Abstract
BACKGROUND Nonnutritive sweeteners, such as aspartame, sucralose and stevioside, are widely consumed, yet their long-term health impact is uncertain. We synthesized evidence from prospective studies to determine whether routine consumption of non-nutritive sweeteners was associated with long-term adverse cardiometabolic effects.
METHODS We searched MEDLINE, Embase and Cochrane Library (inception to January 2016) for randomized controlled trials (RCTs) that evaluated interventions for nonnutritive sweeteners and prospective cohort studies that reported on consumption of non-nutritive sweeteners among adults and adolescents. The primary outcome was body mass index (BMI). Secondary outcomes included weight, obesity and other cardiometabolic end points.
RESULTS From 11 774 citations, we included 7 trials (1003 participants; median follow-up 6 mo) and 30 cohort studies (405 907 participants; median follow-up 10 yr). In the included RCTs, nonnutritive sweeteners had no significant effect on BMI (mean difference −0.37 kg/m2; 95% confidence interval [CI] −1.10 to 0.36; I2 9%; 242 participants). In the included cohort studies, consumption of nonnutritive sweeteners was associated with a modest increase in BMI (mean correlation 0.05, 95% CI 0.03 to 0.06; I2 0%; 21 256 participants). Data from RCTs showed no consistent effects of nonnutritive sweeteners on other measures of body composition and reported no further secondary outcomes. In the cohort studies, consumption of nonnutritive sweeteners was associated with increases in weight and waist circumference, and higher incidence of obesity, hypertension, metabolic syndrome, type 2 diabetes and cardiovascular events. Publication bias was indicated for studies with diabetes as an outcome.
INTERPRETATION Evidence from RCTs does not clearly support the intended benefits of nonnutritive sweeteners for weight management, and observational data suggest that routine intake of nonnutritive sweeteners may be associated with increased BMI and cardiometabolic risk. Further research is needed to fully characterize the long-term risks and benefits of nonnutritive sweeteners. Protocol registration: PROSPERO-CRD42015019749
Obesity is a major public health challenge that contributes to type 2 diabetes and cardiovascular disease.1 Evidence that sugar consumption is fuelling this epidemic2–4 has stimulated the increasing popularity of nonnutritive sweeteners,5 including aspartame, sucralose and stevioside. In 2008, more than 30% of Americans reported daily intake of non-nutritive sweeteners, and this proportion is increasing.6 Researchers have suggested that nonnutritive sweeteners may have adverse effects on glucose metabolism, gut microbiota and appetite control.7,8 Moreover, studies involving animals have reported that chronic exposure to nonnutritive sweeteners leads to increased food consumption, weight gain and adiposity.9
The position of the Academy of Nutrition and Dietetics is that nonnutritive sweeteners can help limit energy intake as a strategy to manage weight or blood glucose.10 However, consumption of nonnutritive sweeteners has been paradoxically associated with weight gain and incident obesity.7,11 A previous meta-analysis12 reported conflicting evidence: randomized controlled trials (RCTs) showed potential benefits (modest weight loss), whereas observational studies showed a small but significant association with increased body mass index (BMI). However, the review did not evaluate outcomes beyond body composition.13 Several studies involving more than 100 000 new participants and representing several new geographic settings have since been published.14–24
Our objective was to synthesize evidence addressing this question: Is routine consumption of nonnutritive sweeteners by adults and adolescents associated with adverse long-term cardiometabolic effects in RCTs and prospective cohort studies?
Methods
This review is reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses25 following a registered protocol.26
Search strategy and selection criteria
The search strategy was developed by an information specialist (M.F.) to overcome the limitations13 of previous reviews. Our MEDLINE strategy (Appendix 1, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161390/-/DC1, Table S1) was peer reviewed and also translated for searches in Embase and The Cochrane Central Register of Controlled Trials. We included the following terms, among others: nonnutritive sweeteners, aspartame, saccharin, sucralose, xylitol, stevia, carbonated beverages, calories and food frequency. We did not limit the search by using terms related to outcomes of interest.
We conducted the searches from the time of database inception to January 2016 with no language restrictions; translation services were accessed to evaluate non-English citations. We also searched conference proceedings from the American Society for Nutrition, American Diabetes Association and Obesity Society. We manually searched reference lists of pertinent reviews and included studies for relevant citations, and we conducted grey literature searches of OpenSIGLE and Google Scholar. We used EndNote (version X6, Thompson Reuters, New York) to perform reference management.
We screened search results in duplicate using a team of 5 reviewers (A.M., A.R., J.L., L.C., M.J.). We included RCTs and observational studies that evaluated consumption of nonnutritive sweeteners in individuals who were more than 12 years of age (Appendix 1, Table S2). Studies evaluating children were reviewed separately.27 We required a minimum study duration of 6 months to reflect routine consumption of nonnutritive sweeteners, to focus on long-term effects and to allow time for metabolic outcomes to develop. For observational studies, we required that associations with baseline intake of nonnutritive sweeteners (not only changes in intake during the course of the study) were reported to confirm temporality and limit confounding by reverse causation. Our primary outcome was change in BMI.
Secondary outcomes included changes in body weight; adiposity; glucose metabolism; and incidence of overweight/obesity, metabolic syndrome, type 2 diabetes, hypertension and other cardiorenal outcomes. If a study reported outcomes at multiple time points, we included the longest available follow-up.
Data extraction
We developed, piloted and deployed a standardized form for data extraction in DistillerSR (version 2, Evidence Partners Inc., Ottawa). A team of 5 reviewers (A.A., B.C., R.R., L.C., M.A.) independently extracted study data in duplicate that included baseline characteristics; interventions for nonnutritive sweeteners and comparators (for trials) or consumption of nonnutritive sweeteners and confounders or covariates (for cohorts); type, dose and duration of exposure to nonnutritive sweeteners; duration of follow-up; and cardiometabolic outcomes. For RCTs, we preferentially extracted data from intention-to-treat analyses or requested the data from authors. For cohorts, we extracted adjusted effect estimates in 2 formats: ratios comparing the highest versus lowest category of nonnutritive sweetener intake, and beta estimates quantifying linear associations per unit of nonnutritive sweetener intake. If multiple adjusted estimates were reported, we extracted the estimate from the statistical model that included the largest number of covariates. Data that were presented in nonextractable formats were requested from authors.
Assessment of study quality
Four reviewers (M.A., J.L., L.C., B.C.) assessed potential bias in RCTs using the Cochrane Collaboration Risk of Bias tool29,30 and evaluated the quality of cohort studies using the 9-point Newcastle–Ottawa Scale.31 Based on previous research32,33 we designated 2 critical confounders for cohort studies: baseline body composition (BMI or other measure of body composition) and diet quality (total energy or sugar intake, or a diet pattern or quality score).
Statistical analysis
For the meta-analysis of continuous outcomes, we calculated mean differences (MD) or standardized MDs. For binary outcomes, we calculated pooled odds ratios (ORs), risk ratios (RRs) or hazard ratios (HRs), and 95% confidence intervals (CIs). When nonnutritive sweetener intake units differed between cohort studies, we converted β estimates to t values (β/standard error) to generate a unitless metric28 and calculated the pooled mean correlation. Subgroup analyses were planned a priori to explore heterogeneity and determine associations in prespecified strata. We conducted the analyses with random-effects models using Comprehensive Meta-Analysis Software (version 2.2.064) or Rev-Man (version 5.3.5). Statistical heterogeneity was quantified using the I2 statistic. We assessed publication bias using funnel plots, and the trim and fill method.
Results
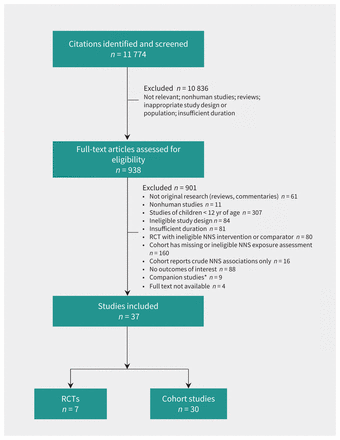
From 11 774 citations, we assessed 938 full-text articles for eligibility, and 37 studies involving a total of 406 910 individuals met our inclusion criteria: 7 RCTs19,20,34–38 and 30 cohort studies14–18,21–24,39–60 (Figure 1).
RISMA flow diagram. NNS = nonnutritive sweetener, RCT = randomized controlled trial. *Companion studies included abstracts, trial registrations and earlier reports from included studies.
The 7 RCTs enrolled a total of 1003 participants who were obese,38 overweight19,20,34,35 or hypertensive36,37 (Table 1). The interventions for nonnutritive sweeteners included beverages sweetened with aspartame or unspecified nonnutritive sweeteners,19,20,34,35 stevioside capsules36,37 or consumption of aspartame at the discretion of the participant.38 The duration of interventions ranged from 6 to 24 months (median 6 mo, interquartile range [IQR] 6–14). Most RCTs were at unclear or high risk of bias (Table 1 and Appendix 1, Table S3).
Randomized controlled trials that evaluated nonnutritive sweetener interventions and long-term cardiometabolic health
The 30 observational studies reported outcomes from 22 distinct cohorts involving a total of 405 907 individuals (Table 2). Most of the studies used food frequency questionnaires to evaluate beverages containing nonnutritive sweeteners. More than 85% controlled for baseline body composition, diet quality, age, sex, smoking and physical activity, whereas less than 50% controlled for ethnicity and socioeconomic status (Appendix 1, Table S4). The duration of follow-up ranged from 1 to 38 years (median 10 yr, IQR 6–22). Most cohort studies were of moderate quality (Table 2 and Appendix 1, Table S5).
Prospective cohort studies evaluating intake of nonnutritive sweetener and long-term cardiometabolic health
Primary outcome: body mass index
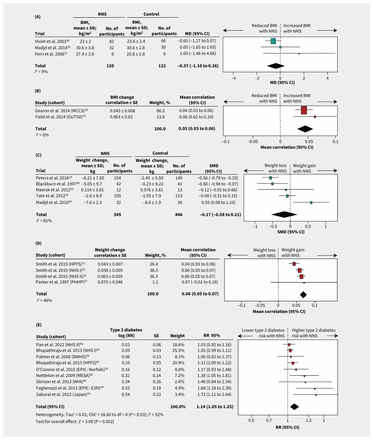
Two RCTs involving hypertensive participants who were taking stevioside capsules36,37 and 1 RCT involving participants who were overweight and consuming artificially sweetened beverages20 showed no significant effect on BMI over 6 to 24 months (MD −0.37 kg/m2, 95% CI −1.10 to 0.36; I2 9%; 3 trials; 242 participants; Table 3, Figure 2A). Two cohort studies that reported continuous nonnutritive sweetener intake in healthy participants14,15 showed a positive correlation with BMI over 3 to 13 years (mean correlation 0.05, 95% CI 0.03 to 0.06; I2 0%; 2 cohorts; 21 256 participants; Table 3, Figure 2B). A third cohort study that reported quantiles of nonnutritive sweetener intake50 found that participants who consumed nonnutrive sweeteners daily had a greater increase in BMI during 8 years of follow-up than those who did not consume them (MD 0.77 kg/m2, 95% CI 0.47 to 1.07 for daily v. no intake; 3371 participants). Overall, there was limited evidence for the effect of nonnutritive sweeteners on BMI, with 3 long-term cohort studies suggesting a modest increase in BMI that was not confirmed in 2 RCTs. The limited number of eligible studies precluded subgroup analyses.
Results from meta-analyses (where possible) or individual studies for intake of nonnutritive sweeteners and long-term cardiometabolic health outcomes in randomized controlled trials and cohort studies
Forest plots of consumption of NNS and selected cardiometabolic health outcomes. (A) Differences in mean BMI between NNS consumption and control groups for RCTs. A value less than 0 represents reduced BMI with NNS consumption. (B) Correlaton of BMI change per unit of NNS intake for cohort studies. A value less than 0 represents a reduced BMI. (C) Standard mean differences in weight between NNS consumption and control groups for RCTs. A value less than 0.0 represents weight loss. (D) Correlation of weight change per unit NNS intake for cohort studies. A value less than 0 favours weight loss. (E) Incidence of type 2 diabetes for highest versus lowest quantiles of NNS intake in cohort studies. A value less than 1.0 represents a lower risk of type 2 diabetes. Additional outcomes are shown in Table 3, and Appendix 1, Figures S1–4. Squares represent effect estimates within each study, with 95% CIs represented by horizontal lines. Square size is proportional to the weight of each study. Diamonds represent the weighted mean effect estimates. Cohort acronyms are defined in Table 2. Note: BMI = body mass index, CI = confidence interval, MD = mean difference, NNS = nonnutritive sweetener, RCT = randomized controlled trial, RR = risk ratio, SD = standard deviation, SE = standard error, SMD = standardized mean difference.
Secondary outcomes
Weight
Among 5 RCTs evaluating interventions using nonnutritive sweeteners in participants who were obese,19,20,34,35,38 there was no consistent effect on change in weight (standardized MD −0.17; 95% CI −0.54 to 0.21; I2 81%; 5 trials; 791 participants) (Table 3, Figure 2C). Heterogeneity across the 5 trials was partially explained by differences in study duration: 2 longer trials19,38 showed significant weight loss over 16 to 24 months of the intervention (standardized MD −0.55, 95% CI −0.75 to −0.34; I2 0%; 2 trials), and 3 shorter (6 mo) trials20,34,35 showed no effect for the use of nonnutritive sweeteners (standardized MD 0.13, 95% CI −0.34 to 0.59; I2 65%; 3 trials) (p for subgroup differences = 0.009; Appendix 1, Table S6). Weight-loss effects also tended to be stronger in RCTs with industry sponsorship19,34,38 (standardized MD −0.37; 95% CI −0.71 to −0.03; I2 77%; 3 trials) compared with RCTs that were not funded by industry20,35 (standardized MD 0.30, 95% CI −0.38 to 0.99; I2 55%; 2 trials) (p for subgroup differences = 0.09; Appendix 1, Table S6). Notably, both longer-term RCTs were funded by industry,19,38 making it impossible to isolate the effect of trial duration and industry sponsorship in subgroup analyses. In addition, all 5 RCTs that evaluated weight change were at high risk of bias, prohibiting subgroup analyses according to this metric.
Two observational studies reported on intake of nonnutritive sweeteners and subsequent weight change in 4 cohorts over periods of 2 to 4 years21,57 (Table 3, Figure 2D). There was a significant positive correlation between intake of nonnutritive sweeteners and weight gain (weighted mean correlation 0.06, 95% CI 0.05 to 0.07; I2 46%; 4 cohorts; 32 405 participants) (Table 3).
Adiposity and overweight
Three RCTs involving participants who were obese and consuming diet soda as part of a weight-loss program reported inconsistent effects on waist circumference (standardized MD −0.16; 95% CI −0.56 to 0.25; I2 83%; 3 trials; 683 participants) (Table 3, Appendix 1, Figure S1A). Heterogeneity across studies was related to the duration of intervention, with one 12-month trial showing a significant reduction in waist circumference19 and two 6-month interventions finding no effect20,34 (p for subgroup differences 0.001). One 6-month trial reported no effect on percentage of body fat.35
In contrast to RCTs, cohort studies with 4 to 9 years of follow-up showed that higher intake of nonnutritive sweeteners was associated with increasing waist circumference (MD 2.27 cm, 95% CI 0.96 to 3.58; 1 cohort; 384 participants)18 (Table 3), higher incidence of abdominal obesity (OR 1.59, 95% CI 1.23 to 2.07; 1 cohort; 5011 participants)60 (Table 3) and higher incidence of overweight (OR 1.84, 95% CI 1.28 to 2.66 for highest v. lowest intake quantiles; I2 0%; 3 cohorts; 7917 participants)22,50,59 (Table 3 and Appendix 1, Figure S1B).
Metabolic outcomes
Incidence for metabolic syndrome and type 2 diabetes was not reported in the RCTs. Pooled data from cohort studies with 4 to 24 years of follow-up showed higher risk of metabolic syndrome (RR 1.31, 95% CI 1.23 to 1.40; I2 0%; 5 cohorts; 27 914 participants)39,47,48,54,60 (Table 3 and Appendix 1, Figure S2A) and type 2 diabetes (RR 1.14, 95% CI 1.05 to 1.25; I2 52%; 9 cohorts; 400 571 participants)16,24,42,49,55,56,58,60 for the highest versus lowest quantiles of nonnutritive sweetener intake (Table 3, Figure 2E). In subgroup analyses, heterogeneity was not explained by baseline weight status, study quality, duration of follow-up or dose of nonnutritive sweeteners (Appendix 1, Table S7). Among 4 cohorts that reported continuous effect estimates, we found a 3% higher relative risk of type 2 diabetes per additional daily serving of nonnutritive sweetener (RR 1.03, 95% CI 1.01 to 1.05; I2 0%; 4 cohorts; 221 363 participants)24,42,53,56 (Table 3 and Appendix 1, Figure S2B). We found no statistically significant associations for insulin resistance (3 trials; Appendix 1, Figure S3), glycosylated hemoglobin (1 trial), glucose tolerance (1 cohort) or gestational diabetes (1 cohort) (Table 3).
Cardiorenal outcomes
Cardiorenal outcomes were not reported in the RCTs. Among cohort studies, we found that high nonnutritive sweetener intake was associated with a higher risk of hypertension over 5 to 38 years of follow-up (HR 1.13, 95% CI 1.06 to 1.20; I2 64%; 5 cohorts; 232 630 participants)45,48,60 (Table 3 and Appendix 1, Figure S4A). In addition, high intake of nonnutritive sweetener was associated with a higher risk of stroke (RR 1.14, 95% CI 1.04 to 1.26; I2 0%; 2 cohorts; 128 176 participants)40 and cardiovascular events (RR 1.32; 95% CI 1.15 to 1.52; I2 0%; 2 cohorts; 62 178 participants),17,52 whereas there was no significant association with coronary heart disease (RR 0.98; 95% CI 0.90 to 1.07; I2 0%; 2 cohorts; 131 403 participants)46,51 (Table 3 and Appendix 1, Figures S4B–4D).
Publication bias
Because of the limited number of studies, we could not assess publication bias for most outcomes, with the exception of type 2 diabetes. Although the pooled RR from 9 published studies that reported incident type 2 diabetes in high versus low consumers of nonnutritive sweeteners was significant (RR 1.14, 95% CI 1.05 to 1.23), it was attenuated to 1.07 (95% CI 0.97 to 1.18) after imputing missing studies (Appendix 1, Figure S5). This suggests potential publication bias that favours studies reporting a positive association between nonnutritive sweetener consumption and type 2 diabetes.
Interpretation
Evidence from small RCTs with short follow-up (median 6 mo) suggests that consumption of nonnutritive sweeteners is not consistently associated with decreases in body weight, BMI or waist circumference. However, in larger prospective cohort studies with longer follow-up periods (median 10 yr), intake of nonnutritive sweeteners is significantly associated with modest long-term increases in each of these measures. Cohort studies further suggest that consumption of nonnutritive sweeteners is associated with higher risks of obesity, hypertension, metabolic syndrome, type 2 diabetes, stroke and cardiovascular disease events; however, publication bias was indicated for type 2 diabetes, and there are no data available from RCTs to confirm these observations.
Previous reviews12,65 concluded that, although data from RCTs support weight-loss effects from sustained nonnutritive sweetener interventions, observational studies provide inconsistent results. Building on these findings, we included new studies14–24 and found that consumption of nonnutritive sweeteners was not generally associated with weight loss among participants in RCTs, except in long-term (≥ 12 mo) trials with industry sponsorship. In addition, we found that consumption of nonnutritive sweeteners was associated with modest long-term weight gain in observational studies. Our results also extend previous meta-analyses that showed higher risks of type 2 diabetes32,33 and hypertension66 with regular consumption of nonnutritive sweeteners.
Our results highlight both the value and challenge of incorporating observational studies when examining the effect of real-world exposures on health outcomes that develop slowly over time. Although RCTs provide the highest quality of scientific evidence, they often fail to recapitulate chronic dietary exposures that are captured in decades-long cohort studies. However, it is not uncommon for hypotheses based on observational evidence to fail when tested in RCTs,67 and these data should therefore be interpreted with caution.
Strengths of our systematic review include use of a registered protocol and sensitive, peer-reviewed search strategy. We synthesized evidence from both RCTs and observational studies, assessed multiple cardiometabolic outcomes and focused on long-term effects.
Limitations
The main limitation of our review is the unavoidable grouping of exposure and outcome variables. We could not evaluate different types or formulations of nonnutritive sweeteners because most studies did not report this information, and we could not assess dose effects owing to the limited number of RCTs and the semi-quantitative nature of the reporting of nonnutritive sweetener intake in cohort studies. In addition, some cardiometabolic outcomes could not be evaluated individually becuse of the way they were combined and reported in the original studies (e.g., “overweight and obesity,” “cardiovascular events”). Finally, meta-analysis was not always possible because of reporting differences and the paucity of eligible studies.
The individual studies included in our review also have limitations. Most RCTs were at high risk of bias, and most cohort studies achieved only moderate quality scores. In the cohort studies, the ascertainment of exposure to nonnutritive sweeteners by self-report was likely incomplete,6 and the comparison of extreme intake quantiles may have yielded biased results. Furthermore, these studies evaluated consumption of artificially sweetened beverages before 2004; however, nonnutritive sweeteners are increasingly found in other foods, and consumption has increased considerably in recent years.6
Observational studies are also subject to confounding bias, particularly when the exposure (e.g., nonnutritive sweeteners) is a potential “treatment” for the outcomes under investigation. However, critical confounders (baseline body composition and diet quality) were largely accounted for in the included studies, and we limited confounding by reverse causation by including only prospective studies that documented intake of nonnutritive sweeteners before weight change and disease incidence.
Randomized controlled trials of nonnutritive sweetener interventions also have known limitations.68 All were relatively short in duration, and the majority were conducted as part of multifaceted weight loss programs in obese individuals, which does not address routine consumption of nonnutritive sweeteners by healthy individuals. In addition, some trials evaluated nonnutritive sweeteners in capsule form, which may alter their physiologic effects, while others were subject to potential bias from lack of blinding and industry sponsorship. Finally, several studies focused on BMI and waist circumference, which are imperfect indices of body composition, despite being established predictors of cardiovascular disease.69,70
Conclusion
Evidence from RCTs does not clearly support the intended benefits of nonnutritive sweeteners for weight management. In contrast, observational data suggest that routine consumption of nonnutritive sweeteners may be associated with a long-term increase in BMI and elevated risk of cardiometabolic disease; however, these associations have not been confirmed in experimental studies and may be influenced by publication bias. New studies are needed to compare different types and formulations of nonnutritive sweeteners, and to evaluate the net effect of substituting nonnutritive sweeteners for sugar. Improved assessment tools and biomarker approaches71 should be used to accurately capture consumption of nonnutritive sweeteners, and confounding bias must be carefully addressed. Given the widespread and increasing use of nonnutritive sweeteners, caution is warranted until the long-term risks and benefits of these products are fully characterized.
Acknowledgements
The authors thank information specialist consultant Becky Skidmore for her critical review of the search strategy. They also thank the following study authors for contributing additional unpublished data: Maira Bes-Rastrollo and colleagues (Seguimiento Universidad de Navarra [SUN] cohort), Emma Gearon and colleagues (Melbourne Collaborative Cohort Study [MCCS] cohort), and Esther Lopez-Garcia and colleagues (Study on Nutrition and Cardiovascular Risk in Spain [ENRICA] cohort).
Footnotes
Competing interests: Jonthan McGavock has received speaker fees from Medtronic. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Meghan Azad conceptualized and coordinated the study. Meghan Azad and Ashleigh Reid drafted the initial protocol. Michelle Fiander developed the search strategy. Ashleigh Reid, Justin Lys, Leslie Copstein, Amrinder Mann and Maya Jeyaraman screened citations and assessed studies for eligibility. Rasheda Rabbani, Bhupendrasinh Chauhan, Ahmed Abou-Setta, Leslie Copstein and Meghan Azad extracted data. Meghan Azad, Justin Lys, Leslie Copstein and Bhupendrasinh Chauhan performed quality assessments. Rasheda Rabbani performed statistical analyses. Dylan MacKay, Jon McGavock and Brandy Wicklow provided content expertise in nutrition and metabolic health. Ryan Zarychanski, Bhupendrasinh Chauhan and Ahmed Abou-Setta provided methodologic expertise in knowledge synthesis and resolved disagreements regarding study eligibility or quality assessments. Dylan MacKay, Jon McGavock, Brandy Wicklow, Ryan Zarychanski, Bhupendrasinh Chauhan and Ahmed Abou-Setta critically reviewed the manuscript for important intellectual content. All of the authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: No funding was specifically obtained for this study. Ryan Zarychanski received a New Investigator Award from the Canadian Institutes of Health Research (CIHR). Jon McGavock holds the CIHR Applied Public Health Chair in Resilience and Childhood Obesity. CIHR had no role in the design, conduct or reporting of the study.
- Accepted March 10, 2017.