Abstract
MANY GENETIC DISORDERS DO NOT MANIFEST themselves until the adult years. Such disorders often involve multiple genetic factors interacting with multiple environmental factors, over time, to produce a phenotype. This paper reviews the modes of inheritance of genetic disorders and describes the types of genetic testing that are currently available. It offers clues that should lead physicians to suspect that an adult patient might have a genetic disorder and raises issues that should be considered in counselling the patient about genetic testing. Resources for patients and their family physicians are also discussed.
Case
A 35-year-old man, who is a new patient, presents at your office complaining of the insidious onset of weakness and clumsiness over the last 2 years. As you greet the patient, you note that his face seems somewhat “droopy” and expressionless. He has difficulty letting go of your handshake. Before he sits down, he tells you that this is a family problem; his sister, mother, maternal uncle and maternal grandfather are similarly affected. The family wonders whether this might be a hereditary problem, but no one has yet sought genetic diagnosis.
The range of genetic disorders is so broad in nature that affected individuals can be found in the practice of virtually all generalists and specialists. Simply put, our genetic blueprint influences our health from the moment of conception until death.
Based on a population study of genetic disorders apparent by the age of 25 years,1 about 0.4% of the population have a single gene (Mendelian) disorder, 0.2% have a chromosomal abnormality and 4.6% have a multifactorial condition. Another 0.1% have an obvious genetic abnormality of unknown inheritance, and 0.3% have congenital problems that are not genetic in nature.
An adult with a genetic disorder diagnosed in childhood, such as congenital heart disease or Down's syndrome, might be seen in a physician's practice. Or a physician might encounter a new patient whose syndrome was not diagnosed in his or her younger years. Alternatively, patients may present with new-onset signs and symptoms of a genetic disorder in early or middle adulthood or even after retirement, because many disorders of adult onset require years to become symptomatic.
In fact, many disorders of genetic origin do not manifest themselves until the adult years. Most of these fall into the multifactorial category.2 That is, multiple genetic factors interact with multiple environmental factors, over time, to produce a phenotype. When one thinks of common disorders such as hypertension, heart disease, diabetes and cancer, one has to wonder whether there is any significant adult-onset disorder that does not have some genetic basis. Indeed, as the genetic code is deciphered, we are gaining an increased appreciation for the ubiquitous genetic contribution to adult health and illness.
Mode of inheritance
Understanding the mode of inheritance of a genetic condition will help the physician to diagnose the disorder and predict recurrence in future generations.
Cytogenetic
Cytogenetic abnormalities,3 also known as chromosomal abnormalities, are responsible for many of the genetic conditions diagnosed in the perinatal period, infancy and childhood. However, other than individuals with cytogenetic disorders who survive to adulthood, or escape diagnosis in childhood, the cytogenetic diagnosis is rare in the adult population. Prominent exceptions are chromosome abnormalities that present with problems of reproduction.
It is important to appreciate that cytogenetic abnormalities can occur in either the germ line or in somatic tissues. When we are looking for a genetic diagnosis, we are usually looking in the germ line. The search for somatic mutations is the basis for much of cytogenetic analysis important to cancer prognosis.
Single gene disorders (or Mendelian disorders)
The concepts of dominant, recessive and X-linked inheritance have been with us for decades. Many single gene disorders produce defined diseases of adult life (Table 1). As our understanding of the mechanisms of gene action has improved, however, it has become clear that the paradigm of “one gene — one protein — one disease” does not hold for many disorders. For example, different mutations to the dystrophin gene4 on chromosome X can produce 2 quite distinct clinical disorders: Duchenne's muscular dystrophy and Becker's muscular dystrophy. A duplication of a specific gene5 on chromosome 17 produces Charcot-Marie-Tooth type Ia; a deletion in the same gene produces “hereditary neuropathy with tendency to pressure palsy.”
Table 1.
Some gene mutations may be capable of both dominant and recessive action, depending on the context.6 In metabolic disorders,7 the heterozygous individual may have mild signs and symptoms of the more serious manifestation of the homozygous or hemizygous state. This is particularly true in the X-linked “recessive” disorders. In Fabry disease,8 the hemizygous male has serious manifestations of vascular, renal and skin disease. The “carrier” female may have milder but still significant manifestations of the same nature. The same situation may be seen in autosomal “recessive” conditions. A single mutation in one of the genes in the homocysteine pathway9 may produce hyperhomocysteinemia that may be inherited in a dominant fashion in the family and predispose to premature atherosclerosis. Should both alleles of a specific gene in the pathway be mutated, the more severe syndrome of homocystinuria is present.
Clearly, many mutations inactivate genes. However, gene mutations do not always result in loss of function. Some mutations stimulate the functioning of genes and this can also be detrimental to health. For example, a mutation to the superoxide dismutase10 gene accelerates gene function and results in a rare form of familial amyotrophic lateral sclerosis.
Genes do not always have the same effect in different circumstances. A mutation to the tumour suppressor gene p5311 may initiate the familial Li-Fraumeni cancer syndrome. But, alterations to p53 are also commonly found in other tumours, both inherited and sporadic, suggesting that p53 mutations modify the course of many types of cancer.
Some gene mutations do not always produce the same effect even in the same family. The mutation for early onset torsion dystonia12 has a penetrance of 30%–40%. That is, 60%–70% of individuals with this gene do not have clinical signs or symptoms of dystonia. When the gene is expressed, the consequences vary from being very mild to devastatingly severe. These phenomena of incomplete penetrance and variable expressivity make the prediction of natural history for any individual or family very difficult.
Germ-line mutations may not always be present in all tissues of the body. Occasionally, a mutation may occur very early in embryogenesis and only be present in tissues derived from a single mutant cell. This condition, known as mosaicism, may produce a limb or organ affected by a genetic disorder while the rest of the body is unaffected. Mosaicism may also account for rare instances in which monozygous twins are discordant for a genetic condition.
Nontraditional
Whereas most genetic material is contained in the cell nucleus, some genetic material resides in the mitochondria. Almost all mutations in mitochondrial DNA13 are inherited from the mother, because sperm have very few mitochondria compared with eggs. In fact, this significant difference between the transmission of mitochondrial and nuclear genetic mutations has allowed archeological geneticists to use mutations in mitochondrial DNA to estimate the time frames for the branching of our global human tree.14 The spectrum of mitochondrial disorders is rapidly expanding and includes many multisystem disorders involving sight, hearing, muscle function and the brain.
Another form of nontraditional inheritance involves imprinted genes.15 That is, some genetic abnormalities function differently depending on the parent of origin. The best examples of this are the childhood syndromes of Pradi-Willi and Angelman. Both result from deletions to the same genetic region, but in Prader-Willi the deletion is always paternally derived, and in Angelman it is always maternally derived.
Multifactorial
As mentioned earlier, the vast majority of genetic disorders that are not exhibited until adulthood have multifactorial causes (Table 2). Indeed, most of the extremely common disorders in adults, such as hypertension, heart disease and cancer, are multifactorial in nature. It is not possible to assess the effects of one gene out of the context of the full genetic complement. Nor can we ignore the environment — past, present and future. The current epidemic in diabetes is not because of a massive populationwide mutation in our genetic makeup. Rather, it is the effect of years of the Western diet that is high in fats and simple carbohydrates exerting its influence on the genetic underpinnings of the ancient hunter–gatherer.
Table 2.
When to consider a genetic disorder
The same considerations that go into adding a possible genetic disorder to a neonate's or child's differential diagnosis apply to adults. Is this disorder genetic or acquired? How significant is the genetic component versus the environmental component? What is the pattern of inheritance? Is there potential for other members of the patient's family to be affected presently or in the future? How can the diagnosis be confirmed? Is any specific treatment available for this genetic disorder, or is the treatment based on clinical grounds regardless of diagnosis?
There are a few clues that will suggest a genetic diagnosis in an adult. The family history might point to a genetic disorder. A great many disorders of adult onset are dominant in nature. That is, a mutation in only one of a gene pair is necessary to produce a genetic problem. (This contrasts with recessive inheritance in which both genes of a pair must have a mutation.) Dominant genetic disorders are very likely to produce a family history, whereas recessive disorders are not. Unfortunately, some dominant conditions have a high new mutation rate, so your patient might well be the first case in the family. Alternatively, a positive family history may not have previously been appreciated, perhaps because of the small size of the family or dispersion of family members. Family members who carried a gene mutation may have died before they developed signs and symptoms, or a genetic diagnosis may have never previously been entertained despite the existence of affected relatives.
Pedigree analysis may be complicated by the phenomena of incomplete penetrance and variable expressivity.16 Incomplete penetrance means that some individuals may carry a genetic mutation but not express any signs and symptoms. Variable expressivity means that even though all affected members of the family have the same mutation, they may not have identical manifestations. Age of onset and severity of the condition may vary considerably.
Many disorders known to have a genetic cause are diagnosed on clinical grounds, such as the biopsy that reveals a neurofibroma17 or the iron-filled liver of hemochromatosis.18
When a disease occurs at a much younger age than one would normally expect, it suggests a possible genetic predisposition. For example, breast cancer in a 25-year-old is very unusual.19 Paired with a history of breast and ovarian cancer in close relatives, this is virtually diagnostic of a mutation to a BRCA gene.20
Physicians should also consider a genetic cause in 3 additional circumstances: cases with multisystem involvement (e.g., the deafness and nephritis associated with Alport's syndrome21), with a multifocal presentation (e.g., the multiplicity of polyps throughout the colon diagnostic of familial adenomatous polyposis22) or with an unusual combination of events (e.g., early onset osteoporosis and conductive hearing loss may be indicative of osteogenesis imperfecta23).
Diagnosis
The possibility of a genetic disorder must be entertained, perhaps triggered by the clues described above, before a diagnosis can be reached. The basis of this diagnosis is still mostly clinical, based on the patient's signs, symptoms and a good family history. Routine clinical investigations may be virtually diagnostic. In fact, in some cases clinical testing may be more sensitive than our currently available genetic testing. For example, extreme multiplicity of polyps makes the diagnosis of familial adenomatous polyposis,24 whereas the molecular testing usually available in hospitals picks up only the 15% of cases caused by the 2 common mutations.
While new molecular diagnostic tests are being developed and the range of available tests is increasing in many centres, we still do not have a genetic test for the vast majority of genetic disorders. Many genes have not yet been localized. Even when a gene location is discovered by a research laboratory, this may not quickly translate into a funded and easily accessible test at your local laboratory.
Current modalities of genetic testing
Cytogenetics
The standard cytogenetic tool is a simple count of chromosomes. In this way it is possible to pick up cases of “aneuploidy,” namely, the presence of supernumerary chromosomes. The most common example is Down's syndrome, which is unlikely to escape detection in infancy or childhood. However, cases of Turner's syndrome25 and Klinefelter's syndrome26 may not be picked up until such patients are seen for infertility. Detailed inspection of the genome3 may pick up large loss (deletion), gain (duplication) or rearrangements (translocations and inversions) of genetic material. These may be important in the diagnosis of individuals with dysmorphic features and an intellectual handicap. The search for translocations and inversions may be important in the investigation of recurrent pregnancy loss27 where, in about 4% of couples, one partner will be found to have a cytogenetic abnormality as an underlying cause. Finally, cytogenetics is moving into ever more sophisticated diagnostics with a technique known as FISH28,29 (fluorescence in situ hybridization) that can pick up deletions, duplications or rearrangements of genetic material not visible by usual cytogenetic techniques.
Specific molecular diagnostics
In its most accurate form, molecular diagnostics29,30 can look for a known specific mutation to a known specific gene such as the single base change in the beta globin gene responsible for sickle cell anemia.29 Another extremely reliable form of molecular diagnosis is in the trinucleotide repeat disorders.31 Most of these disorders are neurodegenerative in nature such as myotonic dystrophy, Huntington's syndrome, spinocerebellar ataxias and fragile X syndrome. Standard numbers of repeats are known for each target gene within a naturally occurring stretch of repeats. When there is expansion of the repeat segment, genetic disease occurs. There is a rough correlation between the increased number of repeats and the severity of the resulting phenotype.
Molecular screening
Most genetic mutations (about 80%) produce truncated proteins.30 The search for these shortened pieces of protein product can yield a likely corollary genetic area to sequence for causative mutations. The techniques of SSCP (single-strand conformation polymorphism) and DGGE (denaturing gradient gel electrophoresis) separate bits of DNA by electrophoretic mobility to target likely areas for total gene sequencing. Microarray technology (also known as gene chips) will streamline the process so that hundreds of procedures can be carried out at once, making molecular diagnostics both faster and cheaper.32
Sequencing
This is the gold standard for finding gene mutations. However, it is prohibitively expensive both in time and equipment, so we are not currently at the point of sequencing for many disorders in service laboratories. One of the confounders of sequencing30 is whether an abnormality is truly pathogenic or merely a polymorphism, a benign change in a few percent of the population that does not produce pathology.
Linkage
This is one of the earliest forms of genetic diagnosis29 and is still used occasionally. Unfortunately, it is also very time and labour intensive and fraught with complications. First, one must have DNA samples (usually from fresh blood) from as many firmly diagnosed affected and unaffected family members as possible. Then, the laboratory chooses markers either within the gene or close to the gene to compare the affected with the unaffected haplotypes. Such investigations are made difficult when sufficient family members cannot be recruited for study, by the quality of the markers in question and their distance from the gene of interest, and the natural tendency of DNA to recombine at the time of reproduction, therefore producing different sequences of genetic information on chromosomes transmitted from parent to child.
Thanks to the Human Genome Project,33 the entire genome has now been sequenced but we do not yet know the function of most sequences. Understanding the genetic code may be compared to constructing a puzzle in which most of the puzzle pieces are missing and the overall picture is unknown. We must await technological advances to reach the stage where a genetic analysis is as simple and inexpensive as a complete blood count.
Management
Replacement therapy has been developed for a small number of inherited disorders of metabolism such as Gaucher's disease,34 but there are few other specific therapies for genetic disease.35 By and large, adult-onset genetic disorders are managed by treating the manifestations of the disorder clinically (e.g., surgery for cancer or drugs for lipid disorders). In many cases, such as most of the inherited neurodegenerative disorders, there is no treatment at all.
Resources again are the limiting factor. For common genetic disorders, there may well be dedicated clinics, especially in large academic centres. For rarer genetic disorders, and certainly in small centres, affected individuals may have easy access only to their family physician. In the latter circumstances, the family physician is best armed by knowledge of the closest genetics referral centre, and Web-based information sites may be of great value.
The knowledge that a patient is predisposed to a specific disorder, whether it be premature atherosclerosis or breast or ovarian cancer, may provide significant opportunity for preventive medicine. Aggressive surveillance programs using diagnostic radiology or endoscopy or specific prophylactic therapy through surgery or pharmaceutical treatment may reduce future morbidity and mortality. The patient may use this information when making important life choices such as those pertaining to choice of occupation and reproduction.
Counselling
Extensive counselling can often help individuals and families understand their predisposition to genetic disease. The following issues should be discussed with patients before they undergo genetic testing.
The genetic test's limitations must be clearly outlined. Testing may not produce a clearly positive or negative result. A noninformative test result does not mean that the patient does not have the disorder in question; it usually means that a definitive answer is beyond the limits of our current technology. For example, a service molecular laboratory may test for the 32 most common mutations (90% of all mutations) in cystic fibrosis, although more than 500 have been described in the literature.36 A patient suspected of having adult-onset cystic fibrosis may well have one clear mutation and one of the very rare mutant alleles that routine testing would not pick up as an abnormal.
The limitations of treatment must be very clear. Just because a genetic disease has been diagnosed does not necessarily translate into a cure or even a surgical or pharmaceutical treatment that may delay disease progression or ameliorate symptoms.
There may be potential for prenatal diagnosis. Would the prospective parents be truly interested in pregnancy termination, even if signs and symptoms would not manifest until adult years? Should a fetus be tested if the parents intend to carry the pregnancy to term but there is no treatment, or treatment would not be started until years later?
Individuals may be discriminated against on the basis of a known genetic diagnosis even before symptoms occur. Many Americans decline presymptomatic genetic testing for fear of losing health care coverage. In both the United States and Canada, individuals may experience difficulty in obtaining life insurance.37
If the candidate for testing is a child or adolescent, additional considerations arise.38 Most geneticists take the position that minors should not be tested unless clear clinical benefit is evident. Otherwise you have merely procured knowledge of no benefit and created the potential for detriment.
Resources
There are genetics clinics in many large hospitals in Canada, particularly those associated with university centres. Questions regarding genetic assessment, whether clinical or laboratory, may be addressed to these centres. Specialized clinics such as adult cystic fibrosis or cancer genetics clinics are also excellent resources. Beyond their clinical functions, disease-specific clinics are often excellent doorways to research for interested patients and their families.
National patient support groups exist for the more common genetic disorders and may be a tremendously valuable resource for affected individuals and their families. For example, the Canadian Muscular Dystrophy Association is an umbrella group for many of the genetically inherited neuromuscular disorders. Some families prefer not to become involved in patient support groups, and this position must be respected. And, for better and for worse, patients and health care providers alike can access the Web to obtain information of wide-ranging validity.
The case revisited
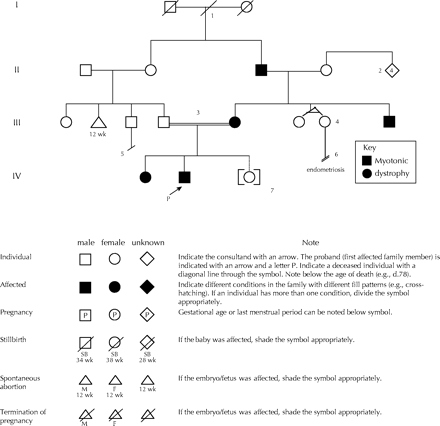
You obtain a history from your patient. He is having difficulty with his employment as a construction worker. He can no longer climb a ladder and is unable to carry any weight greater than 4.5 kg because of muscle fatigue. At home, he has difficulty climbing the stairs and has even had a few falls when his “feet became tangled.” He has started driving his car, which is an automatic, with both feet, using the right for the accelerator and the left for the brake, because he cannot get the right foot to work quickly enough to use it alone. He states that the family members mentioned previously are similarly affected. You also learn that his grandfather is now in a wheelchair at the age of 83 years and that this man has diabetes, cataracts and a pacemaker. Your patient's mother also has diabetes. The patient and his affected male relatives all have frontal balding, whereas other male family members have full heads of hair. You note on physical examination that the patient is generally weaker than you would expect for a 35-year-old male and, in addition to release problems, as in letting go of your handshake, he also has percussion myotonia.
With this history, you are very suspicious of myotonic dystrophy39 (Table 3), which is one of the most common of the neurodegenerative disorders with a population incidence of about 1 in 8000. You decide to refer the patient to your local medical genetics clinic for further counselling regarding the inheritance of this disorder.
Table 3.
Once the referral has been received in the medical genetics clinic, it is usually assigned to a medical genetics counsellor. These are individuals who have a Master's degree in genetic counselling. The counsellor will contact the patient and obtain information regarding the family. The pedigree will be constructed (Appendix 1, Fig. 140) and confirmatory records for family members may be requested. Once all the relevant information is obtained, the patient will be booked for an appointment with the geneticist (an MD with training in medical genetics). The geneticist and genetic counsellor will then meet with the patient (and any family members the patient wishes to accompany him or her). As at any other medical appointment, the physician will obtain a personal history and perform a physical examination. The pedigree will be discussed so that the patient may add or alter information as appropriate. The geneticist will then discuss the diagnosis based on all the information available. Should there be a differential diagnosis, this will also be discussed. Options for investigation, including specific genetic diagnostics, will be explored. Limitations of testing and all the pros and cons of testing will be discussed. Genetic testing will proceed only with the patient's full consent, and written consent for testing may be obtained for certain disorders. Counselling will cover the pattern of inheritance, the manifestations and prognosis of disease, and the risk of recurrence for offspring. For disorders of dominant inheritance, the risk that the gene mutation has been passed by the affected parent to the offspring is 50%. Discussion of further testing for the family, including the advisability (or not) of testing children will be addressed. The patient may be referred to neurology for more specific neurological testing (e.g., electromyography) and advice regarding management. Alternatively, this referral may be left to the judgement of the referring physician. The patient will also be given information about appropriate patient support groups. Usually, a further option for more counselling may be given, particularly when the results of genetic testing become available. The results of genetic testing may be available in weeks, sometimes months. In the case of research genetic testing, results may not be available for years, or perhaps may never become available. All information will be summarized in a letter that will be forwarded to the patient's physician(s).
Fig. 1: Drawing the patient's (P) pedigree. Note: 1. The slash in the relationship line indicates either divorce or separation. 2. This symbol indicates 4 siblings of unspecified sex. 3. The double line indicates consanguinity (in this case first cousins). 4. These siblings are monozygous twins. Dizygous twins are not joined by a horizontal line. If the type of twinning is unknown, a “?” replaces the horizontal line. 5. No children by choice or the reason is not known. 6. Infertility, indicate the reason if known. 7. Adopted into family. Children adopted out are drawn with a solid line of descent. 40
What does the future hold?
We will continue to further elucidate genetic mechanisms for single gene, gene–gene and gene–environment interactions. For known disorders, we may further refine genotype–phenotype correlations. That is, where gene mutations occur in several positions in the gene, there may be variance in the constellation of signs and symptoms.
We will grapple ever more increasingly with ethical issues. Dolly the sheep, cloning humans or body parts, and the use of embryonal tissue for research have all been highlighted in the popular press. Who will control our genetic information? What could happen if decisions about you were made by others based on your genetic blueprint, as in the film GATTACA? Are we entering an entirely new era of eugenics?
Summary
Genetic contribution to adult disease is common but, unfortunately, genetic disorders are often overlooked in the differential diagnosis. We now have a significant ability to diagnose genetic disorders, considerable information regarding prognosis, and some ability regarding treatment or prevention. It is definitely worthwhile to pursue a genetic diagnosis, both for the patient and for the patient's family.
The successful completion of the Human Genome Project marks the end of a decade of exponential growth in our understanding of genetic disorders. The sequencing of the human genome is only the beginning. Progress in identifying the links between specific gene mutations and specific disorders and in understanding the mechanisms by which genes influence health will add to our arsenal of tools to address the health care needs of our patients.
Additional resources
-
General genetics texts
-
Thompson WM, McInnes RR, Willard HF, editors. Genetics in medicine. 5th ed. Philadelphia: WB Saunders; 1991.
-
Harper PS. Practical genetic counselling. 5th ed. Boston: Butterworth-Heinemann; 1998.
-
Kingston HM. ABC of clinical genetics. 2nd rev. ed. London: BMJ Publishing Group; 1997.
-
Dysmorphology texts
-
Jones KL, editor. Smith's recognizable patterns of human malformation. 5th ed. Philadelphia: WB Saunders; 1997.
-
Buyse ML, editor. Birth defects encyclopedia. Dover (MA): Blackwell Scientific Publications; 1990.
-
Cytogenetics text
-
Gardner RJM, Sutherland GR. Chromosome abnormalities and genetic counselling. New York: Oxford University Press; 1996.
-
Molecular diagnostics texts
-
Jameson JL, editor. Principles of molecular medicine. Totowa (NJ) : Humana Press; 1998.
-
Elles R, editor. Molecular diagnosis of genetic diseases. Totowa (NJ): Humana Press; 1996.
-
Disease-specific texts
-
Baraitser M. Genetics of neurological disorders. 3rd ed. New York: Oxford University Press; 1997.
-
Beighton P. McKusick's heritable disorders of connective tissue. 5th ed. Toronto: Mosby; 1993.
-
Foulkes WD, Hodgson SV. Inherited susceptibility to cancer: clinical, predictive and ethical perspectives. New York: Cambridge University Press; 1998.
-
King RA, Rotter JI, Motulsky AG. The genetic basis of common diseases. New York: Oxford University Press; 1992.
-
Morgan SH, Grunfeld JP. Inherited disorders of the kidney. New York: Oxford University Press; 1998.
-
Nora JS, Berg K, Nora AH. Cardiovascular diseases: genetics, epidemiology and prevention. New York: Oxford University Press; 1991.
-
Rosenberg RN, Prusiner SB, DiMauro S, Barchi RL, Kunkel LM. Molecular and genetic basis of neurological disease. Toronto: Butterworth-Heinemann; 1993.
-
Royce PM, Steinmann B. Heritable connective tissue and its disorders: molecular, genetic and medical aspects. Toronto: Wiley-Liss; 1993.
-
Scriver CR, Beaudet AL, Sly WS, Valle D. The metabolic and molecular bases of inherited disease. 7th ed. New York: McGraw-Hill; 1995.
-
Sybert VP. Genetic skin disorders. New York: Oxford University Press; 1997.
-
Tuddenham EGD, Cooper DN. The molecular genetics of haemostasis and its inherited disorders. New York: Oxford University Press; 1994.
-
Web site resources
-
· Online Mendelian Inheritance in Man (OMIM): www.ncbi.nlm.nih.gov/omim
Articles to date in this series
-
This series is now complete.
-
Polifka JE, Friedman JM. Medical genetics: 1. Clinical teratology in the age of genomics. CMAJ 2002; 167(3): 265-73.
-
Hunter AGW. Medical genetics: 2. The diagnostic approach to the child with dysmorphic signs. CMAJ 2002; 167(4):367-72.
𝛃 See related article page 1032
Footnotes
-
This article has been peer reviewed.
Acknowledgements: I wish to acknowledge Stephanie A. Kieffer for assistance in drawing the pedigree information.
Competing interests: None declared.
Series editor: Dr. Judith Hall