Multisystem inflammatory syndrome is an uncommon but severe complication primarily described in children and adolescents after infection with SARS-CoV-2; it can also occur in older individuals.
After recovery from SARS-CoV-2 infection, clinicians should suspect multisystem inflammatory syndrome in adults when a patient has prolonged fever, with multiorgan involvement.
Elevated inflammatory markers support the diagnosis.
Prompt initiation of therapy with immunomodulatory treatment can prevent severe outcomes.
A 60-year-old man presented to the emergency department with a 5-day history of mild shortness of breath, profound fatigue, anorexia and fever of up to 40°C. He also reported a lymph node enlargement over the left side of his neck, which had resolved 2 days before presentation. Four weeks earlier, he had tested positive for SARS-CoV-2 infection, confirmed by polymerase chain reaction (PCR) testing. He had no known comorbidities and had not received vaccination against SARS-CoV-2.
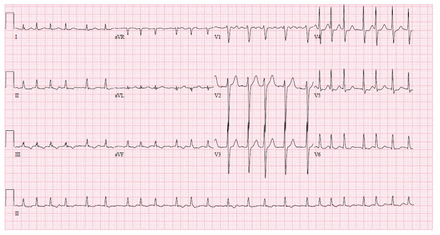
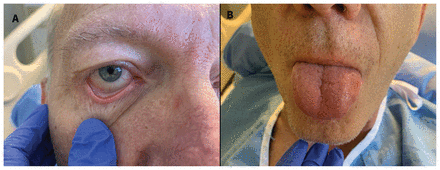
The patient’s heart rate was 150 beats/min, with new-onset atrial fibrillation. His blood pressure was 106/67 mm Hg and his oxygen saturation on room air was normal. His respiratory examination showed good air entry bilaterally, without crepitus, crackles or wheezing on auscultation. He had bilateral nonpurulent conjunctivitis (Figure 1A), erythema and enlargement of his tongue (Figure 1B), bilateral pitting edema, and erythema of the distal portion of his toes bilaterally (Figure 1C). Lesions were not associated with any vesicles, erosive features, crusting, fissures, warmth, swelling or tenderness. A chest radiograph showed right lower lobe opacification. An electrocardiogram showed atrial fibrillation with rapid ventricular response, as well as nonspecific diffuse ST-T wave abnormality (Figure 2). Computed tomography (CT) of the patient’s chest with contrast was negative for pulmonary embolism, but showed right heart enlargement and early pulmonary edema.
(A) Nonpurulent bilateral conjunctivitis in a 60-year-old man (only right eye shown). (B) Glossitis manifesting as nontender diffuse erythema and symmetric enlargement of the tongue. Small enanthem of the anterior third of the dorsum of the tongue (small erythematous bumps [arrow]) with hyperplastic fungiform and filiform papillae. (C) Left toes showing nonblanching maculopapular erythema. All images were taken before treatment was started.
Electrocardiogram showing ventricular rate of 143 beats/min, atrial fibrillation with rapid ventricular response and diffuse nonspecific ST-T wave abnormality.
The patient was admitted and started on empirical piperacillin–tazobactam for possible bacterial pneumonia as a superinfection of his previous SARS-CoV-2 infection. Initial laboratory workup was notable for elevated C-reactive protein, D dimer, ferritin and leukocytosis with neutrophilia (Table 11), indicating acute inflammation. His troponin level was elevated, and he had a substantially elevated pro–brain-type natriuretic peptide level. Blood cultures, urine culture and a respiratory viral panel were drawn and later returned negative. The patient also tested negative for HIV and hepatitis C and was immune to hepatitis B. An autoimmune workup collected before initiation of therapy was negative. COVID-19 immunoglobulin G serology was positive. A transthoracic echocardiogram, done after initiation of corticosteroids, showed normal sinus rhythm and normal right and left ventricular systolic and diastolic function with mild to moderate mitral regurgitation. There were no previous echocardiogram studies with which to compare these findings.
Laboratory data for 60-year-old man with multisystem inflammatory syndrome after SARS-CoV-2 infection*
Given the patient’s recent history of SARS-CoV-2 infection, fevers without localizing symptoms, oral mucosal changes, cervical lymphadenopathy, conjunctivitis and lower extremity changes, we suspected inflammatory post–COVID-19 syndrome. The presentation was similar to reported cases of an uncommon but severe complication in children and adolescents infected with SARS-CoV-2, called multisystem inflammatory syndrome in children (MIS-C), as well as to Kawasaki-like illness (Table 22–4). Both the pediatric and adult rheumatology teams involved in the patient’s care agreed that he met the preliminary Centers for Disease Control and Prevention (CDC) criteria for multisystem inflammatory syndrome in adults (MIS-A) and the criteria for complete Kawasaki disease, and he had multiple features consistent with MIS-C.
We started the patient on acetylsalicylic acid (ASA) and methylprednisolone and discontinued antibiotics. The next day, he received intravenous (IV) immunoglobulin. Within 24 hours of beginning treatment, he reported substantial improvement in his level of energy, shortness of breath and anorexia, with documented resolution of fever, improved conjunctivitis (Figure 3A), and decreased erythema of his tongue (Figure 3B), as well as reduction in inflammatory markers (Table 1). Repeat electrocardiogram was normal (Figure 4). The patient was discharged home 5 days after admission. Fourteen days after discharge, and again at a 6-week follow-up, he continued to improve clinically and biochemically (Table 1).
Near-complete resolution of the patient’s conjunctivitis (A) and improvement in glossitis, and near-complete resolution of enanthem of the tongue (B) on day 1 after initiation of high-dose steroids.
Sinus bradycardia of 54 beats/min and otherwise normal electrocardiogram with resolution of atrial fibrillation and ST-T changes prior to discharge.
Discussion
Since the beginning of the SARS-CoV-2 pandemic, new information has been emerging on the trajectory of the disease. Examples of complications include venous thromboembolism, cardiovascular disease, acute kidney or liver injury, neurologic symptoms and post–COVID-19 syndrome.5 Multisystem inflammatory syndrome in children was first described in April 2020 as a hyperinflammatory syndrome with features resembling Kawasaki disease.2,3,6 As of May 3, 2021, 3742 children in the United States had been formally diagnosed with MIS-C according to the criteria outlined in Table 2, with 35 related deaths.7
In October 2020, the CDC published a review of 27 adult cases that fit the description of a multisystem inflammatory syndrome. 4 The preliminary case definition of MIS-A is shown in Table 2, and cases described to date have been in patients younger than 50 years. As we continue to learn about MIS-A, however, it is prudent not to assume any age limitation when considering the diagnosis, as our case suggests.4,8–10
Many questions about MIS-A remain unanswered. Our patient did not present with the more commonly reported symptoms of stroke, shock or cardiac dysfunction requiring management in the intensive care unit.4 This may be explained by our patient having a milder spectrum illness or by the prompt diagnosis and interventions he received. Several features of our patient’s presentation raised early concern for MIS-A or a Kawasaki-like illness. Our patient met the diagnostic criteria for complete Kawasaki disease (5 d of fever and 4 out of 5 clinical features: extremity changes, conjunctivitis, oral changes and cervical lymphadenopathy; our patient did not have a rash) and MIS-C (except for age). However, evidence of recent SARS-CoV-2 infection and the age of the patient made MIS-A more likely as the cause.
The American College of Rheumatology published a review comparing and contrasting features between MIS-C and Kawasaki disease, and it reported that about 25%–50% of patients who fulfilled criteria for MIS-C also met the full diagnostic criteria for Kawasaki disease.11 Two differences between the conditions were ethnic distribution (highest among people of Japanese descent in patients with Kawasaki disease) and age distribution (usually younger than 5 yr for Kawasaki disease, and a broader range, from 3 months to 21 yr, for MIS-C).2,11,12
Standardized treatment for MIS-A is yet to be established. Our patient improved with 3 days of methylprednisolone 125 mg/d, then an oral prednisone taper of 50 mg/d for 3 days, followed by a tapering dose decreasing by 10 mg/d every 3 days until he was weaned off steroids (for a total oral course of 15 d). He received IV immunoglobulin 1 g/kg/d for 2 days and oral ASA 325 mg/d until clinical resolution, as evidenced by absence of fevers and normal C-reactive protein. This treatment was based on expert opinion and up-to-date evidence on Kawasaki disease and MIS-C.2,6,8,11–13 The American College of Rheumatology recommends a step-wise approach to immunomodulatory treatment in MIS-C with IV immunoglobulin or glucocorticoids considered as first-tier agents,11 and the American Heart Association recommends high-dose immunoglobulin (2 g/kg) as a single IV dose for treatment of Kawasaki disease.2 Although the patient did not have clinically significant thrombocytosis, we prescribed a short course of high-dose ASA to prevent thrombosis and coronary artery aneurysms.11
The optimal follow-up for MIS-A is currently unknown, but experts support monitoring for coronary artery dilatation and aneurysm, as with MIS-C-or Kawasaki-like illness.2,11 Unlike in children, for whom echocardiography is the imaging choice for coronary artery diameter, the visualization of coronary arteries becomes progressively more difficult as body size increases, making CT coronary angiography preferred for surveillance in adults.2 Our patient’s coronary angiogram showed triple-vessel coronary artery disease without clinically important stenosis and no coronary aneurysms or findings suggestive of coronary artery vasculitis. At time of writing, a repeat CT to visualize coronary artery calibre is still pending for 6 months after discharge.
Unlike for MIS-C, there is currently no requirement to report cases of MIS-A to provincial or state authorities, but this should be encouraged to facilitate research and improve patient outcomes.6
The section Cases presents brief case reports that convey clear, practical lessons. Preference is given to common presentations of important rare conditions, and important unusual presentations of common problems. Articles start with a case presentation (500 words maximum), and a discussion of the underlying condition follows (1000 words maximum). Visual elements (e.g., tables of the differential diagnosis, clinical features or diagnostic approach) are encouraged. Consent from patients for publication of their story is a necessity. See information for authors at www.cmaj.ca.
Acknowledgements
The authors acknowledge Drs. Glen Hazlewood, Heinrike Schmeling and Kevin Fonseca for their expert opinion in the management of this case.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
The authors have obtained patient consent.
Contributors: Both authors contributed to the conception and design of the work, drafted the manuscript, revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/