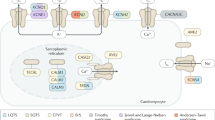
Abstract
Genetic alterations of various ion channels produce heritable cardiac arrhythmias that predispose affected individuals to sudden death. The investigation of such 'channelopathies' continues to yield remarkable insights into the molecular basis of cardiac excitability. The concept of channelopathies is not restricted to genetic disorders; notably, changes in the expression or post-translational modification of ion channels underlie the fatal arrhythmias associated with heart failure. Recognizing the fundamental defects in channelopathies provides the basis for new strategies of treatment, including tailored pharmacotherapy and gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Catterall, W. A. Molecular properties of sodium and calcium channels. J. Bioenerg. Biomembr. 28, 219–230 (1996).
Jan, L. Y. & Jan, Y. N. Voltage-gated and inwardly rectifying potassium channels. J. Physiol. 505, 267–282 (1997).
Philipson, K. D. & Nicoll, D. A. Sodium–calcium exchange: a molecular perspective. Annu. Rev. Physiol. 62, 111–133 (2000).
Marban, E., Yamagishi, T. & Tomaselli, G. F. Structure and function of voltage-gated sodium channels. J. Physiol. 508, 647–657 (1998).
Zeng, J. & Rudy, Y. Early afterdepolarizations in cardiac myocytes: mechanism and rate dependence. Biophys. J. 68, 949–964 (1995).
Robbins, J. KCNQ channels: physiology, pathophysiology, and pharmacology. Pharmacol. Ther. 90, 1–19 (2001).
Näbauer, M., Beuckelmann, D. J. & Erdmann, E. Characteristics of transient outward current in human ventricular myocytes from patients with terminal heart failure. Circ. Res. 73, 386–394 (1993).
Hoppe, U. C. et al. Manipulation of cellular excitability by cell fusion: effects of rapid introduction of transient outward K+ current on the guinea pig action potential. Circ. Res. 84, 964–972 (1999).
Papp, Z. et al. Two components of [Ca2+]i-activated Cl− current during large [Ca2+]i transients in single rabbit heart Purkinje cells. J. Physiol. 483, 319–330 (1995).
Koster, O. F., Szigeti, G. P. & Beuckelmann, D. J. Characterization of a [Ca2+]i-dependent current in human atrial and ventricular cardiomyocytes in the absence of Na+ and K+. Cardiovasc. Res. 41, 175–187 (1999).
Kääb, S. et al. Molecular basis of transient outward potassium current downregulation in human heart failure: a decrease in Kv4.3 mRNA correlates with a reduction in current density. Circulation 98, 1383–1393 (1998).
Näbauer, M. et al. Regional differences in current density and rate-dependent properties of the transient outward current in subepicardial and subendocardial myocytes of human left ventricle. Circulation 93, 168–177 (1996).
Hoppe, U. C., Marban, E. & Johns, D. C. Molecular dissection of cardiac repolarization by in vivo Kv4.3 gene transfer. J. Clin. Invest. 105, 1077–1084 (2000).
Winslow, R. L., Rice, J. & Jafri, S. Modeling the cellular basis of altered excitation–contraction coupling in heart failure. Prog. Biophys. Mol. Biol. 69, 497–514 (1998).
Keating, M. T. & Sanguinetti, M. C. Molecular and cellular mechanisms of cardiac arrhythmias. Cell 104, 569–580 (2001).
Yue, D. T. & Marban, E. A novel cardiac potassium channel that is active and conductive at depolarized potentials. Pflugers Arch. 413, 127–133 (1988).
Patel, A. J., Lazdunski, M. & Honore, E. Lipid and mechano-gated 2P domain K+ channels. Curr. Opin. Cell Biol. 13, 422–428 (2001).
Kim, D. et al. Cloning and functional expression of a novel cardiac two-pore background K+ channel (cTBAK-1). Circ. Res. 82, 513–518 (1998).
Luo, C. H. & Rudy, Y. A dynamic model of the cardiac ventricular action potential. I. Simulations of ionic currents and concentration changes. Circ. Res. 74, 1071–1096 (1994).
Weidmann, S. The electrical constants of Purkinje fibres. J. Physiol. 118, 348–360 (1952).
Nichols, C. G., Ripoll, C. & Lederer, W. J. ATP-sensitive potassium channel modulation of the guinea pig ventricular action potential and contraction. Circ. Res. 68, 280–287 (1991).
Marban, E., Robinson, S. W. & Wier, W. G. Mechanisms of arrhythmogenic delayed and early afterdepolarizations in ferret ventricular muscle. J. Clin. Invest. 78, 1185–1192 (1986).
Cranefield, P. F. & Aronson, R. S. Torsades de pointes and early afterdepolarizations. Cardiovasc. Drugs Ther. 5, 531–537 (1991).
Saffitz, J. E., Laing, J. G. & Yamada, K. A. Connexin expression and turnover: implications for cardiac excitability. Circ. Res. 86, 723–728 (2000).
Berenfeld, O. & Jalife, J. Purkinje-muscle reentry as a mechanism of polymorphic ventricular arrhythmias in a 3-dimensional model of the ventricles. Circ. Res. 82, 1063–1077 (1998).
Hoffman, B. F. & Rosen, M. R. Cellular mechanisms for cardiac arrhythmias. Circ. Res. 49, 1–15 (1981).
Samie, F. H. & Jalife, J. Mechanisms underlying ventricular tachycardia and its transition to ventricular fibrillation in the structurally normal heart. Cardiovasc. Res. 50, 242–250 (2001).
Camm, A. J. et al. Congenital and acquired long QT syndrome. Eur. Heart J. 21, 1232–1237 (2000).
Wang, Q. et al. SCN5A mutations associated with an inherited cardiac arrhythmia, long QT syndrome. Cell 80, 805–811 (1995).
Sanguinetti, M. C. Dysfunction of delayed rectifier potassium channels in an inherited cardiac arrhythmia. Ann. NY Acad. Sci. 868, 406–413 (1999).
Dumaine, R. et al. Multiple mechanisms of Na+ channel-linked long-QT syndrome. Circ. Res. 78, 916–924 (1996).
Hoppe, U. C., Marban, E. & Johns, D. C. Distinct gene-specific mechanisms of arrhythmia revealed by cardiac gene transfer of two long QT disease genes, HERG and KCNE1. Proc. Natl Acad. Sci. USA 98, 5335–5340 (2001).
Splawski, I. et al. Spectrum of mutations in long-QT syndrome genes. KVLQT1, HERG, SCN5A, KCNE1, and KCNE2. Circulation 102, 1178–1185 (2000).
Schwartz, P. J. et al. Genotype–phenotype correlation in the long-QT syndrome: gene-specific triggers for life-threatening arrhythmias. Circulation 103, 89–95 (2001).
Plaster, N. M. et al. Mutations in Kir2.1 cause the developmental and episodic electrical phenotypes of Andersen's syndrome. Cell 105, 511–519 (2001).
Chen, Q. et al. Genetic basis and molecular mechanism for idiopathic ventricular fibrillation. Nature 392, 293–296 (1998).
Antzelevitch, C. The Brugada syndrome: ionic basis and arrhythmia mechanisms. J. Cardiovasc. Electrophysiol. 12, 268–272 (2001).
Rook, M. B. et al. Human SCN5A gene mutations alter cardiac sodium channel kinetics and are associated with the Brugada syndrome. Cardiovasc. Res. 44, 507–517 (1999).
Dumaine, R. et al. Ionic mechanisms responsible for the electrocardiographic phenotype of the Brugada syndrome are temperature dependent. Circ. Res. 85, 803–809 (1999).
Wang, Y. & Rudy, Y. Action potential propagation in inhomogeneous cardiac tissue: safety factor considerations and ionic mechanism. Am. J. Physiol. Heart Circ. Physiol. 278, H1019–H1029 (2000).
Schott, J. J. et al. Cardiac conduction defects associate with mutations in SCN5A. Nature Genet. 23, 20–21 (1999).
Tan, H. L. et al. A sodium-channel mutation causes isolated cardiac conduction disease. Nature 409, 1043–1047 (2001).
Veldkamp, M. W. et al. Two distinct congenital arrhythmias evoked by a multidysfunctional Na+ channel. Circ. Res. 86, E91–E97 (2000).
Marban, E. Heart failure: the electrophysiologic connection. J. Cardiovasc. Electrophysiol. 10, 1425–1428 (1999).
Beuckelmann, D. J., Näbauer, M. & Erdmann, E. Alterations of K+ currents in isolated human ventricular myocytes from patients with terminal heart failure. Circ. Res. 73, 379–385 (1993).
Kääb, S. et al. Ionic mechanism of action potential prolongation in ventricular myocytes from dogs with pacing-induced heart failure. Circ. Res. 78, 262–273 (1996).
Berger, R. D. et al. Beat-to-beat QT interval variability: novel evidence for repolarization lability in ischemic and nonischemic dilated cardiomyopathy. Circulation 96, 1557–1565 (1997).
Studer, R. et al. Gene expression of the cardiac Na+–Ca2+ exchanger in end-stage human heart failure. Circ. Res. 75, 443–453 (1994).
Winslow, R. L. et al. Mechanisms of altered excitation–contraction coupling in canine tachycardia-induced heart failure, II: model studies. Circ. Res. 84, 571–586 (1999).
Sanguinetti, M. C. et al. A mechanistic link between an inherited and an acquired cardiac arrhythmia: HERG encodes the IKr potassium channel. Cell 81, 299–307 (1995).
Mitcheson, J. S. et al. A structural basis for drug-induced long QT syndrome. Proc. Natl Acad. Sci. USA 97, 12329–12333 (2000).
Liu, X. K. et al. Female gender is a risk factor for torsades de pointes in an in vitro animal model. J. Cardiovasc. Pharmacol. 34, 287–294 (1999).
Roden, D. M. & Spooner, P. M. Inherited long QT syndromes: a paradigm for understanding arrhythmogenesis. J. Cardiovasc. Electrophysiol. 10, 1664–1683 (1999).
Roden, D. M. Pharmacogenetics and drug-induced arrhythmias. Cardiovasc. Res. 50, 224–231 (2001).
Abbott, G. W. et al. MiRP1 forms IKr potassium channels with HERG and is associated with cardiac arrhythmia. Cell 97, 175–187 (1999).
Mazhari, R. et al. Molecular interactions between two long-QT syndrome gene products, HERG and KCNE2, rationalized by in vitro and in silico analysis. Circ. Res. 89, 33–38 (2001).
Priori, S. G. et al. Genetic and molecular basis of cardiac arrhythmias: impact on clinical management parts I and II. Circulation 99, 518–528 (1999).
Benhorin, J. et al. Effects of flecainide in patients with new SCN5A mutation: mutation- specific therapy for long-QT syndrome? Circulation 101, 1698–1706 (2000).
Windle, J. R. et al. Normalization of ventricular repolarization with flecainide in long QT syndrome patients with SCN5A:DeltaKPQ mutation. Ann. Noninvasive Electrocardiol. 6, 153–158 (2001).
Compton, S. J. et al. Genetically defined therapy of inherited long-QT syndrome. Correction of abnormal repolarization by potassium. Circulation 94, 1018–1022 (1996).
Shimizu, W. et al. Improvement of repolarization abnormalities by a K+ channel opener in the LQT1 form of congenital long-QT syndrome. Circulation 97, 1581–1588 (1998).
Nuss, H. B. et al. Reversal of potassium channel deficiency in cells from failing hearts by adenoviral gene transfer: a prototype for gene therapy for disorders of cardiac excitability and contractility. Gene Ther. 3, 900–912 (1996).
Nuss, H. B., Marban, E. & Johns, D. C. Overexpression of a human potassium channel suppresses cardiac hyperexcitability in rabbit ventricular myocytes. J. Clin. Invest. 103, 889–896 (1999).
Donahue, J. K. et al. Focal modification of electrical conduction in the heart by viral gene transfer. Nature Med. 6, 1395–1398 (2000).
Acknowledgements
Supported by NIH. E.M. is the Michel Mirowski, M.D. Professor of Cardiology of the Johns Hopkins University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marbán, E. Cardiac channelopathies. Nature 415, 213–218 (2002). https://doi.org/10.1038/415213a
Issue Date:
DOI: https://doi.org/10.1038/415213a
This article is cited by
-
hERG-deficient human embryonic stem cell-derived cardiomyocytes for modelling QT prolongation
Stem Cell Research & Therapy (2021)
-
Inhibitory effects of aloperine on voltage-gated Na+ channels in rat ventricular myocytes
Naunyn-Schmiedeberg's Archives of Pharmacology (2021)
-
Next-generation pacemakers: from small devices to biological pacemakers
Nature Reviews Cardiology (2018)
-
Cardiovascular pharmacology of K2P17.1 (TASK-4, TALK-2) two-pore-domain K+ channels
Naunyn-Schmiedeberg's Archives of Pharmacology (2018)
-
Unexplored therapeutic opportunities in the human genome
Nature Reviews Drug Discovery (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.