Abstract
Background: Statins were initially used to improve cardiovascular outcomes in people with established coronary artery disease, but recently their use has become more common in people at low cardiovascular risk. We did a systematic review of randomized trials to assess the efficacy and harms of statins in these individuals.
Methods: We searched MEDLINE and EMBASE (to Jan. 28, 2011), registries of health technology assessments and clinical trials, and reference lists of relevant reviews. We included trials that randomly assigned participants at low cardiovascular risk to receive a statin versus a placebo or no statin. We defined low risk as an observed 10-year risk of less than 20% for cardiovascular-related death or nonfatal myocardial infarction, but we explored other definitions in sensitivity analyses.
Results: We identified 29 eligible trials involving a total of 80 711 participants. All-cause mortality was significantly lower among patients receiving a statin than among controls (relative risk [RR] 0.90, 95% confidence interval [CI] 0.84–0.97) for trials with a 10-year risk of cardiovascular disease < 20% [primary analysis] and 0.83, 95% CI 0.73–0.94, for trials with 10-year risk < 10% [sensitivity analysis]). Patients in the statin group were also significantly less likely than controls to have nonfatal myocardial infarction (RR 0.64, 95% CI 0.49–0.84) and nonfatal stroke (RR 0.81, 95% CI 0.68–0.96). Neither metaregression nor stratified analyses suggested statistically significant differences in efficacy between high-and low-potency statins, or larger reductions in cholesterol.
Interpretation: Statins were found to be efficacious in preventing death and cardiovascular morbidity in people at low cardiovascular risk. Reductions in relative risk were similar to those seen in patients with a history of coronary artery disease.
Although statins are known to improve survival and relevant clinical outcomes in high-risk populations,1 evidence of their clinical benefit in lower risk populations is more equivocal. Initially, low-risk populations were defined by the absence of known coronary artery disease (and their treatment was termed “primary prevention”). However, it was subsequently recognized that these populations included both patients at very high risk of coronary artery disease (e.g., those with severe peripheral vascular disease) and those at very low risk (e.g., those aged < 40 years who have no diabetes or hypertension and have low-density lipoprotein cholesterol level of less than 1.8 mmol/L). Accordingly, current guidelines for the use of statins are based on the projected risk of an atherosclerotic event rather than solely on the presence or absence of known coronary artery disease.2,3
Results of the recent JUPITER study (Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin)4 have renewed enthusiasm for the use of statins in people without a history of coronary artery disease and have generated further controversy as to whether high-potency statins such as rosuvastatin and atorvastatin lead to better clinical outcomes than low-potency statins such as pravastatin, simvastatin, fluvastatin and lovastatin. We did a systematic review of randomized trials to assess the efficacy and harms of statins in people at low cardiovascular risk, including indirect comparisons of high-potency and low-potency statins.
Methods
We performed a systematic review of published and unpublished randomized controlled trials that compared statins with no statin or placebo. We used accepted methods for literature searches, article selection, data extraction and risk-of-bias assessment. The review was reported according to accepted guidelines.5,6
Literature search
We searched MEDLINE (1950 to Jan. 28, 2011) and EMBASE (1950 to Jan. 28, 2011). Details of the search strategy appear in Appendix 1 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.101280/-/DC1). Because of high search yields, an update of a modified version of the National Institute for Health and Clinical Excellence search7 was performed, with elimination of studies published before 2003. Searches were not restricted by language (when a translator could be found).
We also searched registries of health technology assessments and clinical trials, conducted manual searches of reference lists of relevant reviews, and contacted Canadian manufacturers of statins (Astra Zeneca, Merck Frosst, Pfizer, Bristol Myers Squibb, Novartis) for additional studies and unpublished reports of trials.
Study selection and validity assessment
We included parallel-group randomized controlled trials if they included people 16 years or older who were at low cardiovascular risk (as defined in the next paragraph), the follow-up period was at least six months, and an eligible statin (atorvastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin or simvastatin) was compared with no statin (placebo or standard care). We categorized statins according to their pharmacologic effect on lowering cholesterol as either low potency (fluvastatin, lovastatin, pravastatin and simvastatin) or high potency (atorvastatin and rosuvastatin).8 To be eligible, studies also had to report one or more of the following outcomes: all-cause mortality, unstable angina, acute myocardial infarction (fatal or nonfatal), stroke or transient ischemic attack (fatal or nonfatal), surgical or percutaneous revascularization, length of stay, quality of life, persistence on statin therapy, and adverse events. Trials with fewer than 30 participants per study arm were excluded to improve the efficiency of the work without an appreciable loss of power.9
Trials were considered to have enrolled participants at low cardiovascular risk if the 10-year risk of cardiovascular-related death or nonfatal myocardial infarction among participants was less than 20%,10 as assessed by extrapolation of observed risk in the control group of each trial. In general, this corresponded to participants who were free from cardiovascular disease (i.e., no prior acute coronary syndrome or coronary revascularization, no prior ischemic stroke and no prior revascularization or loss of limb owing to peripheral arterial disease) and diabetes. Data from trials in which some, but not all, participants had known cardiovascular disease were included if the control group had a low cardiovascular risk (as defined above).
In sensitivity analyses, we calculated the estimated 10-year risk of cardiovascular-related death or nonfatal myocardial infarction for the average participant in each trial, using mean baseline characteristics from the control group and two commonly used formulas from D’Agostino and colleagues11 and the Third Adult Treatment Panel.12 We also used a number of other definitions for low risk in sensitivity analyses.
Two reviewers screened each citation. Trials considered to be relevant by one or both reviewers were retrieved, and the full text was independently assessed by two reviewers for inclusion. Disagreements were resolved by a third party through consensus.
Two reviewers independently assessed each study’s risk of bias using a condensed version of the Chalmers Index13 as well as other characteristics that influence the risk of biased estimates of effectiveness in meta-analyses.14–16 Disagreements were resolved by a third party through consensus.
Data extraction
One reviewer extracted data from selected trials, and a second reviewer checked the data for accuracy. Results of intention-to-treat analyses were collected if reported.
We classified adverse events as serious if they were defined as such by the primary authors or if their severity was unspecified but they led to withdrawal from therapy or study. We also collected data on the incidence of new diabetes, cancer and rhabdomyolysis. Myocardial infarctions were classified as fatal, nonfatal or fatal/nonfatal; those in the fatal/nonfatal category were from studies that did not specify the type of myocardial infarction or separate totals for fatal and nonfatal myocardial infarctions. Strokes were classified in a similar manner. Angina was classified as unstable if defined as such by the primary authors, if it required admission to hospital or if it necessitated revascularization. Angina that did not meet these criteria was classified as “unspecified” angina. Types of revascularization included coronary artery bypass graft surgery, percutaneous coronary intervention, percutaneous transluminal coronary angioplasty, interventional vascular procedure, coronary revascularization, arterial revascularization and coronary angioplasty. For studies in which the number of cardiovascular-related deaths or nonfatal myocardial infarctions was unclear, we contacted the original authors for information so that we could calculate the 10-year risk.
Data synthesis and analysis
We pooled the results using the random-effects model described by DerSimonian and Laird.17 We used relative risks (RRs) to summarize dichotomous results. We quantified statistical heterogeneity using the I2 statistic and used “small,” “moderate” and “large” to describe values of 25%, 50% and 75%.18,19 A weighted regression test20 was used to test for publication bias.
We used univariable and bivariable meta-regression analyses18 to examine whether the following variables influenced the association between statin use and all-cause mortality: duration of study, year of study, baseline cholesterol levels, observed annual cardiovascular risk in the control arm, the absolute and percent change in low-density lipoprotein cholesterol from baseline in the statin arm, the point at which low-density lipoprotein cholesterol was measured, daily initial dose of statin, potency of statin (high v. low), mean age of participants, proportion of participants who were male, proportion who had diabetes, proportion who had hypertension, and risk-of-bias items.
The number needed to treat and the absolute risk reduction were calculated for outcomes with statistically significant relative risks. The number needed to treat indicates the number of patients who need to be given statin treatment to prevent one event and is the reciprocal of the absolute risk reduction (difference in probabilities of an event between treatment and control groups).21
In subgroup analyses, we examined all-cause mortality and other clinical outcomes for trials stratified by statin potency (high v. low). We also explored the relative effects of statins using a method of indirect comparison of treatments described by Bucher and colleagues,22 a technique that facilitates the comparison of any two statins not directly compared in any one study. Here, the magnitude of treatment effects (e.g., relative risk) for the direct evidence of treatment A versus B and treatment B versus C were compared with acquired indirect evidence of treatment A versus C.
Analyses were conducted using RevMan Version 4.2 (The Cochrane Collaboration, 2002) and Stata/MP 11.2 software (Stata Corp, 2011).
Results
Literature search
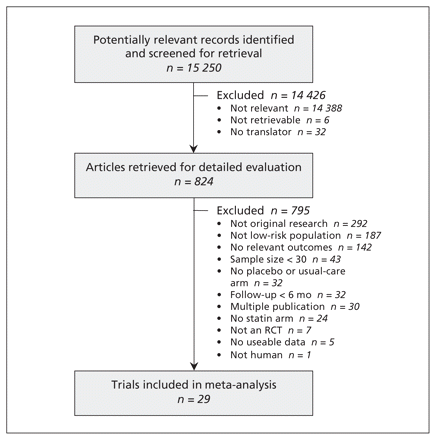
Of the 15 250 articles identified, 29 (n = 80 711) met our inclusion criteria (Figure 1).4,23–50 Atorvastatin was the study drug in six trials (n = 11 894), fluvastatin in four trials (n = 2107), lovastatin in three (n = 15 769), pravastatin in nine (n = 30 974), rosuvastatin in four (n = 19 129) and simvastatin in three (n = 838). Twenty-two of the trials were placebo-controlled, and the remaining seven reported usual care, conventional treatment, standard treatment, no treatment or diet as the comparators. The median year of publication was 2004 (range 1991–2010). The median duration of follow-up was two years (range 0.5–5.3 years). Details of the studies are summarized in Table 1.
Selection of studies for inclusion in the meta-analysis. RCT = randomized controlled trial.
Description of studies included in the systematic review
Risk-of-bias assessment
The 29 trials generally exhibited moderate risk of bias (Table 2). A weighted regression test20 using all-cause mortality results detected no statistical evidence of publication bias (bias = 0.03, p = 0.92).
Risk-of-bias assessment of studies included in the systematic review
Characteristics of participants
The median age of the participants was 58 years (range 51–76), and the proportion who were male ranged from 0% to 100% (median 62%). The median proportion of participants who had diabetes was 7% (range 0%–35%), and hypertension 47% (range 16%–100%) (Table 1).
The baseline lipid levels in the included trials were: total cholesterol, median 6.0 (range 4.8–7.6) mmol/L; low-density lipoprotein cholesterol, median 4.0 (range 2.8–5.2) mmol/L; high-density lipoprotein cholesterol, median 1.3 (range 1.0–1.9) mmol/L; and triglycerides, median 1.7 (range 1.2–3.6) mmol/L. Seven studies reported baseline C-reactive protein levels: median 0.17 (range 0.15–7.7) mg/dL (16.6 [range 13.8–732.4] nmol/L). The mean 10-year risk of cardiovascular-related death or nonfatal myocardial infarction was 6% (range 0%–18%).
Outcomes
Table 3 presents a summary of the relative risks (overall and stratified by high- and low-potency statins), number needed to treat and absolute risk reduction for all outcomes.
Relative risk of serious outcomes associated with the use of statins in patients at low cardiovascular risk*
All-cause mortality
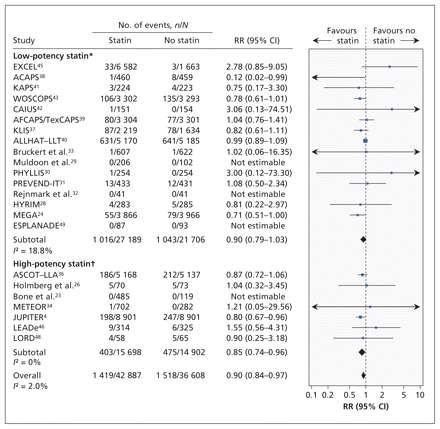
Twenty-three trials (n = 79 495) reported all-cause mortality (Figure 2). The trial-level relative risk could not be estimated for four trials that reported no events in either group.51 From the remaining 19 trials (n = 78 321), the pooled relative risk of death was significantly lower among statin recipients than among controls (RR 0.90, 95% confidence interval [CI] 0.84–0.97; I2 = 2.0%).
Risk of death from any cause associated with the use of statins (versus no statins) in patients at low cardiovascular risk (observed 10-year risk of cardiovascular-related death or nonfatal myocardial infarction < 20%). A relative risk (RR) of less than 1.0 indicates fewer deaths with the use of statins. CI = confidence interval. *Lovastatin, fluvastatin, pravastatin and simvastatin. †Rosuvastatin and atorvastatin. For complete study names, see Box 1.
In the metaregression analyses, none of the characteristics listed in the methods section (including baseline cholesterol levels or change in lipid levels during the trials) significantly modified the association between statin use and all-cause mortality at the level of p < 0.05 (Appendix 2, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.101280/-/DC1).
Myocardial infarction
Thirteen trials (n = 48 023) reported the number of participants with fatal, nonfatal or unspecified myocardial infarctions. The pooled relative risk was significantly lower among statin recipients than among controls (RR 0.63, 95% CI 0.50–0.79; I2 = 13%].
From eight trials (n = 31 424) that provided data on fatal myocardial infarctions, we found that the pooled relative risk did not differ significantly between treatment groups (RR 0.96, 95% CI 0.5–1.85; I2 = 0%).
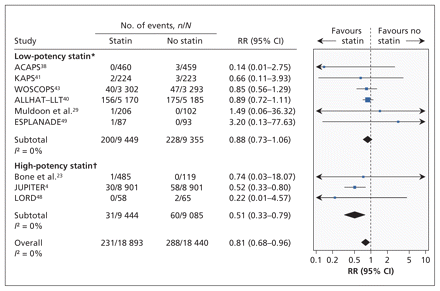
Ten trials (n = 49 222) reported nonfatal myocardial infarctions. The pooled relative risk was significantly lower among statin recipients than among controls (RR 0.64, 95% CI 0.49–0.84; I2 = 44%) (Figure 3). After removing studies that enrolled people with prior myocardial infarction, we found that the point estimate was similar to the estimate from the primary analysis (eight trials, n = 48 595; RR 0.64, 95% CI 0.48–0.86; moderate heterogeneity [I2 = 54%]).
Risk of nonfatal myocardial infarction associated with the use of statins (versus no statins) in patients at low cardiovascular risk (observed 10-year risk of cardiovascular-related death or nonfatal myocardial infarction < 20%). A relative risk (RR) of less than 1.0 indicates fewer events with the use of statins. CI = confidence interval. *Lovastatin, fluvastatin, pravastatin and simvastatin. †Rosuvastatin and atorvastatin. For complete study names, see Box 1.
Stroke
Fourteen trials (n = 60 841) reported the number of participants with fatal, nonfatal or undefined strokes. The pooled relative risk of stroke was significantly lower among statin recipients than among controls (RR 0.83, 95% CI 0.74–0.93; I2 = 0%).
Five trials (n = 36 118) reported on the risk of fatal stroke; the pooled relative risk did not differ significantly between treatment groups (RR 0.91, 95% CI 0.65–1.29; I2 = 0%). Nine trials (n = 37 333) reported the number of participants with nonfatal stroke; the pooled relative risk was significantly lower among statin recipients (relative risk 0.81 [0.68–0.96]; I2 = 0%) (Figure 4).
Risk of nonfatal stroke associated with the use of statins (versus no statins) in patients at low cardiovascular risk (observed 10-year risk of cardiovascular-related death or nonfatal myocardial infarction < 20%). A relative risk (RR) of less than 1.0 indicates fewer events with the use of statins. CI = confidence interval. *Lovastatin, fluvastatin, pravastatin and simvastatin. †Rosuvastatin and atorvastatin. For complete study names, see Box 1.
Coronary revascularization
Eight trials (n = 43 708) reported the number of participants who underwent percutaneous or surgical coronary revascularization. The incidence of revascularization was significantly lower among statin recipients than among controls (RR 0.66, 95% CI 0.57–0.77; I2 = 7%).
Other outcomes
Four trials (n = 35 017) reported on the risk of unstable angina: the pooled relative risk was significantly lower among statin recipients than among controls (RR 0.71, 95% CI 0.55–0.92; I2 = 0%). Three trials (n = 9082) reported on the risk of unspecified angina: the pooled relative risk did not differ significantly between treatment groups (RR 0.83, 95% CI 0.57–1.22; I2 = 0%). Thirteen trials reported on the proportion of participants who adhered to statin therapy (range 63%–98%). Length of hospital stay, persistence on statin therapy and quality-of-life measures were not reported in any of the studies.
Adverse events
Twenty-one trials (n = 47 589) reported the number of participants who had serious adverse events. The trial-level relative risk could not be estimated for four trials that reported no events in either group. From the remaining 17 trials (n = 47 021), the pooled risk of serious adverse events did not differ significantly between treatment groups (RR 1.01, 95% CI 0.96–1.07; I2 = 8%).
Ten trials (n = 45 557) reported the number of participants who experienced rhabdomyolysis. From the three trials in which this outcome occurred (n = 34 712), the pooled risk of rhabdomyolysis did not differ significantly between groups (RR 1.29, 95% CI 0.25–6.68; I2 = 0%).
Ten trials (n = 62 547) reported the number of participants in whom cancer was diagnosed, and four trials (n = 31 818) reported the proportion with new diabetes. The pooled risk of cancer did not differ significantly between groups (RR 1.00, 95% CI 0.93–1.08; I2 = 0%), nor did the pooled risk of diabetes (RR 1.05, 95% CI 0.84–1.32), although heterogeneity between studies in the latter analysis was large (I2 = 64%).
Subgroup and sensitivity analyses
Indirect comparisons between high- and low-potency statins did not show a significant difference in the risk of all-cause mortality (RR for high- v. low-potency statins: 0.94, 95% CI 0.79–1.13), fatal or nonfatal myocardial infarction (RR 0.69, 95% CI 0.43–1.12) or stroke (RR 0.79, 95% CI 0.61–1.02).
The sensitivity analyses, many of which used alternative definitions of low cardiovascular risk, showed findings that were consistent with the results of the primary analysis across a wide variety of assumptions and conditions (Appendices 3 and 4, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.101280/-/DC1). In particular, use of estimates rather than observed data to classify trials with respect to 10-year cardiovascular risk led to similar conclusions regarding the efficacy of statins in reducing all-cause mortality. In addition, when low cardiovascular risk was defined as a 10-year risk of cardiovascular-related death or nonfatal myocardial infarction of less than 10%, the pooled relative risk was similar to that of the primary analysis (RR 0.83, 95% CI 0.73–0.94). The pooled relative risk of all-cause mortality among statin recipients was statistically significant in virtually all of the sensitivity analyses (28 of 33). In addition, the point estimate for the pooled relative risk was relatively stable in these analyses (range 0.78–0.93, as compared with the point estimate of 0.90 in the primary analysis), except for one analysis that included trials with a follow-up period shorter than the median of two years (pooled RR 0.99).
Interpretation
We found that the use of statins by people at low cardiovascular risk reduced the relative risk of death from any cause by 10%. Treatment with statins also reduced the risk of stroke, myocardial infarction, unstable angina and coronary revascularization to an extent similar to that seen in trials involving patients with coronary artery disease.52
In a meta-analysis of the efficacy and safety of statins among patients with coronary artery disease, the number needed to treat was 86 to prevent a single death from any cause and 62 to prevent a single nonfatal myocardial infarction.52 The corresponding numbers needed to treat among people at low cardiovascular risk by our primary definition were 239 and 153, which reflect the generally low rates of vascular events in this population. Because statins might be used indefinitely to reduce cardiovascular risk, the absolute benefit may increase (accompanied by corresponding decreases in the number needed to treat) with longer durations of treatment, although this remains speculative.
Although we sought information from all trials on outcomes associated with serious harm from statins, information on serious adverse events was available for only 59% (47 589/ 80 711) of the trial participants. This low proportion is consistent with previously documented deficiencies in reporting harm among participants in randomized controlled trials.53,54 Our experience with soliciting this information from investigators revealed a lack of clarity among trial authors regarding how serious adverse events were reported in published reports. Because the risk of serious morbidity would be expected to decline among statin users, our finding of a lack of significant difference in the rate of serious adverse events between treatment groups and our nonsignificant estimate of elevated risk among statin users should be interpreted with caution. A complete analysis of published and unpublished data from individual patients on the effect of statins among low-risk patients would be a useful addition to the literature, but it would require cooperation among the various stakeholders.
Our metaregression and indirect comparisons did not show statistically significant differences in the efficacy of high- and low-potency statins. However, metaregression has relatively low statistical power. The as-yet unproven potential for high-potency statins to prevent cardiovascular outcomes more effectively must be balanced against their higher costs compared with low-potency statins. The recent availability of generic (lower cost) atorvastatin makes this issue of particular importance for decision-makers and third-party payers.
Unlike a recent systematic review that included statin trials involving both low- and high-risk people,55 we did not find a significantly increased risk of new diabetes among low-risk statin users, perhaps because of differences in study populations or statistical power. If such an effect exists, this would be expected to further reduce the absolute benefit of statin treatment in low-risk populations over the long term.
Our findings also differ from those of another recently published systematic review of 11 trials involving 65 229 people without cardiovascular disease that found no evidence of a benefit of statin use on all-cause mortality.56 The authors reported a relative risk reduction for all-cause mortality that was similar to ours (9% v. 10%), but their estimate was less precise, leading to a 95% CI that crossed unity (0.83–1.01). Eight trials in that review were included in our study.4,24,28,31,36,39,40,43 However, we included an additional 11 trials that reported on all-cause mortality, and we excluded 2 trials57,58 that exclusively enrolled participants with a diagnosis of diabetes at baseline. A third study included in the prior reviews was excluded from our study because the 10-year risk for cardiovascular-related death or nonfatal myocardial infarction was greater than 20%.59
Strengths and limitations
This is an up-to-date, comprehensive systematic review of the clinical implications of statin use among people at low cardiovascular risk. We included studies in which participants were considered to be at low risk according to an accepted definition — a 10-year incidence of less than 20% for cardiovascular-related death or nonfatal myocardial infarction.10 We excluded trials that predominantly enrolled people at higher cardiovascular risk, such as those with diabetes or prior cardiovascular events. Therefore, our relative risk estimates were unlikely to be influenced by higher-risk subgroups within the trial populations.
Our analysis has limitations. First, as with all meta-analyses, our conclusions are limited by the availability of individual trials. We cannot exclude the possibility of publication bias, although it seems unlikely that there are unidentified trials large enough to influence the direction or significance of our findings.
Second, although we used accepted techniques for metaregression in an attempt to identify factors associated with greater or lesser benefit from statin use among low-risk participants, statistical power for these analyses was relatively low given the number of available trials.
Third, the duration of all trials was relatively short given that people at low cardiovascular risk might require treatment with statins for decades.
Fourth, we used the number needed to treat to summarize the benefits of statins across different analyses. Whether the number needed to treat is appropriate for use in meta-analyses is controversial; however, we believe that the simplicity and transparency of this method outweigh its potential disadvantages.21,60
Fifth, although we reported a wide range of clinically relevant outcomes as specified in our protocol, the definition for some (e.g., unstable angina) will have varied across trials.
Sixth, in an attempt at unbiased identification of people at low cardiovascular risk, we based our inclusion criterion on the observed incidence of events among placebo recipients, anticipating that clinicians could apply our findings in practice by using their risk prediction instrument of choice to identify patients with an estimated 10-year risk of less than 20% for cardiovascular-related death or nonfatal myocardial infarction. Because risk prediction tools may overestimate true rates of events in contemporary practice, we repeated analyses after including only trials with an estimated 10-year risk of less than 20% based on mean participant characteristics and two widely used risk equations;11,12 we reached similar conclusions.
Finally, given that most of the trials included in our review were at moderate risk of bias, we cannot exclude the possibility that other factors besides statin use may have influenced the observed differences in health outcomes between treatment groups.
Conclusion
Both low- and high-potency statins were efficacious in preventing death and cardiovascular-related morbidity in people at low risk of cardiovascular events (whose 10-year risk of cardiovascular-related death or nonfatal myocardial infarction is less than 20%), most of whom did not have a history of coronary artery disease or diabetes. However, the number needed to treat to prevent one adverse outcome was relatively high for any statin. Whether high-potency statins improve outcomes to a greater extent than low-potency statins is uncertain based on current data.
Full names of trials included in the meta-analysis
-
ACAPS: Asymptomatic Carotid Artery Progression Study
-
AFCAPS/TexCAPS: Air Force/Texas Coronary Atherosclerosis Prevention Study
-
ALLHAT–LLT: Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial
-
ASCOT–LLA: Anglo-Scandinavian Cardiac Outcomes Trial – Lipid Lowering Arm
-
ASTRONOMER: Aortic Stenosis Progression Observation: Measuring the Effects of Rosuvastatin
-
CAIUS: Carotid Atherosclerosis Italian Ultrasound Study
-
ESPLANADE: European Study for Preventing by Lipid-lowering Agents aNd ACE-inhibition Dialysis Endpoints
-
EXCEL: Expanded Clinical Evaluation of Lovastatin Study
-
HYRIM: Hypertension High-Risk Management Trial
-
JUPITER: Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin
-
KAPS: Kuopio Atherosclerosis Prevention Study
-
KLIS: Kyushu Lipid Intervention Study
-
LEADe: Lipitor’s Effect in Alzheimer’s Dementia
-
LORD: Lipid Lowering and Onset of Renal Disease
-
MEGA: Management of Elevated Cholesterol in the Primary Prevention Group of Adult Japanese
-
METEOR: Measuring Effects on Intima-Media Thickness: an Evaluation of Rosuvastatin
-
PHYLLIS: Plaque Hypertension Lipid-Lowering Italian Study
-
PREVEND-IT: Prevention of Renal and Vascular Endstage Disease Intervention Trial
-
UK-HARP-I: First United Kingdom Heart and Renal Protection
-
WOSCOPS: West of Scotland Coronary Prevention Study
Acknowledgements
The authors thank Dr. Steven Grover for his helpful comments on an earlier draft of this manuscript.
Marcello Tonelli, Finlay McAlister and Braden Manns are supported by Alberta Innovates — Health Solutions (formerly the Alberta Heritage Foundation for Medical Research (AHFMR) Health Scholar Awards. Brenda Hemmelgarn and Scott Klarenbach were supported by Population Health Investigator Awards from Alberta Innovates — Health Solutions. Marcello Tonelli was supported by a Government of Canada Research Chair in the optimal care of people with chronic kidney disease. Fiona Clement was supported by a postdoctoral fellowship award from the Canadian Health Services Research Foundation and AHFMR. Marcello Tonelli, Brenda Hemmelgarn, Scott Klarenbach, Finlay McAlister and Braden Manns were supported by an alternative funding plan from the Government of Alberta and the Universities of Alberta and Calgary.
Footnotes
-
See related research article by Conly and colleagues at www.cmaj.ca/lookup/doi/10.1503/cmaj.101281 and related commentary by Gupta at www.cmaj.ca/lookup/doi/10.1503/cmaj.111674
-
Competing interests: Marcello Tonelli has received research support from Pfizer; in 2011 he served on an advisory board for Merck, providing expert opinion on global chronic kidney disease and cardiovascular disease. Brenda Hemmelgarn has received research support from Merck and Amgen. Braden Manns has received research support from Merck Canada. No competing interests declared by the other authors.
-
This article has been peer reviewed.
-
Contributors: All the authors contributed to the conception and design. Marcello Tonelli, Fiona Clement, Braden Manns, Anita Lloyd and Jonathan Conly contributed to the acquisition of data and drafted the report. All of the authors contributed to the analysis and interpretation of data, critically revised the report for important intellectual content and approved the final version submitted for publication.
-
Funding: This study was jointly funded by the Canadian Agency for Drugs and Technology in Health and the Alberta Heritage Foundation for Medical Research Interdisciplinary Team Grants Program (which supports the Interdisciplinary Chronic Disease Collaboration). The study sponsors had no role in the design of the study, the collection, analysis or interpretation of data, the writing of the report or the decision to submit the article for publication.