The prevalence of colonic diverticulosis appears to be increasing throughout the world, probably because of changes in lifestyle.1 It is common in developed countries and is slightly more common in the United States than in Europe. In contrast, it is rare in Africa. Diverticular disease is characterized by the presence of sac-like protrusions (diverticula) that form in the colon wall when the mucosa and submucosa herniate through defects in the muscle layer of the wall.2
Although most people with colonic diverticulosis have no symptoms, about 25% will eventually have an episode of symptomatic diverticular disease and up to 5% will have an episode of acute diverticulitis3 (an acute inflammation of the colonic diverticula).2 Along with the increased prevalence of colonic diverticulosis, the prevalence of diverticulitis is increasing.4 Diverticular bleeding is another complication of colonic diverticulosis, but in contrast to diverticulitis, its prevalence is very low.2 In this review, I focus on the diagnosis and management of colonic diverticulitis.
Although we currently have information about the causes, disease course and treatments available to diagnose and manage diverticulitis, treatment options are not well defined because of a lack of evidence. This suggests that there is still considerable work to do in improving our understanding of this disease. Changes in treatment will need to keep pace with new understanding of the disease.
There are few systematic reviews and well-conducted trials to help decision-making in the diagnosis and treatment of diverticulitis. Box 1 summarizes the search strategy used for this review.
Evidence used in this review
This review is based on a search of MEDLINE from January 1990 to March 2012. Proceedings of relevant international congresses were also searched for the same period. The search strategy focused on retrieving articles that addressed diverticular disease or diverticulosis of the colon and on the use of dietary fibre, rifaximin, antibiotics, mesalamine and probiotics to improve symptoms, maintain remission of symptoms, treat acute diverticulitis and prevent acute diverticulitis or recurrent diverticulitis. I excluded retrospective studies and articles addressing the treatment of diverticular hemorrhage, segmental colitis associated with diverticulosis, or outcomes other than clinical outcomes of diverticular disease of the colon.
Few systematic reviews or meta-analyses were found on the treatment of diverticular disease. Several double-blind, placebo-controlled studies were published only in abstract form. Appendix 1 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.120580/-/DC1) provides more details on the search strategy.
How has diagnosing colonic diverticulitis changed?
From a clinical point of view, diverticulitis has generally been considered a “surgical disease.” Abdominal pain or alteration of bowel habits in patients with diverticulosis may have been interpreted as signs of irritable bowel syndrome,5 while a common assumption was that only severe abdominal pain, fever or signs of peritonitis could be interpreted as signs of diverticulitis. In the latter situation, computed tomography (CT) of the abdomen is commonly advised, and diverticulitis is often diagnosed in these patients.
Abdominal CT is still considered the gold standard for diagnosing colonic diverticulitis, in which bowel thickening in the colonic district harbouring diverticula with or without pericolonic fat stranding and, in the case of complicated disease, with abscesses or free perforation may be seen.6 The sensitivity and specificity of abdominal CT for diagnosing colonic diverticulitis are 94% and 81%, respectively, in those with classic symptoms,7 but this approach may underestimate the real prevalence of the disease. Many patients may not show such a dramatic clinical picture but instead may experience milder abdominal pain in the left lower quadrant, diarrhea or constipation, often associated with increased inflammatory markers (e.g., erythrocyte sedimentation rate, C-reactive protein).
Today, the widespread use of colonoscopy in assessing abdominal symptoms has increased the incidence of “uncomplicated” diverticulitis, defined as signs of diverticular inflammation without complications (i.e., abscess, fistula, perforation). Uncomplicated diverticulitis is more frequently diagnosed by endoscopy than would previously have been thought likely. Ten years ago, Ghorai and colleagues reported that findings consistent with diverticulitis may be found in 0.48% of patients who undergo colonoscopy, even if they have no signs or symptoms of the disease.8 These results have been recently confirmed by my group. We found that 1.5% of patients undergoing colonoscopy because of persistent abdominal pain, altered bowel habits or rectal bleeding had findings of diverticulitis.9
If inflamed diverticula are present, there is a risk of colonic perforation during colonoscopy. However, a gentle colonoscopy with minimal air inflation can be carried out safely in patients undergoing a colonoscopy to investigate pain, changes in bowel habits or bleeding. If a diagnosis of diverticulitis is suspected (or confirmed) during colonoscopy, the procedure may be terminated, with a very low risk of perforation.9,10
How is acute diverticulitis currently treated?
The diagnosis and treatment of diverticular disease of the colon are mostly based on single papers; few high-quality randomized trials, systematic reviews or meta-analyses have been published. Despite the lack of evidence from high-quality studies, there is some evidence to support approaches to treatment.
The “blind-pouch” theory, which implicates fecal stasis and bacterial overgrowth in the pathogenesis of such conditions as appendicitis and pouchitis, has been similarly used to explain the development of diverticular inflammation.10 Hence, the first stage of treatment for mild symptoms may involve liquid or low-residue diet initially, along with antibiotic therapy.2,11,12
For outpatients, broad-spectrum antibiotics are usually given for 7–10 days. Various antibiotics may be used in the treatment of acute diverticulitis, ranging from ampicillin to third-generation cephalosporins,2,10,12 as long as there is adequate coverage against gram-positive, gram-negative and aerobic–anaerobic bacterial strains.2,10,12
The combination of ciprofloxacin and metronidazole is a commonly used treatment for uncomplicated diverticulitis,2,10,12 but this combination may be poorly tolerated by some patients. According to a survey by the American Society of Colon and Rectal Surgeons, ampicillin–sulbactam is another common choice for this group of patients.13
If opioid analgesics are required for pain control, guidelines recommend the use of meperidine because morphine causes colonic spasm and may accentuate colonic hypersegmentation.2,10,14
Outpatient treatment is effective in most cases, with a low risk (less than 10%) of emergency department readmission for diverticulitis within 60 days of the initial evaluation.2,10,14 Current guidelines14,15 recommend admission to hospital, with intravenous antibiotic treatment and bowel rest, if the patient is unable to take oral therapy or is affected by severe comorbidity, or if the patient’s condition does not improve with outpatient therapy.2,10
Clinical improvement in patients with acute diverticulitis is generally observed within 3–4 days. For patients admitted to hospital, a 7–10 day course of oral antibiotics is usually given following discharge.2,10,14,15
There are no clear recommendations on whether diverticulitis found incidentally on colonoscopy should be treated. A first diagnosis of diverticulitis by colonoscopy is not rare.8,9 A reasonable approach is to treat diverticulitis found incidentally if there are any relevant symptoms or elevated inflammatory markers (e.g., erythrocyte sedimentation rate, C-reactive protein) as a usual episode of acute diverticulitis as described above. Options for maintenance therapy and monitoring after treatment of the acute episode will be outlined later in this article. If the patient has no symptoms, a reasonable approach could be a short course of antibiotic treatment with clinical follow-up alone.
How is diverticulitis currently managed after an acute episode?
Although recurrence is common, there is little evidence to define the optimal management approach after an acute episode. Several large, long-term studies involving patients initially admitted to hospital for acute diverticulitis have reported that the 5-year recurrence rate is up to 20%.16–18 Several aspects of management after an acute episode (i.e., diet, antibiotic therapy and surgery) are discussed below.
High-fibre diet
After an acute episode of diverticulitis has resolved, patients are generally advised to maintain a high-fibre diet to optimize their bowel movements.15 However, the collective literature investigating the role of dietary modification in preventing diverticular disease or a recurrence of diverticulitis is inconsistent. Even when including only recent studies, the results are conflicting,19–21 and there is not consistent support for recommending a high-fibre diet. Despite this lack of evidence, a high-fibre diet is still commonly recommended.
Despite the classic advice to avoid consuming seeds, popcorn and nuts, which is based on the assumption that such substances could theoretically enter, block or irritate a diverticulum and result in diverticulitis, and possibly increase the risk of perforation, there is no evidence to support this practice.22
Antibiotic therapy
Given the potential involvement of microbial imbalance in the pathogenesis of diverticular disease,23 one option to prevent recurrence after an acute episode may be to use a single, broad-spectrum antibiotic that has activity against both gram-negative and anaerobic bacteria. Recent evidence suggests that rifaximin (a poorly absorbed, broad-spectrum, oral antibiotic) is effective against gram-positive, gram-negative, aerobic and anaerobic bacteria.24 Some studies have shown that cyclic administration of rifaximin (800 mg/d for 10 days every month) can improve symptoms and maintain periods of remission in patients with uncomplicated diverticular disease.24 However, the results of 3 recent systematic reviews in assessing the role of rifaximin in preventing recurrence of diverticulitis are conflicting.24–26 These differing results may be due to varying study quality for this outcome.
From a pathophysiological perspective, the ineffectiveness of rifaximin in preventing diverticulitis recurrence may be caused by the short-lasting effect of the reduction of fecal bacterial during oral treatment with rifaximin. The bacterial population of the colon (mostly Escherichia coli, Bacterioides spp. and anaerobic cocci) recovers within 1–2 weeks after the end of treatment.27 Thus, repeated oral administration of rifaximin may control the colonic bacterial population for only 15–20 days, with higher colonic bacterial concentrations for the last 10 days of the month, therefore leading to a higher risk of recurrence. There is no evidence to support the use of other antibiotic regimens for the prevention of diverticulitis recurrence.
Surgery
Surgery is a therapeutic option after episodes of diverticulitis. According to guidelines by the American Society of Colon and Rectal Surgeons12 and others,28 elective resection should be considered after 1 or 2 well-documented episodes of diverticulitis, depending on the severity of the attack and the age and medical fitness of the patient. However, data indicate that 25% of patients who had elective surgery for diverticulitis experienced persistent abdominal symptoms.29 Neither the stage of disease (complicated or uncomplicated) nor the surgical technique (laparotomy or laparoscopy) were significantly related to the occurrence of symptoms.29 A more individualized approach taking into account the frequency, severity of the attacks and their impact on quality of life should guide the indication for surgery.28
How can new insights into the pathogenesis of diverticular disease guide practice?
The pathophysiology of diverticular disease is extremely complicated because of the multifactorial contributing factors, including diet, colonic wall structure, intestinal motility and possible genetic predisposition.2 However, recent observations suggest that the natural history of the disease bears many similarities to that of chronic inflammatory bowel diseases. For example, low-grade inflammation may be detected in patients with diverticular disease30,31 (although not all studies confirm this32). The extent of this inflammatory infiltrate appears to be related to the severity of the disease,33 and the inflammation may be detected by fecal calprotectin.34
Several studies have suggested that chronic inflammation and its impact on neuromuscular function in the colon may be partially responsible for symptomatic diverticular disease.35 In these studies, symptomatic patients had a greater relative expression of receptors for proinflammatory molecules (e.g., neurokinin 1, tumour necrosis factor-α) compared with asymptomatic patients.36,37
Persistent inflammation may be a risk factor for recurrent disease. We know that diverticulitis recurs in many patients despite surgical treatment and that symptoms persist in 25% of patients following sigmoid resection.29 Recent evidence suggests that the detection of inflammation during follow-up after an acute episode of diverticulitis is a risk factor for recurrence.38 Ongoing inflammation may be detected in patients with symptomatic diverticular disease by fecal calprotectin assessment, an acute phase protein that is increased in diverticular disease, but not in healthy people or those with irritable bowel syndrome.34 (The use of C-reactive protein or erythrocyte sedimentation rate for monitoring after an acute episode of diverticulitis has not been studied in this context.) Data from studies showing that anti-inflammatory agents, such as mesalamine (5-aminosalicylic acid [5-ASA]), improve symptoms in patients with diverticular disease also support an association between inflammation and the onset or persistence of symptoms.39
Because of these advances in our understanding, new therapeutic approaches are under active investigation in treating diverticular disease. In particular, mesalamine and probiotics are currently being investigated. Preliminary results from randomized, placebo-controlled trials on mesalamine are outlined below.
Mesalamine in diverticulitis
Mesalamine is the primary therapy used for induction and maintenance of remission of mild to moderately severe inflammatory bowel disease, in particular ulcerative colitis, thanks to its anti-inflammatory and immunomodulatory properties. The influence of mesalamine on the signs and symptoms of diverticular disease has been investigated in a number of studies. These clinical studies have shown a consistent benefit of mesalamine thus far; however, they have all used an open-label design. The results from some recent randomized, double-blind, placebo-controlled studies have been presented as abstracts. A multicentre 6-week study reported that mesalamine (3 g/d) was significantly better than placebo (56% v. 67%, p = 0.05) in controlling moderate-to-severe abdominal pain caused by diverticular disease.40
The use of mesalamine in preventing the recurrence of diverticulitis, however, may be more interesting. In the 2 open-label studies addressing this issue, the use of mesalamine, in combination with rifaximin or with a probiotic preparation, was effective in preventing recurrence within 3 months (rifaximin plus mesalazine v. rifaximin, 2.8% v. 12.38%, p = 0.005;41 probiotic plus balsalazide v. probiotic, 26.27% v. 40%, p = 0.01).42 Balsalazide acts by releasing mesalamine in the colon.
Three double-blind, placebo controlled studies43–45 have recently assessed the role of mesalamine in preventing recurrence of diverticulitis. The DIVA trial found that 3 months of mesalamine treatment (2.4 g/d) after an episode of acute diverticulitis, confirmed by CT, was superior to placebo in reducing gastrointestinal symptoms for up to 1 year; however, this study was underpowered to evaluate recurrent diverticulitis.43 In particular, mesalamine was significantly better than placebo in reducing a global symptomatic score at 12 weeks (56.3% v. 17.2%, p = 0.001) and at 52 weeks (59.3% v. 27.3%, p = 0.045). There were no differences in diverticulitis recurrence rates, surrogate markers or safety outcomes.
The DIV/04 trial reported similar efficacy with intermittent mesalamine treatment (1.6 g taken for 10 days per month) or placebo (p = 0.021), as well as a decrease in the relative risk of recurrence after 24 months (p = 0.48, 95% confidence interval [CI] 0.20–1.15).44
The most recent trial found that mesalamine (514.7 ± 30.5 mg/d) was significantly better than placebo in reducing the risk of diverticulitis over a 40-month period, as well as the number of diverticulitis Wares and the need for surgery.45 The relative risk of diverticulitis was 2.47 times higher (95% CI 1.38–4.43) in the placebo group than in the mesalamine group.45
Two large double-blind, dose-finding, placebo-controlled studies of multi-matrix system mesalamine (MMX®) in preventing diverticulitis recurrence have recently concluded (Prevention of Recurrence of Diverticulitis [PREVENT-1 and -2]).46,47 The final results are not yet available, but should add to our knowledge in this area, given the studies’ solid design and impressive power.
Although these data about the effectiveness of mesalamine in preventing recurrence of diverticulitis seem promising, the results should be interpreted with caution. In some cases, the study results have been published in abstract form only, and the results from the full study have yet to be published. Even if mesalamine is shown to reduce the recurrence of diverticultitis, an appropriate cost–efficacy analysis has not been completed. We do not know if cyclic, life-long, mesalamine administration to prevent diverticulitis recurrence is cheaper than treating diverticulitis with a 7-day course of antibiotics only for patients with disease recurrence. This important point needs to be addressed in future studies.
Probiotics
Probiotics are living microorganisms that, if consumed in sufficient numbers, can alter host microflora and exert specific health benefits without increasing the risk of antibiotic resistance.44 They commonly include the bacteria Bifidobacterium spp., Lactobacillus spp. and certain strains of E. coli, and the budding yeast Saccharomyces boulardii.44 Probiotic microorganisms appear to have multiple modes of action, including inhibition of pathogen adherence, improving mucosal defence by enhancing the integrity of tight junctions, decreasing bacterial translocation, stimulation of immunoglobulin A secretion in Peyer patches and enhancement of immune system activity by controlling the balance of pro-and anti-inflammatory cytokines.48 Probiotics may also interfere with pathogen metabolism48 and have the ability to restore balance to enteric flora, primarily by decreasing the number of pathogenic gram-negative bacteria.48
Some open-label studies have evaluated the role of probiotics in the treatment of diverticular disease and its complications.49,50 Although most of the strains investigated (e.g., Lactobacillus casei subsp. DG or Lactobacillus paracasei F19) in these studies seem to be effective in preventing the onset or recurrence of diverticulitis, well-designed studies on the role of probiotics in preventing diverticulitis recurrence are lacking.51
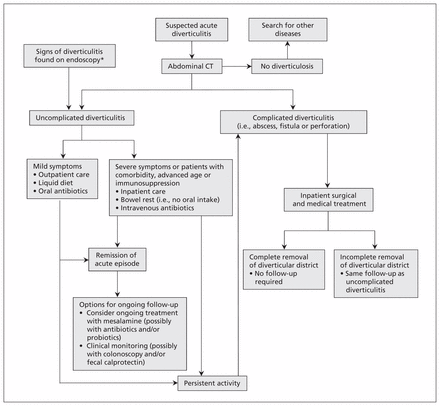
Figure 1 outlines an approach to the management of diverticulitis, based on current guidelines, the available literature and expert opinion.1,2,24–26,39–47,51 Box 2 provides an example of how this algorithm may work in practice.
Applying the results of this review in clinical practice (fictional case)
Mrs. S is a 60-year-old woman who is slightly overweight and has hypertension and hypercholesterolemia. She experiences recurrent episodes of abdominal pain localized in the left lower abdominal quadrant; the pain is associated with diarrhea. Her physician orders laboratory investigations and finds that her levels of C-reactive protein (0.13 [normal < 0.05] mg/L) and erythrocyte sedimentation rate (23 [normal ≤ 10] mm/h) are elevated. Because of these increased inflammatory markers, her physician recommends colonoscopy. The endoscopist reports the following: “Left colon shows several diverticular pockets, most of them located in the sigmoid region. Some diverticular openings show edema; others show intense hyperemia of the peri-diverticular mucosa.” Based on these colonoscopy findings, a diagnosis of uncomplicated diverticulitis is made. Mrs. S undergoes a 7-day course of ampicillin-sulbactam, and she is advised to increase her fibre intake, although the evidence is conflicting for this approach.
Eight months after the endoscopic diagnosis of diverticulitis, and despite normal serological inflammatory values and increased fibre intake, Mrs. S continues to report abdominal pain, associated with recurrent episodes of diarrhea. Under advice of her physician, Mrs. S starts taking mesalamine (1.6 g/d), although this is off-label use. Her symptoms improve shortly after starting the drug. Twelve months later, she continues to feel well. No additional symptoms or diverticulitis recurrence are reported.
Conclusion
The diagnosis and treatment of diverticulitis are changing. Computed tomography remains the gold standard technique for diagnosing acute disease and has a higher predictive value on clinical outcome than clinical assessment. Antibiotics remain the mainstay of treatment of the acute phase.
Because of the increased number of colonoscopies performed, however, most cases of diverticulitis are currently diagnosed by colonoscopy. Most of these patients do not show any clinical signs of acute diverticulitis. The optimum approach to managing these patients is unclear.
Diverticulitis has a substantial rate of recurrence, and long-term treatment to prevent recurrence of the disease or its complications is required. While dietary modifications are often recommended, evidence supporting their use is absent or conflicting. Because surgery does not guarantee complete disappearance of symptoms after an episode of acute diverticulitis, medical treatment may prove to be the best approach in the long term. Monthly courses of antibiotics, such as rifaximin, may be prescribed, but the beneficial effects appear short-term. Mesalamine seems to be very promising for preventing recurrence, but doses and schedules are still unclear, as is the role of probiotics.
With recent advances in our understanding of this complex disease, new therapeutic approaches to the management of diverticulitis may further change practice.
Key points-
About 25% of patients with diverticulosis will develop symptoms, and about 5% will develop an episode of acute diverticulitis.
-
Antibiotics are recommended for the treatment of acute diverticulitis, but the optimal treatment of symptomatic uncomplicated diverticular disease, as well as the medical strategy in preventing diverticulitis recurrence, is unknown.
-
Mesalamine may be effective in treating diverticular disease and preventing recurrence; however, additional studies are needed.
-
Probiotics show promise in treating the disease; however, current data are too preliminary to draw definitive conclusions.
Footnotes
-
Competing interests: None declared.
-
This article has been peer reviewed.