Abstract
Background: Although insulin analogues are commonly prescribed for the management of diabetes mellitus, there is uncertainty regarding their optimal use. We conducted meta-analyses to compare the outcomes of insulin analogues with conventional insulins in the treatment of type 1, type 2 and gestational diabetes.
Methods: We updated 2 earlier systematic reviews of the efficacy and safety of rapid-and long-acting insulin analogues. We searched electronic databases, conference proceedings and “grey literature” up to April 2007 to identify randomized controlled trials that compared insulin analogues with conventional insulins. Study populations of interest were people with type 1 and type 2 diabetes (adult and pediatric) and women with gestational diabetes.
Results: We included 68 randomized controlled trials in the analysis of rapid-acting insulin analogues and 49 in the analysis of long-acting insulin analogues. Most of the studies were of short to medium duration and of low quality. In terms of hemoglobin A1c, we found minimal differences between rapid-acting insulin analogues and regular human insulin in adults with type 1 diabetes (weighted mean difference for insulin lispro: –0.09%, 95% confidence interval [CI] –0.16% to –0.02%; for insulin aspart: –0.13%, 95% CI –0.20% to –0.07%). We observed similar outcomes among patients with type 2 diabetes (weighted mean difference for insulin lispro: –0.03%, 95% CI –0.12% to –0.06%; for insulin aspart: –0.09%, 95% CI –0.21% to 0.04%). Differences between long-acting insulin analogues and neutral protamine Hagedorn insulin in terms of hemoglobin A1c were marginal among adults with type 1 diabetes (weighted mean difference for insulin glargine: –0.11%, 95% CI –0.21% to –0.02%; for insulin detemir: –0.06%, 95% CI –0.13% to 0.02%) and among adults with type 2 diabetes (weighted mean difference for insulin glargine: –0.05%, 95% CI –0.13% to 0.04%; for insulin detemir: 0.13%, 95% CI 0.03% to 0.22%). Benefits in terms of reduced hypoglycemia were inconsistent. There were insufficient data to determine whether insulin analogues are better than conventional insulins in reducing long-term diabetes-related complications or death.
Interpretation: Rapid-and long-acting insulin analogues offer little benefit relative to conventional insulins in terms of glycemic control or reduced hypoglycemia. Long-term, high-quality studies are needed to determine whether insulin analogues reduce the risk of long-term complications of diabetes.
Diabetes mellitus is associated with serious long-term complications and premature death.1 Data from the Health Canada National Diabetes Surveillance System indicate that, in 2004/05, diabetes was diagnosed in about 5.5% (1.8 million) of Canadians aged 20 years and older.2 Because the disease goes undetected in many cases, the true prevalence may approach 1.9 million.3
Tight glycemic control, to maintain a hemoglobin A1c concentration of 7.0% or less, is recommended for all patients with diabetes to reduce the risk of long-term complications such as cardiovascular-related death, retinopathy and nephropathy.4 Insulin is indicated for all patients with type 1 diabetes and for patients with type 2 diabetes if adequate glycemic control cannot be achieved through exercise, diet or oral antidiabetic therapy.4
Conventional insulins include regular human insulin and intermediate-acting neutral protamine Hagedorn insulin. However, these agents do not replicate the pattern of basal and postprandial endogenous secretion of insulin. Insulin analogues are modified human insulins developed to address this limitation.5 The rapid-acting insulin analogues insulin lispro, insulin aspart and insulin glulisine are marketed in Canada as bolus insulins; the long-acting agents insulin glargine and insulin detemir are marketed as basal insulins.6
Systematic reviews of the insulin analogues have been published previously.7–10 However, through our comprehensive search of the literature, we did not identify any reviews of long-acting insulin analogues in the management of type 1 diabetes or gestational diabetes. In this article, we provide an up-to-date, comprehensive systematic review and meta-analysis of outcomes associated with the use of rapid-and long-acting insulin analogues in type 1 and type 2 diabetes (adult and pediatric patients) and gestational diabetes. Detailed methods and complete results are reported elsewhere.11,12
Methods
We based our current study on 2 health technology assessments of the insulin analogues from the Canadian Agency for Drugs and Technologies in Health (CADTH).13,14 Such reports from the agency consist of a systematic review of the available clinical and economic evidence regarding specific drugs or health technologies. We updated the 2 reports to include recently published studies, additional outcomes of interest, and intraclass comparisons of the rapid-and long-acting insulin analogues.
Literature search
We updated the original search strategy used for the health technology assessments to include studies published up to April 2007 (Appendix 1, available at www.cmaj.ca/cgi/content/full/180/4/385/DC2). We developed supplemental searches to include studies that addressed additional comparisons and outcomes of interest (Appendices 2 and 3, available at www.cmaj.ca/cgi/content/full/180/4/385/DC2).
We searched the following databases: MEDLINE (1966 to April 2007), MEDLINE In-Process and Other Non-Indexed Citations, MEDLINE Daily Update, EMBASE (1980 to April 2007), BIOSIS Previews (1989 to April 2007) and the Cochrane Library (Issue 3, 2007). We constructed the search terms using controlled vocabulary, such as the National Library of Medicine's MeSH (Medical Subject Headings), and key words.
The main search concepts were diabetes, long-acting insulin analogues and rapid-acting insulin analogues. We limited our search to randomized controlled trials. We identified “grey literature” by searching the websites of agencies that conduct health technology assessments and other related agencies, as well as endocrine and diabetes associations and their associated conference sites (Appendix 4, available at www.cmaj.ca/cgi/content/full/180/4/385/DC2). Stakeholders, including manufacturers of the agents under review, were given an opportunity to provide additional evidence.
Outcomes of interest
In this article, we present results for hemoglobin A1c concentration, hypoglycemia, quality-of-life, patient satisfaction, complications of diabetes (including death) and adverse effects. We analyzed data on hypoglycemia using the relative risk of experiencing 1 or more episodes of hypoglycemia during the study period; we used the rate ratio for the frequency of episodes (i.e., number of episodes per patient per unit time).15
Results for other outcomes of interest (i.e., fasting plasma glucose level, 2-hour postprandial glucose level, body weight, cholesterol level and blood pressure) are presented elsewhere.11,12
Selection criteria
We included randomized controlled trials published in English if they reported data for one of the following comparisons in patients with type 1, type 2 or gestational diabetes: rapid-acting insulin analogue versus regular human insulin; one rapid-acting insulin analogue versus another; premixed (i.e., biphasic) rapid-acting insulin analogue versus another premixed insulin (either rapid-acting insulin analogue or human insulin); long-acting insulin analogue versus neutral protamine Hagedorn insulin or another intermediate-acting conventional insulin; or one long-acting insulin analogue versus another. We excluded studies of insulin glulisine from the systematic review because this agent had not been marketed in Canada at the time of our analysis.
Quality assessment
Using a modified Jadad scale,16 2 reviewers (F.A. and A.L.) independently assessed the methodologic quality of the included studies of rapid-acting insulin analogues; 2 others (S.R.S and C.Y.) assessed the included studies of long-acting analogues. Specifically, they evaluated the extent of allocation concealment, blinding of assessors and reporting of intention-to-treat analysis.17 Disagreements were resolved by consensus or a third reviewer.
Data extraction and analysis
Each of the reviewers independently extracted data from the articles included in the analysis using a predesigned form. Disagreements were resolved in the same manner as for the quality assessment. Data extraction at the study level was not repeated for studies contained in the 2 original health technology assessments from the Canadian Agency for Drugs and Technologies in Health.13,14
We combined data using a random-effects model.15 We conducted separate analyses for gestational diabetes, pediatric type 1 diabetes, adult type 1 diabetes, pediatric type 2 diabetes and adult type 2 diabetes. We performed subgroup analyses for (a) administration method of rapid-acting insulin analogues (i.e., multiple daily injections or continuous subcutaneous insulin infusion); (b) type of bolus insulin (i.e., regular human insulin or rapid-acting insulin analogue) administered with long-acting analogues; and (c) type of oral antidiabetic agent administered with long-acting insulin analogues in the management of type 2 diabetes. We included the results from studies of premixed insulins in the same meta-analyses with results from studies of bolus insulins. In the absence of reported carryover effects, we combined data from crossover and parallel trials in the same meta-analysis.
We conducted sensitivity analyses to determine whether inclusion of studies deemed to be of low methodologic quality affected the results. We assessed the potential for publication bias, in meta-analyses that included more than 5 studies, using funnel plots.15
We determined heterogeneity using the I2 statistic, which describes the proportion of unexplained variability in effect estimates across studies in a meta-analysis.18 An I2 of 50% represents moderate heterogeneity.18 For analyses above this threshold, we explored possible causes of systematic variability through comparison of population, methodologic and treatment characteristics across included studies.
Results
Study selection
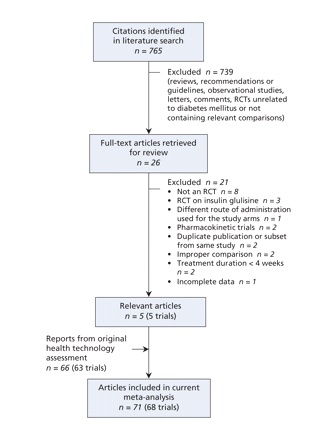
For rapid-acting insulin analogues, we identified 765 citations, of which we reviewed 26 and selected 5 trials19–23 for inclusion in our analysis. We thus had a total of 68 randomized controlled trials19–89 for the current meta-analysis, including 63 trials24–89 from the original health technology assessment13 (Figure 1).
Figure 1: Selection of randomized controlled trials (RCTs) of rapid-acting insulin analogues for inclusion in the meta-analysis.
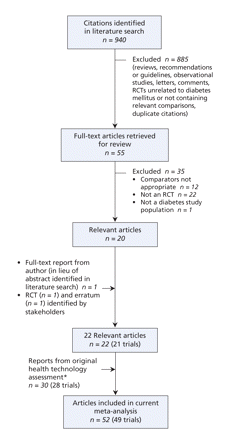
For long-acting insulin analogues, we identified 940 citations, of which we reviewed 55 and selected 20.90–109 A further trial110 was identified by stakeholders. We thus had a total of 49 randomized controlled trials for the analysis,90–102,104–142 including 28 trials111–140,143–146 from the original health technology assessment14 (Figure 2).
Figure 2: Selection of randomized controlled trials (RCTs) of long-acting insulin analogues for inclusion in the meta-analysis. *Thirty-four studies were included in the original health technology assessment;14 however, 4 abstracts143–146 were replaced by full-text publications identified during the update.
We identified no studies of insulin analogues in pediatric type 2 diabetes, or of long-acting insulin analogues in pregnant women with diabetes. Also, we found few studies evaluating the insulin analogues in specific ethnic groups, and none in First Nations populations.
Study characteristics and methodologic quality
Most of the trials included in the current meta-analysis were multinational and sponsored by industry. The number of patients in each study ranged from 7 to 1008. Of the 48 crossover studies,19,20,24,25,30,32,33,35,37–39,42–45,47,48,51,55,56,58,62–67,69–74,78,79,81–83,85,87,89,93,96,100–102,121 most lacked or did not mention a washout period. All studies were of open-label design. Trial duration ranged from 4 weeks to 30 months. Within each population and comparison, we found no major differences across trials in terms of patient characteristics (e.g., sex, degree of obesity, and severity or duration of diabetes).
The methodologic quality of most of the trials was rated as poor (Jadad score 2 or 3). No study was double-blinded, and allocation concealment was rarely described. Detailed ratings of study quality are reported in Appendices 5, 6 and 7 for rapid-acting insulin analogues and in Appendices 8 and 9 for long-acting insulin analogues (appendices are available at www.cmaj.ca/cgi/content/full/180/4/385/DC2).
Efficacy and safety in type 1 diabetes
Adults
Differences between treatments in terms of glycemic control in adults with type 1 diabetes are presented in Table 1 and Appendix 10 (available at www.cmaj.ca/cgi/content/full/180/4/385/DC2). Differences between treatments in terms of severe and nocturnal hypoglycemia in this patient group are presented in Table 2.
Table 1.
Table 2.
Compared with regular human insulin, use of insulin lispro resulted in a marginally lower hemoglobin A1c concentration (weighted mean difference –0.09%, 95% CI –0.16% to –0.02%), a lower risk of severe hypoglycemia (relative risk 0.80, 95% CI 0.67 to 0.96) and a lower rate of nocturnal hypoglycemia (rate ratio 0.51, 95% CI 0.42 to 0.62). There was a high degree of heterogeneity that was not explained by differences in patient characteristics or treatments across the studies that reported rates of nocturnal hypoglycemia in the overall analysis (I2 = 73.1%). For overall hypoglycemia, the rate was similar between the groups receiving insulin lispro and those receiving regular human insulin (data not shown). Subgroup analyses by method of administration did not reveal substantial differences in treatment effects between patients using multiple daily injections and those using continuous subcutaneous infusion (data not shown). In the group using continuous subcutaneous infusion, fewer of the patients given regular human insulin than of those given insulin lispro experienced severe hypoglycemia; however, the difference in risk between treatment groups was statistically nonsignificant.
For insulin aspart, the mean hemoglobin A1c concentration was slightly lower than the concentration with regular human insulin (weighted mean difference –0.13%, 95% CI –0.20% to –0.07%). There were no significant differences between treatments in the risk of severe hypoglycemia or the rate of overall hypoglycemia (data not shown). In the only study reporting data on nocturnal hypoglycemia, the rate among patients given insulin aspart through continuous subcutaneous infusion was significantly lower than the rate among those given regular human insulin (rate ratio 0.55, 95% CI 0.43 to 0.70).27 Subgroup analyses did not reveal important differences in treatment effects between patients using multiple daily injections and those using continuous subcutaneous infusion (data not shown).
Patients generally preferred rapid-acting insulin analogues over regular human insulin because of flexibility in dosing relative to mealtimes.25,39,42,73,83 Some studies that assessed quality of life and patient satisfaction reported statistically significant improvements with the use of rapid-acting insulin analogues compared with regular human insulin, whereas others found no differences between treatments (data not shown).25,29,39,42,57,62,65,67,73,79–83,89
The single study comparing insulin lispro with insulin aspart administered through continuous subcutaneous infusion reported nonsignificant differences in hemoglobin A1c (weighted mean difference 0.25%, 95% CI –0.20% to 0.71%) and rate of nocturnal hypoglycemia (rate ratio 1.20, 95% CI 0.89 to 1.68). However, the rate of overall hypoglycemia significantly favoured insulin aspart (rate ratio for insulin lispro v. insulin aspart 1.49, 95% CI 1.37 to 1.63).
Relative to neutral protamine Hagedorn insulin, insulin glargine provided a small but statistically significant improvement in hemoglobin A1c (weighted mean difference –0.11%, 95% CI –0.21% to –0.02%). There were no significant differences in the risk or rate of any type of hypoglycemia when the same bolus insulin was used in each treatment arm. The relative risk estimate for nocturnal hypoglycemia demonstrated a high degree of heterogeneity (I2 = 65.6%), which was substantially reduced when the study of shortest duration (4 weeks)112 was removed from the meta-analysis. This study demonstrated the largest risk reduction in favour of insulin glargine (relative risk 0.64, 95% CI 0.47 to 0.87).
In the pooled analysis of trials comparing insulin detemir and neutral protamine Hagedorn insulin, we found no significant difference in hemoglobin A1c (weighted mean difference –0.06%, 95% CI –0.13% to 0.02%). We found slight reductions in the risk of severe hypoglycemia (relative risk 0.74, 95% CI 0.58 to 0.96) and nocturnal hypoglycemia (relative risk 0.92, 95% CI 0.85 to 0.98) in favour of insulin detemir, but not overall hypoglycemia (data not shown). Also, we found statistically significant reductions in the rates of nocturnal and overall hypoglycemia in favour of insulin detemir (data not shown).
For insulin glargine and insulin detemir, each compared with neutral protamine Hagedorn insulin, we found that the effect estimates for hemoglobin A1c and hypoglycemia did not differ substantially according to the type of bolus insulin used (data not shown).
A single study reported that insulin glargine was not significantly different from neutral protamine Hagedorn insulin in terms of quality of life; however, it did show significantly greater patient satisfaction with insulin glargine (data not shown).137 No study reported data on quality of life or patient satisfaction with insulin detemir.
We found no significant difference in hemoglobin A1c between insulin detemir and insulin glargine in the single trial that compared the 2 agents (weighted mean difference –0.03%, 95% CI –0.26% to 0.20%). The risk of severe hypoglycemia (relative risk 0.25, 95% CI 0.07 to 0.86) and the rate ratios for severe and nocturnal hypoglycemia (data not shown) were statistically significant in favour of insulin detemir.
There were insufficient data available to compare insulin analogues and conventional insulins in terms of diabetic complications or death.
Children and adolescents
Differences between treatments in terms of glycemic control in children and adolescents with type 1 diabetes are presented in Table 1. Differences in the risk of hypoglycemia are presented in Table 2.
The only trial that compared insulin lispro with regular human insulin in adolescents with type 1 diabetes showed no significant difference in hemoglobin A1c (weighted mean difference –0.01%, 95% CI –0.21% to 0.19%) or risk of severe hypoglycemia (relative risk 1.00, 95% CI 0.29 to 3.43). The rate ratios for nocturnal hypoglycemia (rate ratio 0.61, 95% CI 0.57 to 0.64) and overall hypoglycemia (data not shown) significantly favoured insulin lispro.
In the pooled analysis of trials comparing insulin lispro and regular human insulin in preadolescent patients with type 1 diabetes, we found no significant difference in hemoglobin A1c (weighted mean difference 0.14%, 95% CI –0.18% to 0.46%), risk of severe hypoglycemia (relative risk 0.69, 95% CI 0.24 to 2.01) or rates of nocturnal hypoglycemia (rate ratio 0.96, 95% CI 0.74 to 1.26) and overall hypoglycemia (data not shown).
The only study that compared insulin aspart and regular human insulin in preadolescent patients with type 1 diabetes showed no significant difference in hemoglobin A1c or risk of overall hypoglycemia between treatment groups (data not shown).62 A second study that compared insulin aspart with regular human insulin and insulin lispro in 378 patients aged 6–18 years also reported no significant differences between treatments in terms of hemoglobin A1c or hypoglycemia (data not shown).26
We did not observe statistically significant differences between insulin glargine and conventional intermediate-acting insulins (mostly neutral protamine Hagedorn insulin) in children and adolescents with type 1 diabetes in terms of hemoglobin A1c (weighted mean difference –0.25%, 95% CI –0.55% to 0.05%) or any type of hypoglycemia. We observed a large degree of heterogeneity in the hemoglobin A1c estimate (I2 = 61.8%). This was due, at least in part, to the trial that reported the largest mean difference in hemoglobin A1c in favour of insulin glargine (–0.70%, 95% CI –1.12% to –0.28%).121 This study differed from the others in 2 ways: it involved Japanese patients as old as 21 years, and insulin aspart was used as the bolus insulin in both treatment arms.
The only trial that compared insulin detemir with neutral protamine Hagedorn insulin in children and adolescents with type 1 diabetes showed no significant differences between treatments in hemoglobin A1c (weighted mean difference 0.10%, 95% CI –0.10% to 0.30%) or severe hypoglycemia (relative risk 0.80, 95% CI 0.50 to 1.28). The relative risk of nocturnal hypoglycemia (0.85, 95% CI 0.77 to 0.94) and the rate ratios for nocturnal and overall hypoglycemia (data not shown) demonstrated small, statistically significant benefits in favour of insulin detemir.
No data on quality of life, patient satisfaction, diabetes-related complications or death were reported in any of the studies comparing insulin analogues with conventional insulins in children and adolescents. Also, we found no intraclass comparisons for either the rapid-acting or the long-acting insulin analogues.
Efficacy and safety in type 2 diabetes
Adults
Differences between treatments in terms of glycemic control in adults with type 2 diabetes are presented in Table 3 and Appendix 11 (available at www.cmaj.ca/cgi/content/full/180/4/385/DC2). Differences in terms of hypoglycemia are presented in Table 4. In the pooled analysis of trials comparing insulin lispro and regular human insulin, we observed no significant differences in hemoglobin A1c (weighted mean difference –0.03%, 95% CI –0.12% to 0.06%) or in the risk of severe hypoglycemia (relative risk 0.43, 95% CI 0.08 to 2.37), nocturnal hypoglycemia (relative risk 1.63, 95% CI 0.71 to 3.73) or overall hypoglycemia (data not shown). However, the rate ratio for nocturnal, but not severe, hypoglycemia, was statistically significant in favour of insulin lispro (data not shown).
Table 3.
Table 4.
The pooled analysis of trials comparing insulin aspart and regular human insulin showed no significant differences in hemoglobin A1c (weighted mean difference –0.09%, 95% CI –0.21% to 0.04%) or in the risk of any type of hypoglycemia between the treatment groups. The patients given insulin aspart had significantly fewer events of overall hypoglycemia than did those given regular human insulin (data not shown).
Two studies comparing insulin lispro with regular human insulin reported data on quality of life and patient satisfaction.39,41 They found no significant differences between treatment groups except that “worry related to diabetes” was significantly improved with insulin lispro in one of the studies (data not shown).41 None of the studies of insulin aspart in type 2 diabetes reported data on quality of life or patient satisfaction.
A single study comparing biphasic insulin lispro and biphasic insulin aspart reported no significant difference in hemoglobin A1c (mean difference 0.14%, 95% CI –0.02% to 0.30%) or overall hypoglycemia (data not shown) in adults with type 2 diabetes.
Combined therapy with oral antidiabetic agents was allowed in most trials that compared insulin glargine with neutral protamine Hagedorn insulin in adults with type 2 diabetes. Only one study compared insulin glargine with neutral protamine Hagedorn insulin in combination with a bolus insulin (i.e., without combined therapy with oral antidiabetic agents). Glycemic control was no better in the insulin glargine group regardless of the type of combined therapy (weighted mean difference in hemoglobin A1c –0.05%, 95% CI –0.13% to 0.04%, for insulin glargine with oral antidiabetic therapy; 0.28%, 95% CI 0.07% to 0.49%, for insulin glargine with bolus insulin).
There was no significant difference in the risk of severe hypoglycemia in the studies that used oral antidiabetic therapy (relative risk 0.66, 95% CI 0.29 to 1.48). However, the rate ratio was statistically significant in favour of insulin glargine (data not shown). Both the relative risk (I2 = 64%) and rate ratio (I2 = 83%) estimates demonstrated a high degree of heterogeneity that was due at least in part to opposite effects in the studies combining insulins with sulfonylureas,92,108,109,128 versus those combining insulins with various oral antidiabetic therapies.131,132,140 The risk and rate for severe hypoglycemia were significantly lower among patients given insulin glargine in the sulfonylurea subgroup, but not in the subgroup in which various oral antidiabetic therapies were used (data not shown).
The relative risk for nocturnal hypoglycemia significantly favoured insulin glargine in both the bolus insulin study (relative risk 0.78, 95% CI 0.62 to 0.98) and the studies that allowed oral antidiabetic therapy (relative risk 0.56, 95% CI 0.47 to 0.68). Rate ratio results were similar to those for relative risk in the studies allowing oral antidiabetic therapy, and not estimable in the bolus insulin study. There was a small, statistically significant reduction in risk of overall hypoglycemia in favour of insulin glargine in the studies allowing oral antidiabetic therapy but not in the bolus insulin study (data not shown).
Four studies compared insulin detemir with neutral protamine Hagedorn insulin in adults with type 2 diabetes. Three allowed the use of oral antidiabetic therapy, and 1 study used bolus insulin (insulin aspart) before meals. In the study that used bolus insulin, there was no significant difference between treatment groups in terms of hemoglobin A1c (weighted mean difference 0.10%, 95% CI –0.18% to 0.38%) or risk of overall hypoglycemia (data not shown). The risk of nocturnal hypoglycemia was lower in the insulin detemir group (relative risk 0.66, 95% CI 0.45 to 0.96). The pooled analysis of results from the studies that allowed oral antidiabetic therapy showed a small but statistically significant difference in hemoglobin A1c in favour of neutral protamine Hagedorn insulin (weighted mean difference 0.13%, 95% CI 0.03% to 0.22%). The relative risk for severe hypoglycemia was not statistically significant, although the relative risks for nocturnal hypoglycemia (relative risk 0.53, 95% CI 0.31 to 0.91) and overall hypoglycemia (data not shown) significantly favoured insulin detemir. All 3 relative risk estimates, obtained by pooling data across 2 studies,91,99 demonstrated a high degree of heterogeneity. This may have been due to the fact that one study administered insulin detemir and neutral protamine Hagedorn insulin once daily99 and the other study administered both agents twice daily.91 The study with doses given once daily reported larger reductions in risk of hypoglycemia in favour of insulin detemir than did the study with doses given twice daily. Rate ratios for all types of hypoglycemia were statistically significant in favour of insulin detemir (data not shown).
In terms of patient satisfaction with long-acting insulin analogue treatment, one study92 found a small yet statistically significant benefit in favour of insulin glargine over neutral protamine Hagedorn insulin (data not shown). No studies of long-acting insulin analogues reported data on quality of life.
Two studies compared insulin detemir with insulin glargine in patients with type 2 diabetes. One of the studies allowed the use of oral antidiabetic therapy; it showed no significant difference in terms of hemoglobin A1c (weighted mean difference 0.10%, 95% CI –0.06% to 0.26%) or nocturnal hypoglycemia. The other study used bolus insulin (insulin aspart); it reported a slightly higher hemoglobin A1c with insulin detemir (weighted mean difference 0.20%, 95% CI 0.10% to 0.30%). Neither study reported a difference in risk of overall hypoglycemia (data not shown).
There were insufficient data available for comparisons between insulin analogues and conventional insulins in terms of diabetes-related complications or death.
Efficacy and safety in pregnant women with diabetes
In the pooled analysis of results from studies comparing insulin lispro and regular human insulin in pregnant women, we observed no significant differences in hemoglobin A1c (weighted mean difference 0.20%, 95% CI –1.03% to 1.43%) or risk of severe hypoglycemia (relative risk 0.21, 95% CI 0.01 to 4.10) among women with type 1 diabetes.40,46,84 We also observed no significant difference in hemoglobin A1c among women with gestational diabetes (weighted mean difference 0.06%, 95% CI –0.11% to 0.23%).40,46,84
Results from a single trial comparing insulin aspart with regular human insulin in pregnant women with type 1 diabetes were similar to those for insulin lispro in terms of hemoglobin A1c (weighted mean difference –0.08%, 95% CI –0.28% to 0.12%), risk of severe hypoglycemia (relative risk 1.14, 95% CI 0.76 to 1.71) and risk of overall hypoglycemia (relative risk 1.04, 95% CI 0.98 to 1.11).22
We did not identify randomized controlled trials of long-acting insulin analogues in pregnant women.
Adverse events
Adverse events other than hypoglycemia that were reported in the included studies are presented for rapid-acting and long-acting insulin analogues in type 1 and type 2 diabetes (Appendices 12 to 15; available at www.cmaj.ca/cgi/content/full/180/4/385/DC2). The most commonly reported adverse events were infections of the upper respiratory tract, reactions at the injection site and weight gain. The incidence of adverse events was similar between insulin analogues and conventional insulins. Serious adverse events were uncommon.
Interpretation
Our results suggest that differences between conventional insulins and insulin analogues are minimal in the management of type 1, type 2 and gestational diabetes. Compared with the original health technology assessments,13,14 we included studies published up to April 2007. As well, we assessed more outcomes and conducted intraclass comparisons for both the rapid- and long-acting insulin analogues.
We found that most estimates of differences in hemoglobin A1c between treatment groups were not statistically significant. Where they were statistically significant in favour of insulin analogues, the differences were smaller than minimal clinically important differences described in the literature.10,147
We found statistically significant benefits of insulin analogues over conventional insulins in terms of hypoglycemia for some comparisons, populations and hypoglycemia types. However, we did not consistently observe a major clinical advantage in terms of hypoglycemia for either the rapid-acting or the long-acting insulin analogues over conventional insulins. In particular, no relative risk or rate ratio estimates for hypoglycemia were statistically significant for insulin glargine versus neutral protamine Hagedorn insulin in type 1 diabetes, for insulin aspart versus regular human insulin in pediatric type 1 diabetes or for rapid-acting insulin analogues versus regular human insulin in gestational diabetes. Furthermore, several trials excluded subjects with a history of recurrent major hypoglycemia; therefore, the benefits of insulin analogues in such patients remain uncertain. This was particularly the case for trials that compared insulin detemir with neutral protamine Hagedorn insulin.
Few of the studies reported on patient satisfaction with treatment or quality of life. This suggests that these outcomes are rarely measured or are selectively reported. When data for these outcomes were available, substantial heterogeneity in methods across studies precluded pooling of the results. Some studies reported insulin analogues to be statistically significantly superior to conventional insulins in terms of quality of life; however, results were inconsistent, and differences often appeared to be small and of uncertain clinical significance.
All of the head-to-head comparisons between insulin analogues of the same class showed little or no differences in glycemic control or risk of hypoglycemia.
Studies of insulin analogues were not sufficiently powered or of adequate duration to measure differences in long-term diabetes-related complications or death.
Our results regarding the effects of rapid-acting insulin analogues in type 1, type 2 and gestational diabetes and of long-acting insulin analogues in type 2 diabetes are similar to those reported by others.7–10 In a recent systematic review, biphasic insulin analogues were found to be similar to biphasic human insulin in terms of glycemic control and hypoglycemia rates among patients with type 2 diabetes.148 In our analysis, such trials were pooled with data from trials comparing rapid-acting insulin analogues and regular human insulin. However, subgroup analysis of trials of biphasic insulin did not yield substantially different results from the overall analysis (data not shown). Consistent with our findings, previous reviews observed that most trials of insulin analogues had methodologic limitations.7,10 For example, allocation concealment was adequate in only 10 of 117 trials included in our review (Appendices 5 to 9, available online at www.cmaj.ca/cgi/content/full/180/4/385/DC2). Thus, the potential for ascertainment bias is heightened,149 especially for subjective outcomes such as patient-reported hypoglycemia and quality of life.
Limitations
Like all systematic reviews, our analysis has limitations. First, we restricted our search to trials published in English; therefore, we may have missed articles published in other languages. However, empirical evidence suggests that exclusion of non-English trials has minimal impact on the results of systematic reviews and meta-analyses.150–152 Furthermore, additional trials published in languages other than English were not identified by stakeholders.
Second, there was heterogeneity across the trial results, as indicated by high I2 values. The degree of heterogeneity was particularly high for hypoglycemia outcomes. Although we could not always identify reasons for heterogeneity, we observed that studies reporting outlying estimates of effect differed in terms of patient characteristics or treatment strategies in some analyses. In most cases, results were qualitatively similar across studies in terms of direction of effect, even in the presence of large I2 values.
Third, we pooled data for hemoglobin A1c and hypoglycemia separately. Investigators instituting more aggressive glycemic control may have been less likely to find differences in hemoglobin A1c between treatment groups but more likely to observe benefits in hypoglycemia. By pooling studies without accounting for this correlation, we may have underestimated the benefit of the insulin analogues. However, the results of a recent metaregression analysis of insulin glargine using patient-level data suggest that adjustment for hemoglobin A1c does not greatly affect estimates of relative risk for hypoglycemia.153
The remaining limitations pertain to the available evidence on insulin analogues. More studies are needed to understand better the impact of insulin analogues on long-term diabetes-related complications, death, quality of life and patient satisfaction. Improvements in methodologic quality of trials are also necessary to produce valid assessments of the efficacy and safety of these agents. Furthermore, the relative safety and effectiveness of insulin analogues versus conventional insulins requires study in patients with a prior history of significant hypoglycemia, children with type 2 diabetes and, for the long-acting insulin analogues, pregnant women.
Conclusion
Our results indicate that insulin analogues offer few clinical advantages over conventional insulins in the management of most patients with type 1, type 2 or gestational diabetes. Although the evidence supporting the benefit of insulin analogues in terms of hypoglycemia is weak, these agents may be an option for patients with problematic hypoglycemia despite optimization of conventional insulin therapy. In a companion paper (see page 369 of this issue),154 we report on the cost-effectiveness of insulin analogues in the management of type 1 and type 2 diabetes in adults. The results of the cost-effectiveness analysis serve to clarify further the optimal place of insulin analogues relative to conventional insulins in the management of diabetes in the Canadian health care system.
@@ See related commentary by Siebenhofer-Kroitzsch and colleagues, page 369
Footnotes
-
Une version française de ce résumé est disponible à l'adresse www.cmaj.ca/cgi/content/full/180/4/385/DC1
Funding: This research was supported through a financial contribution from Health Canada to COMPUS.
This article has been peer reviewed.
Contributors: All of the authors contributed to the conception and design of the study. Sumeet Singh, Fida Ahmad, Avtar Lal, Changhua Yu and Zemin Bai extracted data from primary studies and analyzed and interpreted the results. Heather Bennett provided oversight for the extraction, analysis and interpretation of the data. Sumeet Singh, with the help of Avtar Lal, Changhua Yu and Heather Bennett, drafted the manuscript. All of the authors critically reviewed the manuscript. Zemin Bai ensured that the analysis was conducted appropriately and verified the accuracy of results presented in the manuscript. All of the authors approved the final version submitted for publication.
Acknowledgements: We thank Greg Bak and Michelle Fiander for developing and implementing the literature search strategies, and Samantha Verbrugghe for assistance with data management.
This systematic review was conducted by researchers at the Canadian Optimal Medication Prescribing and Utilization Service (COMPUS), a directorate of the Canadian Agency for Drugs and Technologies in Health (CADTH). COMPUS identifies optimal drug therapy, develops intervention tools and provides services to promote and encourage the use of evidence-based clinical and cost-effectiveness information in decision-making by health care providers and patients. COMPUS is a collaborative, pan-Canadian service funded by Health Canada. For more information, visit www.cadth.ca.
Competing interests: None declared.
REFERENCES
- 1.↵
- 2.↵
- 3.↵
- 4.↵
- 5.↵
- 6.↵
- 7.↵
- 8.
- 9.
- 10.↵
- 11.↵
- 12.↵
- 13.↵
- 14.↵
- 15.↵
- 16.↵
- 17.↵
- 18.↵
- 19.↵
- 20.↵
- 21.
- 22.↵
- 23.↵
- 24.↵
- 25.↵
- 26.↵
- 27.↵
- 28.
- 29.↵
- 30.↵
- 31.
- 32.↵
- 33.↵
- 34.
- 35.↵
- 36.
- 37.↵
- 38.
- 39.↵
- 40.↵
- 41.↵
- 42.↵
- 43.
- 44.
- 45.↵
- 46.↵
- 47.↵
- 48.↵
- 49.
- 50.
- 51.↵
- 52.
- 53.
- 54.
- 55.↵
- 56.↵
- 57.↵
- 58.↵
- 59.
- 60.
- 61.
- 62.↵
- 63.
- 64.
- 65.↵
- 66.
- 67.↵
- 68.
- 69.↵
- 70.
- 71.
- 72.
- 73.↵
- 74.↵
- 75.
- 76.
- 77.
- 78.↵
- 79.↵
- 80.
- 81.↵
- 82.
- 83.↵
- 84.↵
- 85.↵
- 86.
- 87.↵
- 88.
- 89.↵
- 90.↵
- 91.↵
- 92.↵
- 93.↵
- 94.
- 95.
- 96.↵
- 97.
- 98.
- 99.↵
- 100.↵
- 101.
- 102.↵
- 103.
- 104.↵
- 105.
- 106.
- 107.
- 108.↵
- 109.↵
- 110.↵
- 111.↵
- 112.↵
- 113.
- 114.
- 115.
- 116.
- 117.
- 118.
- 119.
- 120.
- 121.↵
- 122.
- 123.
- 124.
- 125.
- 126.
- 127.
- 128.↵
- 129.
- 130.
- 131.↵
- 132.↵
- 133.
- 134.
- 135.
- 136.
- 137.↵
- 138.
- 139.
- 140.↵
- 141.
- 142.↵
- 143.↵
- 144.
- 145.
- 146.↵
- 147.↵
- 148.↵
- 149.↵
- 150.↵
- 151.
- 152.↵
- 153.↵
- 154.↵