health Bureau of the Government of the Hong Kong Special Administrative Region, through the Health and Medical Research Fund Research on COVID-19 (COVID1903011). The funder was not involved in the study design, data collection, data analysis, data interpretation and writing of the report. Francisco Tsz Tsun Lai and Ian Chi Kei Wong are partially supported by the Laboratory of Data Discovery for Health, funded by the AIR@InnoHK and administered by the Innovation and Technology Commission.
Data sharing: Data will not be available for others as the data custodians have not given permission.
- Accepted November 24, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/