Abstract
Background: Apixaban (2.5 mg) taken twice daily has been shown to substantially reduce the risk of venous thromboembolism (VTE) compared with placebo for the primary thromboprophylaxis of ambulatory patients with cancer who are starting chemotherapy and are at intermediate-to-high risk of VTE. We aimed to compare the health system costs and health benefits associated with primary thromboprophylaxis using apixaban with those associated with the current standard of care (where no primary thromboprophylaxis is given), from the perspective of Canada’s publicly funded health care system in this subpopulation of patients with cancer over a lifetime horizon.
Methods: We performed a cost–utility analysis to estimate the incremental cost per quality-adjusted life-year (QALY) gained with primary thromboprophylaxis using apixaban. We obtained baseline event rates and the efficacy of apixaban from the Apixaban for the Prevention of Venous Thromboembolism in High-Risk Ambulatory Cancer Patients (AVERT) trial on apixaban prophylaxis. We estimated relative risk for bleeding, risk of complications associated with VTE treatment, mortality rates, costs and utilities from other published sources.
Results: Over a lifetime horizon, apixaban resulted in lower costs to the health system (Can$7902.98 v. Can$14 875.82) and an improvement in QALYs (9.089 v. 9.006). The key driver of cost–effectiveness results was the relative risk of VTE as a result of apixaban. Results from the probabilistic analysis showed that at a willingness to pay of Can$50 000 per QALY, the strategy with the highest probability of being most cost-effective was apixaban, with a probability of 99.87%.
Interpretation: We found that apixaban is a cost-saving option for the primary thromboprophylaxis of ambulatory patients with cancer who are starting chemotherapy and are at intermediate-to-high risk of VTE.
Patients with cancer are 4–7 times more likely to acquire venous thromboembolism (VTE) than the general population, 1 based on the hypercoagulable state associated with the cancer itself, patient characteristics and antineoplastic treatments. 2 Despite this increased risk for VTE, clinical guidelines do not recommend the use of primary thromboprophylaxis in unselected ambulatory patients with cancer,3–5 because this strategy has been associated with a small absolute reduction in symptomatic VTE and a nonstatistically significant trend in increased major bleeding events.6
The Khorana score uses the cancer type and individual patient characteristics to predict the risk of VTE in patients who are about to begin chemotherapy.7 This score has been evaluated prospectively for its capacity to identify patients with cancer who are at higher risk for VTE and, therefore, may be used to select those patients who are more likely to benefit from primary thromboprophylaxis.8,9 The 2019 Apixaban for the Prevention of Venous Thromboembolism in High-Risk Ambulatory Cancer Patients (AVERT) trial assessed the use of a low-dose direct oral factor Xa inhibitor (apixaban 2.5 mg twice daily) for primary thromboprophylaxis in ambulatory patients with cancer who were starting chemotherapy and were at intermediate-to-high risk of VTE (with a Khorana score ≥ 2).10 The study found that patients randomly assigned to apixaban had a significantly lower risk of VTE compared with placebo. The study also reported that apixaban was not associated with an increase in major bleeding during the on-treatment period. Subsequent to the published results of the AVERT trial and the Efficacy and Safety of Rivaroxaban Prophylaxis Compared with Placebo in Ambulatory Cancer Patients Initiating Systemic Cancer Therapy and at High Risk for Venous Thromboembolism (CASSINI) trial,11 clinical guideline recommendations were updated to endorse the consideration of primary thromboprophylaxis in high-risk ambulatory patients with cancer (Khorana score ≥ 2) before the start of chemotherapy. 4,5 Given the novelty of this recommendation and supporting data, individualized discussions regarding the risk of bleeding, expected benefits and overall costs are also encouraged.
To provide a better framework to support societal discussions on primary thromboprophylaxis in this patient population, we aimed to compare the health system costs and health benefits associated with the use of apixaban primary thromboprophylaxis with those associated with the current standard of care (where no primary thromboprophylaxis is given), from the perspective of Canada’s publicly funded health care system.
Methods
Study design and population
We conducted a cost–utility analysis that compared health system costs and clinically relevant outcomes relating to apixaban primary thromboprophylaxis with usual care (which did not include the use of primary thromboprophylaxis) among ambulatory patients with cancer who were starting chemotherapy and were at intermediate-to-high risk of VTE. Our analysis addressed the decision problem relating to whether apixaban should be reimbursed for this subpopulation of patients within Canada’s publicly funded health care system.
Consistent with the AVERT trial, our study population included ambulatory patients aged 18 years or older, with a new diagnosis or progression of cancer, who were starting chemotherapy and had a modified Khorana score of 2 or more (modified by the inclusion of myeloma and renal cancer as high-risk, and brain cancers as very high-risk cancer types).10 In the AVERT trial, patients were randomly assigned apixaban 2.5 mg twice a day or usual care (i.e., patients received only a placebo). Our primary efficacy outcome was the first major venous thromboembolic event (defined as a proximal deep vein thrombosis [DVT] or pulmonary embolism [PE]). Our primary safety outcome was major bleeding, as defined by the International Society on Thrombosis and Haemostasis (ISTH).12 Patients took apixaban for a median of 157 days and were followed for 180 days.
Model design and inputs
We used a decision tree and a Markov model (Figure 1) to simulate costs and outcomes for patients who received either apixaban for primary thromboprophylaxis or placebo over a lifetime horizon. We conducted scenario analyses to address the model’s structural uncertainty relating to the extrapolation of data from a trial with a limited follow-up period (6 mo in the AVERT trial) and the emerging use of direct oral Xa inhibitors for the treatment of cancer-associated thrombosis. Efficacy and costs associated with apixaban were applied over the trial duration (6 mo) for the base-case (lifetime model) and scenario analyses. To account for uncertainty, our model conservatively assumed that there was no difference in the risk of VTE for the 2 treatment arms beyond the trial period. We used a cycle length of 1 month based on the dosing schedules for the treatment of VTE and the expected duration of symptoms in an acute VTE event (before transitioning to more long-term symptoms in some patients). The model was run over a patient’s lifetime horizon (20.6 yr), based on the life expectancy in Canada.13
Model structure. Note: CRNMB = clinically relevant non-major bleeding, CTEPH = chronic thromboembolic pulmonary hypertension, ICH = intracranial hemorrhage, PTS = postthrombotic syndrome, VTE = venous thromboembolism. *Patients can transition to death at any point in the model because of age-specific mortality, cancer or complications.
We separated the model into 2 parts to better capture the dynamic risk of VTE in patients with cancer, and to incorporate differences in costs and risk of complications during different stages of the disease. In the first part, we described the disease pathway for patients with cancer who received primary thromboprophylaxis. The second part described the disease pathway for patients with cancer who had a first VTE event and who were receiving secondary thromboprophylaxis with anticoagulants (beyond the first month of treatment for cancer-associated thrombosis).
Our model included 17 discrete health states (Appendix 1, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.210523/tab-related-content) and was based on the following assumptions: patients received 1 chemotherapy regimen for 6 months before moving into complete remission; patients were assumed to be cured of cancer if they survived 5 years from the start of chemotherapy (meaning that their risk for VTE returns to that of the general population after 5 yr); primary thromboprophylaxis using apixaban was stopped if a first VTE or major bleeding event occurred; patients in the “first VTE” state received treatment-dose dalteparin (at 200 IU/kg) for cancer-associated thrombosis; patients in the “secondary thromboprophylaxis state without complications” continued to receive anticoagulation therapy with dalteparin over their remaining lifetime at a reduced dose (150 IU/kg; patients who had recurrent VTE received a dose escalation of dalteparin); and anticoagulation therapy with dalteparin was stopped if a patient had major bleeding or intracerebral hemorrhage. We chose these assumptions to best fit the current practice in the management of cancer-associated thrombosis3–5 and to coincide with the available evidence from the AVERT trial.
We identified transition probabilities, costs and utility values through a targeted literature review. The model structure and input parameters were validated by clinical experts to ensure that they coincided with current clinical practice. All costs were measured in 2020 Canadian dollars. Model input parameters are described in Table 1 and Appendix 1.
Model input parameters
We discounted costs and quality-adjusted life-years (QALYs) annually at a discount rate of 1.5%, which is recommended by the Guidelines for the economic evaluation of health technologies: Canada.61
Sensitivity analyses
We performed a series of sensitivity analyses to assess uncertainty in study results. A one-way sensitivity analysis was performed for event rates, costs, proportion of patients with VTE who were treated as inpatients, length of stay for VTE in hospital and utility values used in the model. We also conducted a probabilistic analysis for all parameters in the model using the Monte Carlo method with 10 000 iterations. We used the results of the probabilistic analysis to create a cost-effectiveness acceptability curve, which shows the probability of apixaban being cost-effective over a range of willingness-to-pay (WTP) thresholds.
Results
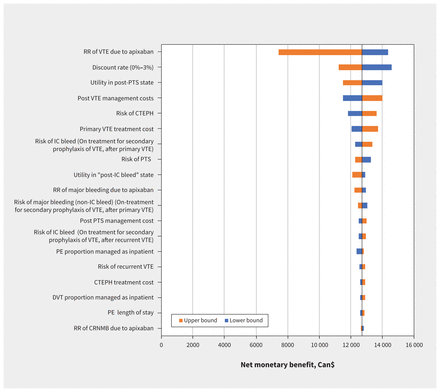
Over a lifetime horizon, we found that apixaban resulted in lower health system costs (Can$7902.98 v. Can$14 875.82) and an improvement in QALYs (9.089 v. 9.006) in patients with cancer who received chemotherapy, from the perspective of Canada’s health system (Table 2). The key driver of the cost-effectiveness results was the relative risk of VTE when receiving apixaban (Figure 2). Apixaban remained cost-effective compared with usual care across all 1-way sensitivity analyses.
Results from base-case analysis
One-way sensitivity analyses. Note: CRNMB = clinically relevant non-major bleeding, CTEPH = chronic thromboembolic pulmonary hypertension, DVT = deep vein thrombosis, ICH = intracranial hemorrhage, PE = pulmonary embolism, PTS = postthrombotic syndrome, RR = relative risk, VTE = venous thromboembolism.
In a cohort of 1000 patients, we found that apixaban resulted in fewer VTE events (57 v. 131), but an increase in major bleeding (129 v. 111) and clinically relevant non-major bleeding (1242 v. 1133) over a lifetime horizon.
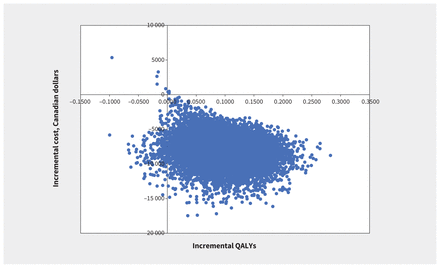
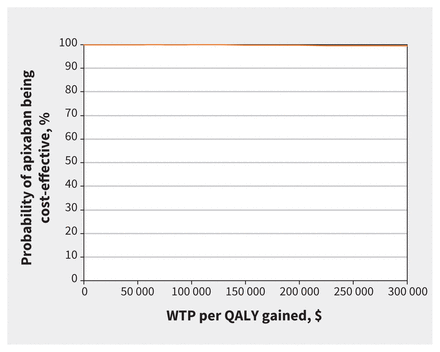
Results from the probabilistic analysis showed most iterations lead to cost savings and improved QALYs (Figure 3). At a commonly used WTP of Can$50 000 per QALY, the strategy with the highest probability of being most cost-effective was apixaban (probability of 99.87%; Figure 4).
Cost-effectiveness plane for base-case analysis. Note: QALY = quality-adjusted life-year.
Cost-effectiveness acceptability curve for primary analysis. Note: QALY = quality-adjusted life-year, WTP = willingness to pay.
Our results are robust to changes in time horizon and model assumptions. We performed a scenario analysis over the trial follow-up period of 6 months (scenario analysis 1). Over 6 months, apixaban was associated with a reduction in costs to the health system of Can$257.37 and an improvement in QALYs by 0.001 units. However, we observed wide confidence intervals around the mean incremental QALYs over a 6-month time horizon.
We also performed scenario analyses by varying the proportion of patients with cancer and a first VTE who received direct oral anticoagulants for treatment of cancer-associated thrombosis: 50% of patients received dalteparin and 50% of patients received edoxaban (scenario analysis 2), and 100% of patients received edoxaban (scenario analysis 3). Results from these scenario analyses were consistent with our base case results, showing that apixaban lowered health system costs and improved QALYs over a lifetime horizon in all scenarios (Appendix 2, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.210523/tab-related-content).
Interpretation
We found that the use of apixaban resulted in lower health system costs by Can$6973 and improved QALYs by 0.083 units (30.40 quality-adjusted life-days) compared with usual care. The primary driver of the results was the relative efficacy of apixaban. The relative safety of apixaban, costs and utility values had minimal affect on the cost-effectiveness findings. We also performed scenario analyses by varying the time horizon from lifetime to the trial follow-up period (6 mo) and the regimen used for the treatment of cancer-associated thrombosis. Apixaban remained dominant across all scenario analyses. However, owing to the limited data available about apixaban, we observed a high level of uncertainty around the estimates of cost-effectiveness for apixaban over the 6-month time horizon.
In 2020, a cost-effectiveness analysis involving a hypothetical cohort in the United States suggested that low-dose direct oral Xa inhibitors were cost-effective for the prevention of VTE in ambulatory patients with cancer who started chemotherapy and were at increased risk of VTE (Khorana score ≥ 2), with an incremental cost-effectiveness ratio of US$11 947 per QALY gained.32 This analysis was based on the pooled efficacy and safety results of both the AVERT and CASSINI trials. Our results are consistent with those from the US study. However, our cost-effectiveness analysis differs from the US study in several noteworthy ways. First, our analysis involved Canada’s publicly funded health care system. Therefore, costs and health system usage are reflective of this environment. Second, we made use of patient-level data from the AVERT trial, instead of summary results, which increases the specificity of our results for this patient population in Canada. Third, although combining the efficacy and safety results from the AVERT and CASSINI trials has the advantage of decreasing the uncertainty around these estimates, there are important differences between the 2 trials, which should lead to a more nuanced interpretation of each trial’s results and caution when combining their data. The patient populations in the 2 trials differed, most likely because of the use of the modified Khorana score in the AVERT trial and the Khorana score in the CASSINI trial to identify potential participants and the exclusion of patients with intracranial disease in the CASSINI trial. In addition, the CASSINI trial mandated the use of serial screening compression ultrasonography of the lower extremities throughout the follow-up period, which is not standard of care in ambulatory patients with cancer. The cost, impact on patients’ quality of life and most appropriate use of screening compression ultrasonography are unknown. Finally, the definition of an outcome was inconsistent between the 2 studies. Although both the AVERT and CASSINI trials used a composite outcome (by combining end points with varying degrees of clinical severity), the CASSINI trial included VTE events that were of lesser or unknown clinical significance, such as symptomatic distal or proximal DVT found on screening compression ultrasonography. These differences in patient populations, use of screening compression ultrasonography to identify asymptomatic DVT, and the divergence in composite outcomes to include less clinically significant and screening-detected events may account for the lack of significant efficacy of rivaroxaban in the CASSINI trial, which is contrary to the findings in the AVERT trial.
Although additional data on the optimal duration and effectiveness of primary thromboprophylaxis using apixaban are needed, our results support the coverage of low-dose apixaban by Canada’s publicly funded health care system for primary thromboprophylaxis in ambulatory patients with cancer who are starting chemotherapy and have a modified Khorana score of 2 or more. The prevention of VTE during the first 6 months after the start of chemotherapy translates to cost savings, which persist throughout the patient’s lifetime, by avoiding future complications including VTE, chronic complications of VTE and increased bleeding risk when receiving anticoagulation for the management of VTE. The use of apixaban for primary thromboprophylaxis appears to be a well-tolerated intervention, which further increases quality of life for patients with cancer. Given the importance and increasing costs associated with novel cancer treatment,62 strategies that alleviate the financial impact of providing high-quality patient-centred care to patients with cancer must be sought and adopted to promote the sustainability of the health care system. Primary thromboprophylaxis in appropriately chosen ambulatory patients with cancer appears to be such an intervention.
Limitations
There is uncertainty regarding the efficacy of apixaban beyond the trial follow-up period because data are lacking on long-term outcomes of thromboprophylaxis using apixaban. To account for this uncertainty, our model conservatively assumed that there was no difference in the risk of VTE for the 2 treatment arms beyond the trial period. In addition, there was a high level of uncertainty around the estimate of the relative safety of apixaban derived from the AVERT trial because of the small number of outcomes. Therefore, our study used synthesis-based estimates to supplement the patient-level data from the AVERT trial for the relative safety of apixaban by combining the major bleeding events from the AVERT and CASSINI trials that evaluated the use of oral anticoagulants for thromboprophylaxis among this patient population. This practice was justified because both trials used the same definitions for bleeding outcomes. Furthermore, we were unable to perform subgroup analysis by Khorana score or tumour type because the AVERT trial was not sufficiently powered to detect statistically significant differences by patient subgroups. Our study used data on resource use for the treatment of VTE, clinically relevant non-major bleeding and major bleeding derived from populations of patients without cancer. Previous research has shown that length of stay for treatment of VTE among patient populations with cancer is longer than that for those without cancer.63 This suggests that our finding of cost savings due to reduction in incidence of VTE attributable to apixaban may be an underestimation. In addition, we did not consider low-molecular-weight heparin as a comparator because of the lack of direct or indirect evidence comparing the efficacy of apixaban to low-molecular-weight heparin among ambulatory patients with cancer. Finally, our results are not generalizable to patients with cancer who have undergone surgery but have not received chemotherapy because only patients who started a new course of chemotherapy were eligible for the AVERT trial.
Conclusion
We found that within Canada’s publicly funded health system, primary thromboprophylaxis with apixaban in selected ambulatory patients with cancer is cost saving.
Footnotes
Competing interests: Marc Carrier has received consulting fees from Bayer, Sanofi, Servier, BMS, Pfizer, Leo Pharma and Valeo. Philip Wells has received personal fees in the form of payment or honoraria from the Bristol Myers Squibb (BMS)–Pfizer Alliance, Sanofi, Bayer Healthcare and Medscape. He is also the board director for the Bruyère Research Institute and Ottawa Department of Medicine Not-for-Profit Corporation. No other competing interests were declared.
This article has been peer reviewed.
Contributors: All of the authors contributed to the study’s concept and design, methodological choices and interpretation of the data. Miriam Kimpton and Srishti Kumar curated the data. Srishti Kumar, Miriam Kimpton, Doug Coyle and Kednapa Thavorn contributed to the formal analysis. Miriam Kimpton, Srishti Kumar and Kednapa Thavorn drafted the manuscript. All authors contributed substantially to the critical revision of the manuscript, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: Miriam Kimpton has received grants from Thrombosis Canada and the CanVECTOR network for her fellowship in adult thrombosis medicine (Canadian Institutes of Health Research [CIHR] CDT-142654). Marc Carrier has received grants from Bristol Myers Squibb (BMS), Pfizer and Leo Pharma. Philip Wells has received a grant from the BMS–Pfizer Alliance.
Data sharing: Our study is based on data from published literature and patient-level data from the AVERT trial. Data obtained from published literature have been reported in our study. Patient-level data from the AVERT trial can be requested from the first author (M.K.) upon request.
- Accepted June 25, 2021.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/