Abstract
Background: Starting in the 2006/2007 influenza season, the US Advisory Committee on Immunization Practices expanded its recommendations for seasonal influenza vaccination to include healthy children aged 24–59 months. The parallel Canadian organization, the National Advisory Committee on Immunization, did not at that time issue a similar recommendation, thereby creating a natural experiment to evaluate the effect of the policy in the United States.
Methods: We examined data for 2000/2001 through 2008/2009 and estimated relative changes in visits to the emergency department for influenza-like illness at two pediatric hospitals, one in Boston, Massachusetts, and the other in Montréal, Quebec, following the US policy change. Models were adjusted for virologic factors, seasonal trends and all-cause utilization of the emergency department.
Results: Of 1 043 989 visits to the emergency departments of the two hospitals for any reason during the study period, 114 657 visits were related to influenza-like illness. Adjusted models estimated a 34% decline in rates of influenza-like illness among children two to four years old in the US hospital relative to the Canadian hospital (rate ratio 0.66, 95% confidence interval 0.58–0.75) following the 2006 policy change of the Advisory Committee on Immunization Practices. This was accompanied by more modest declines of 11% to 18% for the other age groups studied.
Interpretation: The divergence in influenza rates among children in the US and Canadian sample populations after institution of the US policy to vaccinate children two to four years of age is evidence that the recommendation of the US Advisory Committee on Immunization Practices resulted in a reduction in influenza-related morbidity in the target group and may have indirectly affected other pediatric age groups. Provincial adoption of the 2010 recommendation of teh National Advisory Committee on Immunization in Canada to vaccinate childen two to four years of age might positively affect influenza morbidity in Canada.
See related commentary by Peltola and colleagues at www.cmaj.ca/lookup/doi/10.1503/cmaj.111055
Seasonal influenza is an important cause of visits to the emergency department among children during winter months, and its control and prevention rely on annual vaccination.1–4 In the United States, the Advisory Committee on Immunization Practices is responsible for guiding immunization practices, and it revises its recommendations annually. Until the 2004/2005 influenza season, vaccination was targeted to primarily older individuals and those with certain medical conditions.5 On the basis of evidence that young children are at elevated risk for admission to hospital because of influenza-related complications, the recommendations were expanded for the 2004/2005 season to include healthy children aged 6–23 months.6
Starting in the 2006/2007 season, the recommendations of the Advisory Committee on Immunization Practices regarding influenza vaccination were expanded again to include healthy children aged 24–59 months, a shift that added 10.6 million children to the target group.7 For the 2008/2009 season, recommendations were further expanded to include all persons 6 months to 18 years old,8 and for the 2010/2011 season, the influenza vaccine was advised for all individuals over 6 months of age.9
In Canada, the National Advisory Committee on Immunization is the federal organization responsible for guiding the use of vaccines. Until the 2006/2007 season, the recommendations of this committee for the use of seasonal influenza vaccines were harmonized with those of its US counterpart. However, it was not until the 2010/2011 season that the Canadian committee began recommending that children two to four years old be routinely vaccinated against influenza, and not all Canadian provinces have adopted this recommendation.
We aimed to examine the effect of the 2006 recommendation of the Advisory Committee on Immunization Practices to expand influenza vaccination coverage to preschool-aged children. Interannual variation in severity of disease and effectiveness of the vaccine make it difficult to directly estimate the effect of an intervention on the incidence of influenza in any given community. Instead, we compared surveillance data from the emergency departments of two pediatric hospitals, one in Boston, Massachusetts, and the other in Montréal, Quebec, cities with similar epidemiologic dynamics for seasonal influenza.10 Our specific objective was to estimate the relative effect of the 2006 US recommendation on influenza-related emergency department visits to Children’s Hospital Boston, with the Montreal Children’s Hospital as an untreated control.
Methods
Study period and settings
We examined emergency department visits for the seasons 2000/2001 through 2008/2009 at two large urban pediatric hospitals, Children’s Hospital Boston in Boston and Montreal Children’s Hospital in Montréal. Greater Boston has a population of 4.5 million, and the emergency department of Children’s Hospital Boston receives about 50 000 visits per year. Montréal has a greater metropolitan area with a population of 3.8 million. The emergency department of the Montreal Children’s Hospital receives about 65 000 visits per year. We analyzed visits in four pediatric age groups: 0–1, 2–4, 5–9 and 10–18 years. Our study period ended at the onset of the April 2009 outbreak of influenza A/H1N1.
Detailed data on the uptake of influenza vaccine are not available, but surveys provide estimates of self-reported vaccination coverage. The Canadian Community Health Survey11 indicated that in Quebec in 2005, 7% of children over 11 years of age and 25% of the overall population received a vaccine against seasonal influenza (http://cansim2.statcan.ca/). In Massachusetts in 2005, according to the National Immunization Survey,12 36% of age-eligible children (i.e., those 6–23 months old) were vaccinated (www.cdc.gov/vaccines/stats-surv/nis/). According to the Behavioural Risk Factor Surveillance System,13 in 2005, 30% of adult survey participants in Massachusetts reported having received an influenza vaccination in the previous 12 months (www.cdc.gov/brfss).
Data sources
Visits to emergency departments
Quantifying the burden of influenza is difficult, because of its nonspecific symptoms and the lack of routine laboratory confirmation for most patients. In young children, quantifying the burden of disease is particularly complicated because influenza in this age group is most routinely associated with visits to outpatient clinics and emergency departments, where it is often not recognized clinically.4 Previous studies have relied on measurements of influenza-like illness based on syndromic definitions that have been shown to be valuable indicators of influenza activity.3 We analyzed weekly visits to the emergency department of Children’s Hospital Boston that were related to influenza-like illness as captured by the AEGIS Flu system, part of the real-time syndromic surveillance system used by the Massachusetts Department of Public Health.14 AEGIS Flu is based on a set of respiratory illness–related chief complaints that have been previously described and validated.15 We retrospectively extracted from the electronic records of the Montreal Children’s Hospital a dataset defined by the set of chief complaint codes best matching those used by AEGIS Flu (Appendix 1, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.110241/-/DC1). The institutional review boards at Children’s Hospital Boston and Montreal Children’s Hospital approved use of the data for this study.
Virologic surveillance data
Because viral type and subtype have been associated with the intensity of influenza epidemics,16 we controlled for regional differences in the composition of circulating viruses. We used respiratory virus surveillance summaries reported by local authorities and published by the US Centers for Disease Control and Prevention to characterize the composition of influenza viruses circulating in New England and similar summaries published by the Public Health Agency of Canada to characterize the composition of viruses circulating in Quebec. We examined the proportion of isolates characterized as influenza type A (versus B), the proportion of influenza A viruses characterized as H3N2 (versus other A subtypes) and the proportion of characterized viruses that antigenically matched the strains included in that season’s trivalent influenza vaccine for the northern hemisphere. Weekly series of counts of influenza A and B isolates were transformed into relative counts. Other data were available as seasonal summaries. Seasonal summaries of all three indicators were used in the statistical analyses.
Statistical analyses
For each age group, we used Poisson regression models to estimate the effect of the 2006 policy change on emergency department visits related to influenza-like illness at the Children’s Hospital Boston relative to Montreal Children’s Hospital, which was used as the reference group. Each model included one indicator variable signifying whether the timing of the observation was before or after the US policy change (set at the start of the 2006/2007 influenza season, assumed to be the 35th week of 2006) and another indicator variable for the hospital. Each of these were modelled with a binary variable (for policy shift, before the start of the 2006/2007 influenza season = 0 and after the start of the 2006/2007 influenza season = 1; for hospital, Montreal Children’s Hospital = 0 and Children’s Hospital Boston = 1). The exponentiated coefficient of an additional term describing the interaction of these two indicator variables was calculated to assess relative differences in responses to the policy change between the two hospitals, expressed as rate ratios. We included total weekly all-cause counts of emergency department visits as model offset terms, to account for all-cause utilization of the emergency department.
We adjusted our multivariable models for other factors known to affect variation in influenza activity. In addition to the virologic variables described above, we included linear, quadratic and annual sine and cosine terms to model and adjust for temporal trends and seasonal cycles. We included indicator variables to adjust for fluctuations in utilization during the weeks of the Christmas and New Year holidays.
We assessed the goodness of fit using Akaike information criterion. In all models, we estimated standard errors using the heteroskedasticity and autocorrelation consistent estimator to account for temporal autocorrelation.17
Sensitivity analyses
We evaluated the sensitivity of our results to certain model assumptions. First, we reran the models after excluding, one at a time, individual influenza seasons, to assess whether the results we observed might have been driven by a single irregular season. Next, to evaluate the robustness of our results to our definition of influenza-like illness, we performed a separate analysis using a narrower, more specific definition that included only influenza, pneumonia, and cold, upper respiratory infection or congestion. Finally, we ran the analysis using hypothetical alternative policy change points at the start of each season, with the exception of 2000/2001, the first season in our study period. We used adjusted models in all sensitivity analyses.
Results
Virologic surveillance
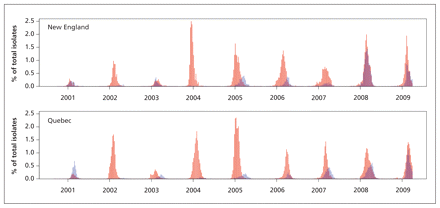
Patterns of virologic isolates were variable from season to season, yet were similar between the two regions. In both regions, influenza A predominated in all seasons except 2000/2001 (Appendix 2, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.110241/-/DC1). There was substantial influenza B activity in both New England and Quebec regions in all seasons except 2001/2002 and 2003/2004, and the timing of the epidemic peak was similar between regions for each virus type (Figure 1; see also Appendix 2, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.110241/-/DC1). Both New England and Quebec experienced little to no H3N2 activity in the 2000/2001 season, and both had an H3N2-dominant season in 2003/2004. In 2003/2004 and 2007/2008, the influenza vaccine for each season and region exhibited a poor match with strains circulating in both regions.
Time series of isolates of influenza A (orange bars) and influenza B (blue bars), as weekly percentage of all virologic isolates, in New England (top) and Quebec (bottom). Note: Where the orange and blue bars overlap, the colour appears purplish-red.
Emergency department visits related to influenza-like illness
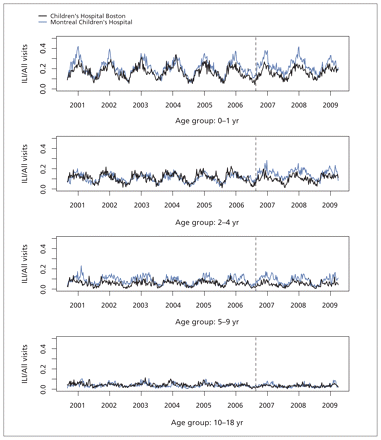
We analyzed 114 657 visits related to influenza-like illness, out of 1 043 989 total visits to the emergency departments of Children’s Hospital Boston and Montreal Children’s Hospital during our study period. Of these, 42 375 (37%) of the visits related to influenza-like illness and 456 371 (44%) of the all-cause visits were to Children’s Hospital Boston. The weekly series of visits for influenza-like illness as a proportion of all visits exhibited similar temporal dynamics: both hospitals had strong seasonal fluctuations in visits related to influenza-like illness in younger age groups, with more subtle seasonal patterns in older pediatric age groups and similar seasonal increases, declines and peak timing of the epidemic curves (Figure 2).
Weekly visits to the emergency departments of Children’s Hospital Boston and Montreal Children’s Hospital that were related to influenza-like illness (ILI), as a proportion of all-cause visits for four pediatric age groups. Dashed vertical line indicates the point at which US policy was changed to recommend vaccination for children 24 to 59 months of age.
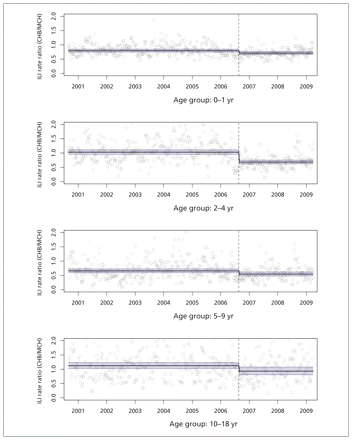
Following the policy change in the United States, we observed a decline in the rate of emergency department visits for influenza-like illness at Children’s Hospital Boston relative to the Montreal Children’s Hospital in the target age group, children two to four years old (unadjusted rate ratio 0.66, 95% confidence interval [CI] 0.51–0.85; adjusted rate ratio 0.66, 95% CI 0.58–0.75) (Table 1, Figure 3). The variation in both series and the potential for confounding due to differences in the composition of circulating strains motivated our effort to model and adjust for seasonal and virologic effects, but these factors did not change the overall result that we observed in this age group. However, the model’s goodness of fit was better after adjustment in this and all other age group models, so we report both unadjusted and adjusted results.
Model-based estimates of the effect of expanded recommendations for pediatric vaccination on visits to the emergency department for influenza-like illness at Children’s Hospital Boston relative to Montreal Children’s Hospital
Visits to the emergency department for influenza-like illness (ILI), as a proportion of all visits to the emergency department, expressed as the ratio of Children’s Hospital Boston (CHB) to Montreal Children’s Hospital (MCH). Circles = raw data, horizontal lines = adjusted model-based predictions (with 95% confidence interval, shaded lines), dashed vertical lines = point at which US policy was changed to recommend vaccination for children 24 to 59 months of age.
We also observed significant relative declines of 11%–18% for the nontarget age groups (Table 1; Figure 3) following the 2006 policy change in the United States, although this statement reflects the estimates of adjusted models only (the unadjusted models did not detect this effect).
Sensitivity analyses
We performed a series of sensitivity analyses on data for the target age group to evaluate the robustness of our results to model assumptions. Repeating the analysis after removal of individual seasons did not change the results qualitatively, which suggests that the effect we observed was not due to a single irregular season. Using a narrower definition of influenza-like illness for our study population, we observed a rate ratio for children two to four years old that was still significantly lower than 1 (adjusted rate ratio 0.48, 95% CI 0.40–0.58). Thus, our finding of an effect of the policy change is robust to our choice of definition for influenza-like illness. Repeating our analysis for eight hypothetical change points revealed that the maximum effect size was found with the change point set at the start of the 2006/2007 season.
Interpretation
We identified an association between the 2006 policy shift of the US Advisory Committee on Immunization Practices to recommend routine seasonal influenza vaccination to children two to four years old and an estimated 34% decline in visits related to influenza-like illness to Children’s Hospital Boston relative to Montreal Children’s Hospital, accompanied by a less pronounced decline in other pediatric age groups.
Preschool-aged children have been identified as important to the spread of influenza in households and communities, partly because of infection rates averaging 25%–43% per season, which is higher than in any other age group.18,19 Vaccination of healthy children is particularly effective because they mount excellent immune responses to the vaccine,20 making them desirable targets for vaccination when the goal is preventing the spread of disease. Therefore, it is possible that part of the reason for the decline that we observed in the nontarget age groups was due to reductions in household and community transmission from preschool-aged children to their contacts. Another explanation for this observation is that the Advisory Committee on Immunization Practices recommendation may have improved vaccination coverage among other pediatric age groups through vaccine uptake by the siblings of two- to four-year-old children or by promoting general awareness among health care providers and parents of the value of seasonal influenza vaccination for children.
Limitations
Our study had certain limitations. First, with data from only two pediatric medical centres, we were limited in our ability to make inferences regarding the effects of the US recommendations at a national scale. Second, influenza diagnoses are rarely confirmed by laboratory testing, which meant we had to rely on a syndromic definition of influenza-like illness to ascertain cases for this study, a method that has been shown to produce a valid estimate of influenza activity in the community.3 Repeating our analysis with a narrower definition of influenza-like illness resulted in a strengthening of the effect from a 34% reduction to a 52% reduction in emergency department visits for influenza-like illness at Children’s Hospital Boston relative to the Montreal Children’s Hospital, which suggests that our broad definition resulted in a conservative estimate of the effect. Third, our analysis would have benefited from data on vaccine uptake. Detailed data of this type are limited, but surveys have found that influenza vaccination coverage in two- to four-year-old children increased in the United States by about 49% between the 2005/2006 and 2006/2007 seasons and has remained stable since then.9,21 According to the same surveys, there have been increases in vaccine uptake in other pediatric and adult age groups since the 2005/2006 season, although the increases in coverage for these age groups have been more modest.9,21 Finally, there are several incongruencies between our two study populations that we did not adjust for directly, including demographic differences and differences between the health care systems in the United States and Canada that would have consequences for emergency department utilization. However, we know of no evidence that these effects might have changed over the course of our study period.
Conclusions
A number of factors influencing the observable trends in our data sets may be independent of the policy shift that was of interest for our study. Nonetheless, our findings provide evidence that, in our US study community (i.e., Boston), the recommendation of the Advisory Committee on Immunization Practices to routinely vaccinate preschool-aged children against seasonal influenza is improving pediatric influenza-related outcomes, as evidenced by reduction in emergency department visits. This adds to existing evidence that broadening vaccination recommendations toward inclusion of young children reduces the number of influenza cases and associated complications.22–29 Taken together, all of this evidence suggests that the recent expansion of the Advisory Committee on Immunization Practices recommendations on seasonal influenza vaccination is likely to result in additional reductions in influenza morbidity in the United States and that the adoption of parallel policies in Canada would be effective.
Acknowledgements:
The authors are grateful to Lucy Hadden, Aman Verma and Robert Rodrigues for database support and to Tracy Lieu, Jeffrey C. Kwong and Al Ozonoff for helpful discussions.
Footnotes
-
Competing interests: Caroline Quach has served as a consultant to Novartis regarding meningococcal B vaccines, and her institution has received a grant from Merck Frosst for a study of nosocomial rotavirus. No competing interests are declared by Anne Gatewood Hoen, David L. Buckeridge, Katia M.L. Charland, Kenneth D. Mandl or John S. Brownstein.
-
This article has been peer reviewed.
-
Contributors: Anne Gatewood Hoen, David L. Buckeridge, Katia M.L. Charland and John S. Brownstein conceived and designed the study and analyzed and interpreted the data; Kenneth D. Mandl and Caroline Quach contributed data; and Anne Gatewood Hoen drafted the article. All authors critically revised the article for important intellectual content and gave final approval of the version to be published.
-
Funding: This study was supported by the Canadian Institutes of Health Research (PAN-83152 and CAT-86857), the National Institutes of Health National Institute of Allergy and Infectious Diseases (1R21A1073591-01) and the National Library of Medicine (R01LM007677). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.