Abstract
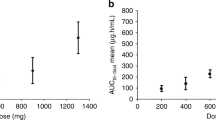
Purpose: MG98 is a second generation phosphorothioate antisense oligodeoxynucleotide which is a highly specific inhibitor of translation of the mRNA for human DNA MeTase I (DNMT 1). This phase I study examined the toxicity and pharmacologic profile of MG98 administered as a continuous 21-day intravenous infusion every 4 weeks. Patients and methods: Fourteen patients with solid cancers received a total of 25 cycles of MG98 at doses ranging from 40 to 240 mg/m2/day. Steady-state concentrations of MG98 were measured as were several pharmacodynamic assessments including mRNA of the target gene, DNMT1, in PBMC. In addition, other potential surrogate markers of drug effects were explored, including hemoglobin F, Vimentin and GADD45. Results: Dose limiting effects were drug-related reversible transaminase elevation and fatigue seen at doses of 240, 200 and 160 mg/m2/day. The dose level of 80 mg/m2/day was felt to be safe and tolerable when delivered on this schedule. No evidence of antitumor activity was observed. Although pharmacokinetic analysis revealed that at the higher dose levels, mean Css values of MG98 were approximately 10-fold times the IC50 values associated with target inhibition in vitro, the extent of MG98 penetration into target tumors in this trial was not determined. No consistent, dose-related changes in correlative markers including DNMT1 mRNA, hemoglobin F, Vimentin and GADD45, were observed. Conclusions: This schedule of MG98 given as a 21-day continuous intravenous infusion every 4 weeks was poorly tolerated in the highest doses; therefore, further disease-site specific evaluation of the efficacy of this agent will utilize a more favorable, intermittent dosing schedule. Pharmacodynamic evaluations undertaken in an attempt to explore and validate the biological mechanisms of MG98 did not show dose-related effects.
Similar content being viewed by others
References
Bestor TH, Laudano A, Mattaliano R, Ingram V: Cloning and sequencing of a cDNA encoding DNA methyltransferase of mouse cells. The carboxyl-terminal domain of the mammalian enzymes is related to bacterial restriction methyltransferases. J Mol Biol 203: 971–983, 1988
Jones PA, Laird PW: Cancer epigenetics comes of age. Nat Genet 21: 163–167, 1999
Eng C, Herman JG, Baylin, SB: A bird's eye view of global methylation. Nat Genet 24: 101–102, 2000
Adams RL, McKay EL, Craig LM, Burdon RH: Mouse DNA methylase: methylation of native DNA. Biochem Biophys Acta 561: 345–357, 1979
Razin A, Szyf M: DNA methylation patterns. Formation and function. Biochem Biophys Acta 782: 331–342, 1984
Yoder JA, Soman NS, Verdine GL, Bestor TH: DNA (cytosine-5)-methyltransferases in mouse cells and tissues. J Mol Biol 270: 385–395, 1997
Yisraeli J, Szyf M: The pattern of methylation of eukaryotic genes. In: Razin A, Cedar H, Riggs AD (eds) DNA Methylation: Biochemistry and Biological Significance. 1984, pp 353–378
Laird P: Oncogenic mechanisms mediated by DNA methylation. Mol Med Today 3: 223–229, 1997
Jones PA: The DNA methylation paradox. Trends Genet 15: 34–37, 1999
Szyf M: DNA methylation properties: consequences for pharmacology. Trends Pharmacol Sci 15: 233–238, 1994
Nan X, Campoy FJ, Bird A: MeCP2 is a transcription repressor with abundant binding sites in genomic chromatin. Cell 88: 471–481, 1997
Szyf M: The DNA methylation machinery as a target for anticancer therapy. Pharmacol Ther 70: 1–37, 1996
Costello JF, Fruhwald MC, Smiraglia DJ, Rush LJ, Robertson GP, Gao X, Wright FA, Feramisco JD, Peltomaki P, Lang JC, Schuller DE, Yu L, Bloomfield CD, Caligiuri MA, Yates A, Nishikawa R, Su Huang H-J, Petrelli NJ, Zhang X, O'Dorisio MS, Held WA, Cavenee WK, Plass C: Aberrant CpG-island methylation has non-random and tumor-type specific patterns. Nat Genet 25: 132–138, 2000
Strathdee G, MacKean MJ, Illand M, Brown R: A role for methylation of the hMLH1 promoter in loss of hMLH1 expression and drug resistance in ovarian cancer. Oncogene 18: 2335–2341, 1999
Esteller M, Garcia-Foncillas J, Andion E, Goodman SN, Hidalgo OF, Vanaclocha V, Baylin SB, Herman JG: Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N Engl J Med 343: 1350–1354, 2000
Orr RM, Monia BP: Antisense therapy for cancer. Curr Res Mol Ther 1: 102–108, 1998
Shaw JP, Kent K, Bird J, Fishback J, Froehler, B: Modified deoxyoligonucleotides stable to exonucleases degradation in serum. Nucleic Acids Res 19: 747–750, 1991
Inoue H, Hayase Y, Imura A, Iwai S, Miura K, Ohtsuka, E: Synthesis and hybridization studies on two complementary nona (2′-O-methyl) ribonucleotides. Nucleic Acid Res 15: 6131–6148, 1987
Zhang R, Iyer RP, Yu D, Tan W, Zhang X, Lu Z, Zhao H, Agrawal, S: Pharmacokinetics and tissue disposition of a chimeric oligodeoxynucleoside phosphorothioate in rats after intravenous administration. J Pharmacol Exp Ther 278: 971–979, 1996
Agrawal S, Jiang Z, Zhao Q, Shaw D, Cai Q, Roskey A, Channavajjala L, Saxinger C, Zhang, R: Mixed-backbone oligonucleotides as second generation antisense oligonucleotides: in vitro and in vivo studies. Proc Natl Acad Sci USA 94: 2620–2625, 1997
Beaulieu N, Fournel M, MacLeod AR: Antitumor activity of MG98, an antisense oligodeoynucleotide targeting DNA Methyltransferase-1 (DNMT1). Proc AACR-NCI-EORTC 728: 28,148, 2001
DeSimone J, Heller P, Hall L, Zwiers, D: 5–Azacytidine stimulates fetal hemoglobin synthesis in anemic baboons. Proc Natl Acad Sci USA 79: 4428–4431, 1982
Ley TJ, Anagnou NP, Noguchi CT, Schechter AN, DeSimone J, Heller P, Nienhuis AW: DNA methylation and globin gene expression in patients treated with 5–azacytidine. Prog Clin Biol Res 134: 457–474, 1983
Ley TJ, DeSimone J, Noguchi CT, Turner PH, Schechter AN, Heller P, Nienhuis AW: 5–Azacytidine increases gamma-globin synthesis and reduces the proportion of dense cells in patients with sickle cell anemia. Blood 62: 370–380, 1983
Carr BI, Rahbar S, Doroshow JH, Blayney D, Goldberg D, Leong L, Asmeron, Y: Fetal hemoglobin gene activation in a phase II study of 5,6–dihydro-5–azacytidine for bronchogenic carcinoma. Cancer Res 47: 4199–4201, 1987
Bourque AJ, Cohen, AS: Quantitative analysis of phosphorothioate oligonucleotides in biological fluids using direct injection fast anion-exchange chromatography and capillary gel electrophoresis. J Chromatogr B Biomed Appl 662: 343–349, 1994
Betke K, Marti HR, Schlicht I: Estimation of small percentages of foetal haemoglobin. Nature 194: 1877–1878, 1959
Ho PTC, Parkinson DR: Antisense oligonucleotides as therapeutics for malignant diseases. Semin Oncol 24: 187–202, 1997
Benimetskaya L, Tonkinson J, Stein CA: Determination of cellular internalization of fluoresceinated oligonucleotides. Meth Enzymol 313: 287–297, 2000
Henry SP, Bolte A, Auletta C, Kornbrust DJ: Evaluation of the toxicity of ISIS 2302, a phosphorothioate Oligonucleotide, in a four-week study in Cynomolgus monkeys. Toxicology 120: 145–155, 1997
Yuen AR, Halsey J, Fisher GA, Holmlund JT, Geary RS, Kwoh TJ, Dorr A, Sikic BI: Phase I study of an antisense oligonucleotide to protein kinase C-α (ISIS 3521/CGP 64128A) in patients with cancer. Clin Cancer Res 5: 3357–3363, 1999
Cunningham CC, Holmlund JT, Schiller JH, Geary RS, Kwoh TJ, Dorr A, Nemunaitis J: A phase I trial of c-raf kinase antisense oligonucleotide ISIS 5132 administered as a continuous intravenous infusion in patients with advanced cancer. Clin Cancer Res 6: 1626–1631, 2000
Waters JS, Webb A, Cunningham D, Clarke PA, Raynaud F, diStefano F, Cotter FE: Phase I clinical and pharmacokinetic study of bcl-2 antisense Oligonucleotide therapy in patients with non-Hodgkin's lymphoma. J Clin Oncol 18: 1812–1823, 2000
Stewart D, Donehower R, Eisenhauer E, Wainman N, Moore M, Bonfils C, Reid G: A phase I and pharmacokinetic (PK) study of MG98, a human DNA methyltransferase (MeTase) antisense oligonucleotide, given as a 2–hour twice weely (BIW) infusion 3 out of every 4 weeks. Proceedings of the 11th NCI/EORTC/AACR Symposium, 148 (abstract no. 528), 2000
O'Dwyer PJ, Stevenson JP, Gallagher M, Cassella A, Vasilevskaya I, Monia BP, Holmlund J, Dorr FA, Yao KS: c-raf-1 depletion and tumor responses in patients treated with the c-raf-1 antisense oligodeoxynucleotide ISIS 5132 (CGP 69846A). Clin Cancer Res 5: 3977–3982, 1999
Geary RS, Leeds JM, Henry SP, Monteith DK, Levin A: A. Antisense oligonucleotide inhibitors for the treatment of cancer: 1. Pharmacokinetic properties of phosphorothioate oligodeoxynucleotides. Anticancer Drug Des 12: 383–393, 1997
Pfeifer GP: p53 mutational spectra and the role of methylated CpG sequences. Mutat Res 450: 155–166, 2000
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Davis, A.J., Gelmon, K.A., Siu, L.L. et al. Phase I and Pharmacologic Study of the Human DNA Methyltransferase Antisense Oligodeoxynucleotide MG98 given as a 21-day Continuous Infusion Every 4 Weeks. Invest New Drugs 21, 85–97 (2003). https://doi.org/10.1023/A:1022976528441
Issue Date:
DOI: https://doi.org/10.1023/A:1022976528441