The number of people with diabetes is expected to double within a decade. Most people with diabetes have or will develop hypertension, and observational and randomized trial evidence has proven a causal relation between raised blood pressure and clinically important micro- and macrovascular complications, particularly kidney disease.
The Canadian Hypertension Education Program (CHEP) recommendations specifically target hypertension and diabetes, since control of hypertension can lead to a dramatic reduction in target organ damage.1 The recommendations advise keeping blood pressure to less than 130 mm Hg systolic and 80 mm Hg diastolic for all patients with diabetes, using an angiotension-converting enzyme inhibitor or angiotensin-receptor blocker for patients with nephropathy (either microalbuminuria or overt nephropathy), and expecting to use combination drug therapy with most patients to bring their blood pressure to target levels. In studies in which blood pressure control was achieved in people with diabetes and hypertension, rates of cardiovascular disease were reduced and progression to kidney disease was delayed or prevented.
The Case: A 63-year-old man has returned to your office for review of his diabetes and blood pressure. He has had type 2 diabetes mellitus for 15 years, which was managed first with oral hypoglycemic agents and then, for the last 5 years, with subcutaneous insulin, with reasonable control of his diabetes achieved (hemoglobin A1c concentration 0.68%). He has had high blood pressure for 15 years as well, which, until recently, was controlled with 3 agents: an angiotensin- converting enzyme inhibitor (trandolapril 2 mg daily), a long-acting calcium-channel blocker (amlodipine 5 mg daily), and a diuretic (hydrochlorothiazide 25 mg daily). However, in the last 2 months his home blood pressure readings have crept up from their previous level of 130/80 mm Hg and now consistently average 145/85 mm Hg, his reading in the office today. His weight (90 kg), alcohol intake and diet have remained consistent, and his only other medications are ASA 81 mg daily and atorvastatin 20 mg daily. His heart rate is 72 beats per minute and regular, he has normal fundi and no renal artery bruits, and his cardiac examination results are normal. His creatinine level was 130 mmol/L 2 years ago and 150 mmol/L 2 weeks ago (creatinine clearance rate 55.4 mL/min [0.9 mL/ sec] by Cockroft Gault equation; range for stage 3 chronic kidney disease 30–60 mL/min [0.5–1.0 mL/ sec]). He has microalbuminuria with a random urine albumin/creatinine ratio of 5 mg/mmol. He states that he dislikes taking medications and questions whether lowering his blood pressure any further is really necessary. He asks whether a change in medication might be a better way of managing his blood pressure, rather than increasing any doses.
Question: The threshold for initiating antihypertensive treatment and the target blood pressures are lower in people with diabetes compared with people with uncomplicated hypertension. Why is this?
Comment: Cardiovascular disease is the most common cause of death in people with diabetes, and the association between blood pressure and cardiovascular risk is both continuous and graded in people with diabetes. Even with blood pressures in the high– normal range (130–139/80–89 mm Hg), diabetic patients are at increased risk of cardiovascular disease. Antihypertensive therapy reduces the risk of both cardiovascular disease and renal dysfunction in people with diabetes. The results of the UKPDS study showed that reducing blood pressure from 154/87 mm Hg to 144/82 mm Hg among diabetic patients reduced the risk of myocardial infarction and death by about 3.5%, with a number needed to treat of 28 to prevent either event over 8.4 years (Table 1).2 Furthermore, in another study, diabetic patients who at baseline had a blood pressure of 130/80 mm Hg or higher had odds ratios for new microalbuminuria of 10 and for cardiovascular outcomes of 20 compared with those with blood pressure less than 130/80 mm Hg over 7.8 years.3 Similarly, in the Hypertension Optimal Treatment (HOT) study diabetic subgroup, patients randomly assigned to a diastolic blood pressure target of less than 90 mm Hg (85 mm Hg was achieved) had a risk of myocardial infarction of 2.8% and of death of 6.0% over 3.8 years, whereas those with a diastolic blood pressure target of less than 80 mm Hg (81 mm Hg achieved) had a risk of myocardial infarction of 1.4% and of death of 3.4%. This translates to a number needed to treat of 72 to prevent a myocardial infarction and of 39 to prevent one death (Table 1).4 The patient in the hypothetical case study is currently at risk of target organ damage, and his target blood pressure is less than 130 mm Hg systolic and 80 mm Hg diastolic using office-based measurements. Currently, there is insufficient evidence to recommend a therapeutic blood pressure target using home or ambulatory blood pressure monitoring for people with diabetes.
Table 1.
Question: Which antihypertensive therapies are recommended for people with diabetes?
Comment: The first step with all patients with hypertension should be to recommend lifestyle modifications proven to lower blood pressure (see article 3 in this series, available online at www.cmaj.ca/cgi/content/full/173/7/749). If these lifestyle modifications are ineffective in lowering office blood pressure to less than 130/80 mm Hg in diabetic patients, then medication should be started. The timing of drug therapy initiation depends on the degree of blood pressure elevation and the presence or absence of other cardiovascular risk factors or nephropathy. The CHEP recommendations advise that medications should not be delayed in patients with either nephropathy or other cardiovascular risk factors. In our experience, when medication is adjusted in a patient whose case is similar to that described here, dosages should be increased no sooner than every 4 weeks.
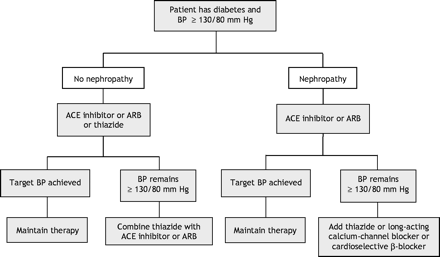
For people with hypertension and diabetes without nephropathy, angiotensin-converting enzyme inhibitors, angiotensin-receptor blockers and thiazide diuretics have all been shown to improve prognoses in large randomized trials and are considered appropriate choices for initial therapy (Fig. 1).1 Combination drug therapy is typically needed to meet the CHEP-recommended blood pressure targets. For example, in the UKPDS 38 trial, one-third of those patients randomly assigned to tight control required at least 3 antihypertensive drugs to achieve target levels.2 If monotherapy fails to achieve the target blood pressure, we recommend that an angiotensin-converting enzyme inhibitor or angiotensin-receptor blocker be combined with a thiazide.1 If blood pressure remains uncontrolled, the 2005 CHEP recommendations endorse adding a cardioselective β-blocker (e.g., atenolol or metoprolol) or long-acting calcium-channel blocker (e.g., amlodipine or the long-acting formulation of nifedipine or diltiazem in a slow-release formulation) to the treatment regimen. The evidence for these recommendations is highlighted in the full guideline publications.1 Caution should be exercised when combining verapamil or diltiazem with a β-blocker in patients with kidney disease because of the risk of heart block or other severe bradyarrhythmia.1
Fig. 1: Hypertension treatment in patients with diabetes mellitus. BP = blood pressure, ACE = angiotensin-converting enzyme, ARB = angiotensin-receptor blocker. Lifestyle interventions (weight loss, sodium restriction, aerobic exercise and alcohol restriction) are recommended for all patients with hypertension in advance of any medications. Nephropathy is the presence of microalbuminuria or overt nephropathy; urine albumin > 30 mg/day or an albumin/creatinine ratio > 2.0 for men or > 2.8 for women.
The patient in the case study is taking appropriate drugs but at low doses. The angiotensin-converting enzyme inhibitor, trandolapril, should be increased to 4 mg daily and possibly to 8 mg daily, and the calcium-channel blocker, amlodipine, should be increased to 10 mg daily. If this still does not bring his blood pressure to target, adding a β-blocker or long-acting α-blocker should be considered. Referral to a hypertension specialist is indicated when blood pressure remains uncontrolled with 3 medications at higher doses after drug adherence has been verified (see article 4 in this series, available online at www.cmaj.ca/cgi/content/full/173/8/865).
Question: Should an angiotensin-converting enzyme inhibitor and angiotensin-receptor blocker be combined because of the nephropathy?
Comment: Although it is possible that combining these agents may provide further benefits for hypertensive patients with diabetic nephropathy, the evidence is preliminary, and this strategy cannot be endorsed until the results of upcoming cardiovascular outcome studies are released. The focus should remain on blood pressure lowering and other proven strategies to reduce renal and cardiovascular risk before attempts are made to lower urinary albumin levels.
Question: What about other therapies?
Comment: Blood pressure control, although important, is only one component of the anti-atherosclerotic strategy when caring for a diabetic patient with hypertension. Cardiovascular risk reduction should include lifestyle changes, cholesterol management and use of low-dose ASA. Aspirin should be considered for all diabetic patients with hypertension, particularly those with other cardiovascular risk factors; however, we recommend that the initiation of aspirin be delayed until adequate blood pressure control is achieved to reduce the risk of hemorrhagic stroke.1 Glycemic and lipid control will help preserve renal health, which in turn maintains both healthy blood pressure and cardiovascular health. Smoking cessation is also important.
Question: When should renal vascular disease be considered, and which screening tests are appropriate?
Comment: Up to 30% of patients with resistant hypertension have renal vascular disease. Our patient has some features that suggest the possibility of atherosclerotic renal artery disease (a prolonged history of diabetes, recently increasing hypertension, and an elevated serum creatinine level). However, he does not demonstrate any of the signs or symptoms that are often associated with renal vascular disease and that would trigger us to order screening tests (Box 1. CHEP currently recommends screening only those patients with at least 2 of the clinical signs found in Box 1. The captopril-enhanced renal scan, doppler sonography, magnetic resonance angiography, or CT angiography (provided patients have a glomerular filtration rate > 60 mL/min and stop taking metformin before the test) are all appropriate diagnostic techniques.
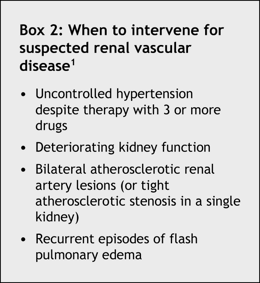
The decision to screen for renal vascular disease is complicated by the fact that interventions to revascularize affected kidneys (angioplasty with stent or surgery) are currently recommended only for patients with the conditions listed in Box 2. Thus, it is unnecessary to pursue investigations for renal vascular disease for patients whose blood pressure and creatinine levels are controlled and stable.1
For patients with renal artery stenosis who do not fulfill the criteria for revascularization, the recommended treatment regimen is similar to that for patients with uncomplicated hypertension (see article 4 in this series), although angiotensin-converting enzyme inhibitors and angiotensin- receptor blockers should be used with caution.1
-
McAlister FA, Wooltorton E, Campbell NRC. The Canadian Hypertension Education Program (CHEP) recommendations: launching a new series. CMAJ 2005;173(5):508-9.
-
Bolli P, Myers M, McKay D. Applying the 2005 Canadian Hypertension Education Program recommendations: 1. Diagnosis of hypertension. CMAJ 2005;173(5):480-3.
-
Hemmelgarn BR, Grover S, Feldman RD. Applying the 2005 Canadian Hypertension Education Program recommendations: 2. Assessing and reducing global atherosclerotic risk among hypertensive patients [published erratum appears in CMAJ 2005;173(7):738]. CMAJ 2005;173(6):593-5.
-
Padwal R, Campbell N, Touyz RM. Applying the 2005 Canadian Hypertension Education Program recommendations: 3. Lifestyle modifications to prevent and treat hypertension. CMAJ 2005;173(7):749-51.
-
Khan NA, Hamet P, Lewanczuk RZ. Applying the 2005 Canadian Hypertension Education Program recommendations: 4. Managing uncomplicated hypertension. CMAJ 2005;173(8):865-7.
Articles to date in this series
Footnotes
-
This article has been peer reviewed.
Competing interests: Sheldon Tobe has received funding for non-peer-reviewed investigator-initiated research from Pfizer and Merck and for industry-supported contract research from Astra Zeneca, Pfizer, Ortho Biotec, Novartis, Bristol Myers Squibb, Sanofi, Amgen, Roche, Merck Frosst and Boehringer-Ingelheim, as well as speaker fees from Pfizer, Bristol Myers Squibb, Merck, Sanofi-Aventis, Amgen, Abbott and Fournier. Finlay McAlister has received operating grant funding from Pfizer Canada for an ongoing trial cofunded by the Heart and Stroke Foundation of Canada and Pfizer Canada. Lawrence Leiter has had paid consultancies with Astra Zeneca, Biovail, Bristol Myers Squibb, Fournier, Merck, Pfizer, Sanofi-Aventis and Servier and has received research support from Astra Zeneca, Bristol Myers Squibb, Novartis, Pfizer, Sanofi-Aventis and Servier as well as speaker fees from Astra Zeneca, Biovail, Merck, Pfizer, Sanofi-Aventis and Servier.
REFERENCES
- 1.
- 2.
- 3.
- 4.