Antiplatelet therapy is a key component in the management of many medical diseases to decrease the risk of cardiovascular events.1 Acetylsalicyclic acid (ASA) is the most common antiplatelet agent used and is usually first-line therapy. Use of clopidogrel, especially in combination with ASA, has become commonplace in the management of several cardiovascular diseases in the past decade. Recently, newer, more potent antiplatelet agents, chiefly prasugrel and ticagrelor, have become available.
Antiplatelet therapy has an established role in disease states such as coronary artery disease, myocardial infarction and diabetes, by preventing myocardial infarction, stroke and death.1 In atrial fibrillation and stroke, it reduces the incidence of subsequent cerebrovascular events.1 Although antiplatelet therapy is important in the management of various vascular disease states (including peripheral vascular disease), it is particularly useful in acute coronary syndromes where rupture of coronary artery plaque leads to release of inflammatory mediators, platelet activation and subsequent thrombosis. Although ASA is well established as first-line therapy in acute coronary syndromes,2 new evidence supports the role of newer antiplatelet agents.
In this article, we review recent advances and practical applications of the new antiplatelet agents, specifically prasugrel and ticagrelor because they are currently available on the market worldwide. Because the strongest evidence supporting their use is currently limited to acute coronary syndromes, we have focused our review in this area. The use of these agents in acute coronary syndromes is supported by large, randomized controlled trials and recent clinical guidelines. The evidence used in this review is described in Box 1.
Evidence used in this review
We searched MEDLINE, the Cochrane Database of Systematic Reviews and Embase using the terms “clopidogrel,” “prasugrel,” “ticagrelor,” “antiplatelet,” “thienopyridine,” “myocardial infarction,” “acute coronary syndrome,” “percutaneous coronary intervention,” “coronary stent” and “coronary artery disease” for landmark studies, reviews and meta-analyses on antiplatelet use in myocardial infarction published from 2000 to 2012.
We reviewed guidelines from the American College of Cardiology Foundation/American Heart Association (including the latest 2013 guideline for the management of ST-elevation myocardial infarction), the National Institute for Clinical Excellence, the European Society of Cardiology, the Canadian Cardiovascular Society and the American College of Chest Physicians on the use of antiplatelet therapies in myocardial infarction and percutaneous coronary intervention. The randomized trials discussed in the guidelines were retrieved for further review. As well, we searched for subsequent articles in this area by key authors and groups involved in the publication of the landmark trials.
What is the current standard for antiplatelet therapy after acute coronary syndromes?
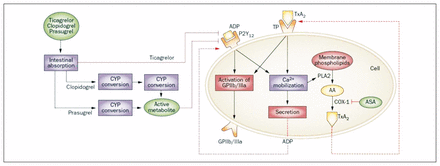
The use of clopidogrel in combination with ASA is the current standard for dual antiplatelet therapy after acute coronary syndromes.3 Although ASA inhibits platelet activation through suppression of the cyclooxygenase enzyme, platelet activation can occur through other pathways (Figure 1). Thus, dual antiplatelet therapy is used to suppress platelet activation further in acute coronary syndromes.
Inhibition of platelet activation by P2Y12 receptor antagonists. P2Y12 receptor antagonists bind irreversibly (clopidogrel and prasugrel) or reversibly (ticagrelor) to the P2Y12 receptor, thereby inhibiting calcium ion (Ca2+) mobilization and activation of the glycoprotein IIb/IIIa receptor (GPIIb/IIIa). Decreased Ca2+ mobilization also results in a reduction in secretion of vasoactive and proaggregatory substances from platelets and prevents conformational changes of the GPIIb/IIIa receptor that is required for platelet aggregation. Acetylsalicylic acid (ASA) binds irreversibily to cyclooxygenase-1 (COX-1), thus blocking the synthesis of thromboxane A2 (TxA2). Clopidogrel and prasugrel (thienopyridines) are prodrugs that require bioactivation by hepatic cytochrome P450 (CYP) enzymes. Ticagrelor (cyclopentyl-triazolo-pyrimidine) is an active drug molecule that directly and reversibly inhibits the P2Y12 receptor. [Reproduced, with permission, from Paikin JS, Eikelboom JW, Cairns JA, et al. New antithrombotic agents — insights from clinical trials. Nat Rev Cardiol 2010;7:498–509. Copyright © 2010 Macmillan Publishers Ltd.]
The CURE (Clopidogrel in Unstable Angina to Prevent Recurrent Events) and COMMIT (Clopidogrel and Metoprolol in Myocardial Infarction Trial) randomized trials showed that dual antiplatelet therapy with ASA and clopidogrel was effective in reducing recurrent myocardial infarction and cardiovascular events over ASA alone in patients with medically managed acute coronary syndromes.4,5 The PCI-CURE study, a subgroup analysis of the CURE trial, and the CREDO (Clopidogrel for the Reduction of Events During Observation) trial further established the role of dual antiplatelet therapy with ASA and clopidogrel after coronary artery stenting.6,7
The recommended duration of dual antiplatelet therapy after acute coronary syndromes is 1 year, with or without coronary stent insertion; thereafter, ASA should be continued indefinitely.8,9
The rates of recurrent myocardial infarction and stent thrombosis remain high despite dual antiplatelet therapy. Clopidogrel is a thienopyridine prodrug, requiring multistep metabolic bioactivation to its active molecule to exert its antiplatelet effects. Genetic polymorphisms and drug–drug interference of the enzymes that metabolize clopidogrel can affect platelet inhibition in some patients.10,11 Suboptimal platelet inhibition can subsequently lead to recurrent myocardial infarction and stent thrombosis, which has high mortality and morbidity.12 The CURRENT–OASIS 7 (Clopidogrel and Aspirin Optimal Dose Usage to Reduce Recurrent Events-Seventh Organization to Assess Strategies in Ischemic Syndromes) trial examined whether doubling the clopidogrel dose (150 mg/d) would maximize platelet inhibition; however, the benefits were modest compared with the standard dose of 75 mg/d.13
The pursuit of more potent antiplatelet agents after acute coronary syndromes and after coronary artery stenting, to maximize platelet inhibition while minimizing the risk of bleeding, led to the development of new antiplatelet agents.
How do the new antiplatelet agents differ from clopidogrel?
Prasugrel is a novel thienopyridine prodrug similar to clopidogrel that exerts its antiplatelet effects by binding to the P2Y12 receptor for adenosine diphosphate (ADP), thereby inhibiting platelet aggregation (Figure 1). Unlike clopidogrel, which requires 2 steps for conversion to its active metabolite, prasugrel requires only a single step (Figure 1, Table 1). This single-step bioactivation provides faster antiplatelet activity with less varied effects between patients compared with clopidogrel.14 As well, prasugrel is a more potent thienopyridine than clopidogrel and thus achieves greater platelet suppression.15 Similar to clopidogrel, its platelet inhibition is irreversible: 5–7 days are required for platelet function to return to normal after use is stopped.
Characteristics of clopidogrel and the new antiplatelet agents prasugrel and ticagrelor 15
Ticagrelor is a novel cyclopentyl-triazolo-pyrimidine agent. It is a reversible, direct inhibitor of the P2Y12 receptor that blocks ADP-mediated platelet activation and aggregation by binding to the receptor at a site different from the ADP-binding site (noncompetitive antagonism). Because ticagrelor is not a prodrug, it does not require metabolic bioactivation and is less influenced by variations in patients’ metabolic activity, genetic polymorphisms of enzymes and drug–drug interactions.16 In addition to providing faster and more consistent platelet inhibition than clopidogrel, ticagrelor also achieves greater platelet inhibition.17 The antiplatelet effect of ticagrelor is reversible, with platelet function returning to normal 2–5 days after its use is stopped. Ticagrelor has been shown to provide greater platelet inhibition than prasugrel.18
The increased platelet inhibition by the new antiplatelet agents is thought to provide superior clinical benefit over clopidogrel. However, the clinical relevance of greater platelet inhibition as measured by laboratory outcomes has recently been called into question and is a point of continued debate.19,20
What is the evidence for the new antiplatelet agents in acute coronary syndromes?
Prasugrel was studied in the randomized double-blind TRITON–TIMI 38 trial (Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel–Thrombolysis in Myocardial Infarction 38), in which 13 608 patients presenting with acute coronary syndromes were randomly assigned to receive either prasugrel (60-mg loading dose, 10 mg/d thereafter) or clopidogrel (300-mg loading dose, 75 mg/d thereafter) (Table 2).21 Most patients had a stent inserted after percutaneous coronary intervention, and all patients received ASA. At 15 months, the composite outcome of cardiovascular-related death, nonfatal myocardial infarction or nonfatal stroke was significantly reduced in favour of prasugrel (hazard ratio [HR] for prasugrel v. clopidogrel 0.81, 95% confidence interval [CI] 0.73–0.90). The difference was largely related to a significant reduction in myocardial infarction in the prasugrel group. Prasugrel use was also associated with lower rates of stent thrombosis (HR 0.48, 95% CI 0.36–0.64). However, patients in the prasugrel group had higher rates of major bleeding (HR 1.32, 95% CI 1.03–1.68) and life-threatening bleeding (HR 1.53, 95% CI 1.08–2.13) than those in the clopidogrel group. Thus, prasugrel was more effective than clopidogrel in reducing cardiovascular events in patients with acute coronary syndromes who had coronary stent insertion, but at an increased risk of major bleeding.
Trials of prasugrel and ticagrelor in acute coronary syndromes
Recently, the randomized double-blind TRILOGY ACS (Targeted Platelet Inhibition to Clarify the Optimal Strategy to Medically Manage Acute Coronary Syndromes) study investigated the benefit of prasugrel versus clopidogrel in 7243 patients with acute coronary syndromes who did not undergo percutaneous coronary intervention but who were medically managed.22 All patients received ASA 100 mg/d and optimal medical therapy for acute coronary syndromes. The use of prasugrel did not show a benefit in the reduction of the composite outcome (death from cardiovascular causes, nonfatal myocardial infarction or nonfatal stroke) when compared with clopidogrel. The rates of major bleeding did not differ between the 2 groups. Thus, in this study, prasugrel showed no advantage or disadvantage over clopidogrel in patients with medically managed acute coronary syndromes.
Ticagrelor was studied in the randomized double-blind PLATO trial (Study of Platelet Inhibition and Patient Outcomes), which enrolled 18 624 patients presenting with acute coronary syndromes.23 A substantial proportion of the patients underwent percutaneous coronary intervention (61% with and 4% without coronary stent insertion); the remainder underwent coronary artery bypass grafting (CABG, 10%) or medical management (25%). Ticagrelor (180-mg loading dose, 90 mg twice daily thereafter) was compared with clopidogrel (300–600-mg loading dose, 75 mg/d thereafter). All of the patients received ASA. At 12 months, the composite outcome of vascular-related death, myocardial infarction or stroke occurred significantly less often in the ticagrelor group than in the clopidogrel group (9.8% v. 11.7%; HR 0.84, 95% CI 0.77–0.92). The difference was driven by a reduction in myocardial infarction and all-cause death. Rates of stent thrombosis were significantly lower in the ticagrelor group than in the clopidogrel group (HR 0.77, 95% CI 0.62–0.95). The incidence of major bleeding did not differ between the study groups. Thus, ticagrelor was more effective than clopidogrel in patients with acute coronary syndromes, without an increased risk in major bleeding.
Overall, the evidence from these 3 large randomized trials shows that the newer thienopyridine agents are superior to clopidogrel in patients with acute coronary syndromes, particularly those undergoing percutaneous coronary intervention and stent insertion, in reducing recurrent cardiovascular events and stent thrombosis.
The European Society of Cardiology 2012 guidelines for ST-segment elevation myocardial infarction, the American College of Cardiology Foundation/American Heart Association 2011 guidelines for unstable angina and non–ST-segment elevation myocardial infarction and the Canadian 2012 guidelines on antiplatelet therapy strongly recommend the use of prasugrel or ticagrelor over clopidogrel in addition to ASA for all patients with acute coronary syndromes undergoing percutaneous coronary intervention with coronary stent insertion.8,24,25
For patients whose condition is medically managed and those undergoing percutaneous coronary intervention without a coronary stent, the guidelines strongly recommend the use of dual antiplatelet therapy with ASA and either clopidogrel or ticagrelor.8,24,25 Prasugrel is an option for dual antiplatelet therapy with ASA, but it does not offer an advantage over clopidogrel and its use in this situation is not encouraged by the guidelines.8,24,25
Current guidelines recommend that these new antiplatelet agents be considered in patients who experience coronary stent thrombosis while taking dual antiplatelet therapy with ASA and clopidogrel.25 However, this recommendation is largely based on expert opinion, consensus guidelines and extrapolation from the recent antiplatelet trials.
The indefinite use of low-dose ASA (81 mg/d) is recommended as part of dual antiplatelet therapy, based on the recommendations for secondary prevention and the evidence from the CURRENT–OASIS 7 trial, which showed no difference in cardiovascular-related outcomes between patients receiving a high dose of ASA (325 mg/d) and those given a low dose (75–100 mg/d) after coronary stent insertion.9,13,25
What are the harms of the new antiplatelet agents?
The risk of major bleeding is increased with the new antiplatelet agents, specifically prasugrel. Ticagrelor has a higher rate of non–CABG-related major bleeding, whereas prasugrel has higher rates of major and life-threatening bleeding compared with clopidogrel. Prasugrel should be avoided in patients at high risk of bleeding, but if necessary, a lower dose should be considered.
Dual therapy with ASA and clopidogrel increases the risk of major bleeding by an absolute of 1% compared with ASA alone.26 In the TRITON–TIMI 38 trial, ASA and prasugrel significantly increased the risk of non–CABG-related major bleeding (HR 1.32, 95% CI 1.03–1.68) and life-threatening bleeding (HR 1.53, 95% CI 1.08–2.13) after coronary stent insertion compared with ASA and clopidogrel.21 In a post hoc analysis of the TRITON–TIMI 38 trial, patients at highest risk of major bleeding with prasugrel were those with low body weight (≤ 60 kg), age greater than 75 years or a history of stroke. Thus, use of prasugrel should be avoided in such patients. If prasugrel use is necessary, Canadian and US guidelines recommend that a lower maintenance dose of 5 mg/d be used in such patients to minimize the risk of bleeding.24,25 This recommendation is largely based on pharmacokinetic studies and has not been clinically validated.
The combination of ASA and ticagrelor has been shown to have similar rates of overall major bleeding compared with ASA and clopidogrel after coronary stent insertion (HR 1.04, 95% CI 0.95–1.13), but it is associated with a higher rate of non–CABG-related bleeding (HR 1.19, 95% 1.02–1.38).23 Dypsnea (HR 1.84, 95% CI 1.68–2.02) and ventricular pauses longer than 3 seconds (5.8% in the ticagrelor group v. 3.6% in the clopidogrel group, p = 0.01) were more evident with the use of ticagrelor than with clopidogrel.23
How are these new agents used with oral anticoagulants?
The risk of bleeding is particularly high when “triple therapy” — dual antiplatelet therapy after coronary stent insertion plus oral anticoagulant treatment (e.g., with warfarin) for atrial fibrillation or mechanical heart valves — is required.25,27 The risk of thrombosis and reinfarction must be weighed against the risk of bleeding when considering triple therapy.
Several strategies can be used to minimize the risk of bleeding in patients who require triple therapy.9 The dose of ASA should be lowered to 81 mg/d, and the minimum duration of dual antiplatelet therapy with thienopyridine agents should be used after acute coronary syndromes. Guidelines recommend at least 1 month of dual antiplatelet therapy after insertion of a bare-metal coronary stent and at least 6 months after insertion of a drug-eluting stent.8,25 The US 2013 guidelines for ST-segment elevation myocardial infarction also recommend targeting the lower range of the therapeutic international normalized ratio in patients taking anticoagulant therapy, to minimize the risk of bleeding.9 Guidelines recommend that prophylactic gastroprotective therapy with proton pump inhibitors be started in patients given triple therapy who are at high risk of bleeding.28
The recently published WOEST (What is the Optimal Antiplatelet and Anticoagulant Therapy in Patients with Oral Anticoagulation and Coronary Stenting) study assessed the rates of bleeding associated with antithrombotic therapy among patients taking oral anticoagulant therapy who underwent percutaneous coronary intervention with coronary stent insertion.29 This open-label, multicentre randomized controlled study randomly assigned 573 patients to receive clopidogrel alone with oral anticoagulation (double therapy) or clopidogrel plus ASA plus oral anticoagulation (triple therapy). At 1 year, the rates of any bleeding event were significantly less with double therapy than with triple therapy (19.4% v. 44.4%, p < 0.0001); the rates of thrombotic and thromboembolic events did not differ between the 2 groups. Thus, use of clopidogrel alone in patients taking oral anticoagulation is an option for those at high risk of bleeding. Because prasugrel and ticagrelor have not been as well studied as part of triple therapy, it is unclear whether the rates of bleeding with such therapy are greater than those with clopidogrel and oral anticoagulation.
What are other considerations in the use of these new agents?
Because coronary stent insertion has become commonplace, the use of potent antiplatelet agents will become more prominent in efforts to reduce recurrent ischemic events. However, the ubiquitious use of these new agents over clopidogrel is hindered by their cost. The daily cost of prasugrel and ticagrelor ($2.60 and $3.16 respectively) is 3–4 times higher than that of generic clopidogrel ($0.71).30 The higher costs of prasugrel and ticagrelor are a greater financial burden to patients who require dual antiplatelet therapy for up to 1 year. As well, limited or restricted coverage for prasugrel and ticagrelor by provincial drug programs may shift the financial burden to the patient and potentially lead to non-compliance with antiplatelet therapy.
However, these new antiplatelet agents have been found to be more cost-effective than clopidogrel to the health care system. In a pharmacoeconomic analysis of data from the TRITON–TIMI 38 study, use of prasugrel reduced the total costs of hospital readmission because of a reduced rate of readmission for repeat percutaneous coronary intervention.31 As well, prasugrel was associated with life-expectancy gains of 0.102 years as a consequence of the decreased rate of nonfatal myocardial infarction. These results translated to a total cost of care (drug and readmission costs) that was $221 lower per patient with prasugrel than with clopidogrel. The incremental cost-effectiveness ratio for prasugrel was $9727 per life-year gained, which is below the commonly accepted threshold of $50 000 per life-year gained to be deemed a cost-effective medical intervention.27
Ticagrelor is also an economically attractive treatment strategy. Analysis of data from the PLATO study showed that non–drug-related health care costs were $665 lower per patient with ticagrelor than with clopidogrel, mainly because of the reduced number of readmission days and interventions.32 The incremental cost-effectiveness ratio for ticagrelor was $3193 per life-year gained, largely driven by the drug’s mortality benefit. Thus, although the drug costs of prasugrel and ticagrelor are higher than that of clopidogrel, their use may be economically attractive in reducing hospital costs and overall health care costs.
Gaps in knowledge
The evidence supporting prasugrel and ticagrelor is limited to the use of these agents in acute coronary syndromes. Their use in other cardiovascular disease states is currently being investigated. The PEGASUS trial (Prevention with Ticagrelor of Secondary Thrombotic Events in High-risk Patients with Prior Acute Coronary Syndrome) is currently investigating the role of ticagrelor versus placebo in patients 1–3 years after acute coronary syndromes. The EUCLID trial (Examining Use of Ticagrelor in Peripheral Arterial Disease) is evaluating the role of ticagrelor versus clopidogrel in patients with symptomatic peripheral arterial disease. Prasugrel is being studied in patients with acute coronary syndromes undergoing percutaneous coronary intervention in the ACCOAST study (A Comparison of Prasugrel at the Time of Percutaneous Coronary Intervention or as Pre-treatment at the Time of Diagnosis in Patients with Non-ST-Elevation Myocardial Infarction).
Other antiplatelet agents such as cangrelor (reversible, intravenous ADP P2Y12 inhibitor) and vorapaxar (protease-activated receptor 1 [PAR-1] antagonist) are being studied and are the next advancement in novel antiplatelet therapy.33,34 Cangrelor and vorapaxar are not currently available in North America, and their exact role in cardiovascular disease remains to be elucidated.
Conclusion
Guidelines recommend use of the new antiplatelet agents prasugrel and ticagrelor as part of dual antiplatelet therapy with ASA in acute coronary syndromes, setting a new standard. However, the benefits of decreased ischemic events and stent thrombosis with the use of these agents must be considered against the risk of increased bleeding and financial costs to the patient.
Resources for clinicians
2013 ACCF/AHA (American College of Cardiology Foundation/American Heart Association) guidelines for the management of ST-segment elevation myocardial infarction: http://circ.ahajournals.org/content/127/4/e362
2012 European Society of Cardiology guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: www.escardio.org/guidelines-surveys/esc-guidelines/GuidelinesDocuments/AMI-STEMI.aspx
Focused 2012 update of the Canadian Cardiovascular Society guidelines for the use of antiplatelet therapy: www.onlinecjc.ca/article/S0828-282X(13)00443-1/fulltext
American College of Chest Physicians evidence-based clinical practice guidelines on antithrombotic therapy and prevention of thrombosis, 9th edition: http://journal.publications.chestnet.org/issue.aspx?journalid=99&issueid=23443#12096ArticleTypeName
2013 National Institute for Health and Care Excellence (NICE) clinical guidelines for the acute management of myocardial infarction with ST-segment elevation: http://publications.nice.org.uk/myocardial-infarction-with-st-segment-elevation-cg167
2012 ACCF/AHA focused update of the guideline for the management of patients with unstable angina/non-ST-elevation myocardial infarction (updating the 2007 guideline and replacing the 2011 focused update): http://circ.ahajournals.org/content/126/7/875.full
Dual antiplatelet therapy, typically with ASA and clopidogrel, is recommended after acute coronary syndromes and after percutaneous coronary intervention with coronary stent insertion.
Prasugrel and ticagrelor, new P2Y12 antagonists, are more potent and exhibit less pharmacodynamic variability than clopidogrel.
These new agents are more effective than clopidogrel as part of dual antiplatelet therapy with ASA in reducing recurrent myocardial infarction and cardiovascular events after acute coronary syndromes and coronary stent insertion.
Prasugrel is associated with higher rates of major bleeding compared with clopidogrel and should not be used in patients at high risk of bleeding.
Evidence for the use of new antiplatelet agents in disease states other than acute coronary syndromes is currently limited.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Doson Chua wrote and revised the manuscript. Cesilia Nishi reviewed the manuscript for accuracy and clarity. Both authors approved the final version submitted for publication.