Anal cancer is generally uncommon,1 but some groups at high risk have higher prevalence. It is usually caused by the human papillomavirus (HPV) and can occur along with other HPV-related cancers in the same patient or in his or her sexual partners. Anal cancer is potentially preventable, and precancers can be detected and treated in the office.
There were 580 cases of anal cancer in Canada in 2010, 195 (33.6%) of which occurred in men.1 Although anal cancer is more common in women,1,2 high rates of the disease are developing among people with HIV infection, particularly among men who have sex with men.3,4
Similar to cervical cancer, 80%–90% of anal cancers are caused by HPV, and premalignant changes, termed anal intraepithelial neoplasias (AINs), occur.5,6 Although screening for anal cancer and its precursors is recommended,7,8 there are no consensus guidelines for doing so in Canada.
The incidence of anal cancer in Canada is 1.7 per 100 000 person-years.1,9 However, among people with HIV infection, the rates range from 49 to 144 per 100 000 person-years.3,4,10 This figure is comparable to the rate of colorectal cancer in men (60 per 100 000 person-years).1 Among 13 cohorts, unadjusted rates of anal cancer were 131, 46 and 2 per 100 000 for men with HIV infection who have sex with men (rate ratio [RR] 80.3, 95% confidence interval [CI] 42.7–151.1), men with HIV infection (RR 26.7, 95% CI 11.5–61.7) and men without HIV infection, respectively.11
Here, we review the epidemiology, screening and prevention of anal cancer. We conducted a broad literature search to inform this review, and the level of evidence ranged from expert opinion to randomized controlled trials (Box 1). The term anal cancer as used in this review refers to the predominant histologic form of anal cancer, which is squamous cell carcinoma.
Evidence used for this review
We performed a PubMed and Embase literature search from the earliest possible date to June 2014. We used the search terms “anal cancer” and “anal intraepithelial neoplasia” with the following medical subject headings to identify the most relevant research: “HIV,” “incidence,” “HPV,” “HPV vaccine,” “treatment,” “HAART,” “cART,” “trends,” “cigarette smoking,” and “solid organ transplantation.” We manually reviewed the abstracts and bibliographies of relevant studies to identify additional articles. We included all types of reports (randomized controlled trials, meta-analyses, reviews and prospective cohort, retrospective cohort, case–control and cross-sectional studies). We included only articles in English. Where possible, we selected the most recent articles (published within the last 5 yr) with the highest level of evidence (e.g., randomized controlled trials; meta-analysis of randomized controlled trials) for inclusion. We reviewed 232 articles for relevance, 49 of which are included in this review.
How are anal cancers classified, and what are the risk factors?
The anal canal extends 4 cm from the anorectal junction caudally to the anal verge, beyond which is the anal margin or perianal skin. The canal is lined by squamous tissue that transitions to the columnar or glandular tissue of the rectum at the squamo–columnar junction. Squamous cell carcinoma and precancerous lesions (i.e., AINs) are most likely to arise at this junction. Some squamous cancers also occur in the perianal area, but they behave more like skin cancers.12 In this review, we will focus on squamous cell carcinoma of the anal canal, for which risk factors (Box 2)12 overlap with those for HPV infection.
Risk factors for anal cancer12
Human papillomavirus infection
HIV infection*
Multiple sexual partners*
Anal-receptive intercourse*
Anal warts
History of cervical, vulvar or vaginal carcinoma
Chronic immune suppression not caused by HIV infection*
Cigarette smoking
↵* Risk factors that overlap with those for HPV infection.
Oncogenic strains of HPV, particularly types 16 and 18, cause 80%–90% of anal cancers. HPV encodes tumour suppressor proteins E6 and E7, and their interactions with intracellular proteins p53 and retinoblastoma lead to changes in cell growth and regulation, apoptosis and immortalization leading to malignant disease.13,14
What is the connection between immune suppression and anal cancer?
With the advent of combined antiretroviral treatment, people with HIV infection are living longer, and AIDS-defining cancers such as Kaposi sarcoma, non-Hodgkin lymphoma and cervical cancer have declined. However, non–AIDS defining cancers are increasing in this population.10,15–17 Despite very effective antiretroviral treatments, there has been a progressive increase in the incidence of anal cancer, which appears to have plateaued recently.3,4,11,16,18–21
In one large study, there was no relationship between CD4+ T helper cell counts at HIV registration and risk of anal cancer.22 Conversely, other studies have reported associations between lower CD4+ T helper cell counts at baseline or nadir CD4+ T helper cell counts and anal cancer.21,23 Mortality from non–AIDS defining cancers decreased from 6.0 (95% CI 3.3–10.1) to 0.6 (95% CI 0.4–0.8) per 1000 person-years when the CD4+ T helper cell count rose to more than 500 cells/μL.24 Prolonged undetectable HIV viral loads were also associated with a decreased risk of anal cancer.25
Recipients of solid organ transplantation also have an increased incidence of HPV-related cancers, including anal cancer (11.6 per 100 000 person-years, increased standardized incidence ratio 2.77–10.26).26,27
Should we screen for premalignant lesions, and if so, how?
Lack of evidence that screening reduces rates of anal cancer and mortality has led to a lack of guidelines for screening. Uncertainty exists as to which at-risk populations should undergo screening, and there are differing views on the cost-effectiveness of screening and variable screening methods.
The idea behind screening for anal cancer is to detect precancerous histopathologic changes (dysplasia) and ablate these lesions before invasive cancer occurs, as with screening for cervical cancer.
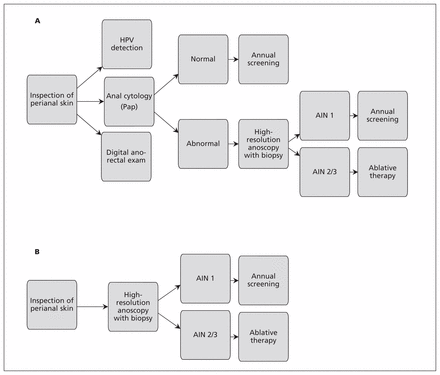
Screening can involve an initial anal Papanicolaou (Pap) test or cytology using a swab inserted into the anal canal past the squamo–columnar junction. The swab is vigorously rotated to acquire cells, then placed in liquid-based medium for cytologic examination. The same sample can be used to test for HPV. A digital anorectal exam is then performed to detect warts, induration or masses. If the results of the Pap test are abnormal, high-resolution anoscopy is typically done to examine the anal canal, with a bright light and magnification being provided by a colposcope. Staining with vinegar and Lugol’s iodine aids identification of possible dysplastic lesions for biopsy. The pathology grading system mimics that of cervical pathology. For cytology, this includes atypical squamous cells of undetermined significance (ASCUS), low-grade squamous intraepithelial lesions (LSILs) and high-grade squamous intraepithelial lesions (HSILs). For histology, the lesions can be designated as either low-grade AIN (AIN 1; LSIL) or high-grade AIN (AIN 2/3; HSIL).
Although some clinicians do anal cytology first and proceed to high-resolution anoscopy only if the cytology is abnormal, others go directly to high-resolution anoscopy because cytology has low specificity, and high-resolution anoscopy with directed biopsy is the gold standard for detecting AIN lesions (Figure 1).28,29 The preferred approach depends on the population undergoing screening and the resources and expertise available.
Two approaches to screening for anal cancer. (A) High-resolution anoscopy with abnormal results on anal cytology. (B) High-resolution anoscopy done directly. Adapted from Lam et al.29 and Palefsky and Rubin.30 Note: AIN = anal intraepithelial neoplasia, HPV = human papillomavirus, Pap = Papanicolaou test.
The New York State Department of Health AIDS Institute and European guidelines suggest annual anal Pap screening of men with HIV infection who have sex with men, with referral for high-resolution anoscopy for any abnormal cytology.7,8,29 The reason for this approach is the high sensitivity of Pap screening at this cytologic cut-off (84%). However, the specificity is only 39%,31 with a positive likelihood ratio of 1.38 and negative likelihood ratio of 0.41.
If a small number of people at high risk are undergoing screening and the resources are available, some experts prefer to go directly to high-resolution anoscopy without cytology.29 However, the lack of experienced providers to perform the procedures and the associated costs do not allow for the screening of large at-risk populations using this approach.
If many patients are being offered screening, triaging by cytology is likely the best approach. Anoscopy should then be reserved for those with HSIL on cytology because the specificity approaches 91%.31
Whether cytology or high-resolution anoscopy is used initially, a threshold of any abnormal cytology leads to unnecessary high-resolution anoscopies, whereas using an HSIL threshold misses patients with internal high-grade disease. The addition of biomarkers such as HPV typing or E6 RNA transcripts may help to better determine who should undergo high-resolution anoscopy.32
Digital anal examination should be emphasized as a method for detecting early cancers, because these smaller cancers have a better outcome. However, a digital examination does not detect precancerous lesions.
There have been no clinical trials showing the utility of screening for anal cancer precursors. Although much of the evidence is drawn through parallels to screening for cervical cancer, progression from anal dysplasia to cancer may not occur at the same rate as the progression to cervical cancer.33 About 30% of inadequately treated cervical intraepithelial neoplasia progresses to cancer over 30 years.34 In comparison, studies have shown rates of progression of high-grade dysplasia to anal cancer in men with HIV who have sex with men to be around 15% over two to five years.35,36 Others, however, have calculated the current progression rate to be slower (0.3% per year).30
An Ontario study looking at the costeffectiveness of detecting AIN 2/3 in men with HIV who have sex with men concluded that direct use of high-resolution anoscopy without preceding cytology was the best strategy. Although this approach was initially more costly, the expense was offset by a higher sensitivity and the low specificity of both HPV and cytology testing.29 A recent study confirmed these findings, with AIN 2/3 diagnosed in 28 patients (52.7%) who did not have HSIL on anal cytology.37 The approvpriate screening interval is unclear: an American systematic review showed that screening for anal cancer among men with HIV who have sex with men every one to two years was cost-effective; this result was not confirmed in the United Kingdom.38
Screening for anal cancer in populations at high risk is acceptable and feasible.39 In addition to men with HIV who have sex with men, we suggest that other groups at risk should also undergo screening, including men and women with HIV, women with genital dysplasia or cancers, people with perianal high-grade lesions, and recipients of solid organ transplants.
Is anal cancer preventable?
Prevention can be categorized according to behavioural and nonbehavioural interventions (Box 3). Safer sex practices, such as a reduced number of partners and regular condom use, can reduce the risk of HPV-related diseases40 and facilitates clearance of HPV-associated penile lesions in men whose partners have cervical dysplasia.41 Cigarette smoking is linked to anal cancer and cytologic abnormalities.42,43 There is some protection against AIN in patients with HIV who have high CD4+ T helper cell counts and controlled viral loads.25 Male circumcision reduces penile oncogenic HPV infection, markedly reduces the risk of penile cancer and reduces the risk of cervical cancer in female partners.44,45 High consumption of fruits, vegetables, vitamins and minerals with antioxidant properties helps to reduce progression of HPV infections to high-grade lesions.46
Preventing anal cancer
Behavioural interventions
Nonbehavioural interventions
The quadrivalent HPV vaccine (types 6, 11, 16 and 18) protects against high-grade cervical lesions47,48 and against high-grade anal dysplasia among men who have sex with men.49 Rates of high-grade dysplasia related to the types of HPV against which the vaccine protects were reduced by 54.2% (95% CI 18.0–75.3) in the intention-to-treat population and by 74.9% (95% CI 8.8–95.4) in the per-protocol population.49 The vaccine also reduced persistent anal infection. In addition to its role in preventing cervical cancer among women, the quadrivalent HPV vaccine is recommended in Canada for boys and men between the ages of 9 and 26 years for the prevention of AIN grades 1–3, anal cancer and anogenital warts.9 The bivalent vaccine (types 16 and 18) has been proven to help protect against high-grade cervical lesions; however, there have been no randomized controlled trials looking at the bivalent vaccine in preventing high-grade anal dysplasia.
It seems intuitive that, in populations with high rates of anal cancer, high-grade lesions are preinvasive and should be removed. In clinics where screening and ablation are done, anal cancers are rarely seen.50,51
Gaps in knowledge
Although screening and early treatment should help prevent anal cancer, similar to what has been accomplished with screening for cervical cancer, the efficacy of this approach has not yet been proven in clinical trials.
Conclusion
Anal cancer is a preventable sexually transmitted disease. It is important to recognize populations at risk and counsel patients on early detection, safer sex practices, smoking cessation and HPV vaccination. The current quadrivalent HPV vaccine prevents AIN, anal cancer and anogenital warts.
Key pointsAnal cancer and anal intraepithelial neoplasia are sexually transmitted diseases associated with certain oncogenic strains of human papillomavirus (HPV), particularly strains 16 and 18.
Anal cancer is prevalent among people with HIV infection, particularly men who have sex with men.
Populations at high risk of acquiring anal cancer should undergo regular screening, although the efficacy of this approach has not yet been proven in clinical trials.
The HPV vaccine is effective in reducing rates of anal intraepithelial neoplasia and anal cancer.
Footnotes
See also CMAJ Open research article at www.cmajopen.ca/content/2/3/E127.full
Competing interests: Irving Salit has received fees for lectures from Merck. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Richard Medford reviewed all of the relevant publications in the English literature, extracted that information and wrote the initial draft of the article. Irving Salit concieved the idea for the article and revised the article for important intellectual content. Both of the authors approved the final version submitted for publication.