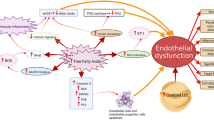
Abstract
By partially replacing the corresponding omega-6 analogues in membrane phospholipids, omega-3 fatty acids have been shown to decrease the transcriptional activation of genes — e.g., adhesion molecules, chemoattractants, inflammatory cytokines — involved in endothelial activation in response to inflammatory and pro-atherogenic stimuli. This regulation occurs, at least in part, through a decreased activation of the nuclear factor-κB system of transcription factors, secondary to decreased generation of intracellular hydrogen peroxide. Such regulation by omega-3 fatty acids is likely linked to the presence of a higher number of double bonds in the fatty acid chain in omega-3 compared with omega-6 fatty acids. By similar mechanisms, omega-3 fatty acids have been recently shown to reduce gene expression of cyclooxygenase-2, an inflammatory gene involved, through the activation of some metalloproteinases, in plaque angiogenesis and plaque rupture. The quenching of gene expression of pro-inflammatory pro-atherogenic genes by omega-3 fatty acids has consequences on the extent of leukocyte adhesion to vascular endothelium, early atherogenesis and later stages of plaque development and plaque rupture, ultimately yielding a plausible comprehensive explanation for the vasculoprotective effects of these nutrients.


Similar content being viewed by others
References
Ait-Said F., Elalamy I., Werts C., Gomard M.T., Jacquemin C., Couetil J.P., Hatmi M. 2003. Inhibition by eicosapentaenoic acid of IL-1beta-induced PGHS-2 expression in human microvascular endothelial cells: involvement of lipoxygenase-derived metabolites and p38 MAPK pathway. Biochim. Biophys. Acta. 1631:77–84
Akarasereenont P., Bakhle Y.S., Thiemermann C., Vane J.R. 1995. Cytokine-mediated induction of cyclo-oxygenase-2 by activation of tyrosine kinase in bovine endothelial cells stimulated by bacterial lipopolysaccharide. Br. J. Pharmacol. 115:401–408
Al-Fakhri N., Wilhelm J., Hahn M., Heidt M., Hehrlein F.W., Endisch A.M., Hupp T., Cherian S.M., Bobryshev Y.V., Lord R.S., Katz N. 2003. Increased expression of disintegrin-metalloproteinases ADAM-15 and ADAM-9 following upregulation of integrins alpha5beta1 and alphavbeta3 in atherosclerosis. J. Cell Biochem. 89:808–823
Arthaud B. 1970. Cause of death in 339 Alaskan natives as determined by autopsy. Arch. Pathol. 90:433–438
Bagga, D., Wang, L., Farias-Eisner, R., Glaspy, J.A., Reddy, S.T. 2003. Differential effects of prostaglandin derived from omega-6 and omega-3 polyunsaturated fatty acids on COX-2 expression and IL-6 secretion. Proc. Natl. Acad. Sci USA 100:1751–1756. Epub 2003 Feb 1710
Bang H.O., Dyerberg J., Hjorne N. 1976. The composition of food consumed by Greenland Eskimos. Acta Med. Scand. 200:69–73
Barbieri S.S., Cavalca V., Eligini S., Brambilla M., Caiani A., Tremoli E, Colli S. 2004. Apocynin prevents cyclooxygenase 2 expression in human monocytes through NADPH oxidase and glutathione redox-dependent mechanisms. Free Radic. Biol. Med. 37:156–165
Barger A.C., Beeuwkes R., 3rd, Lainey L.L., Silverman K.J. 1984. Hypothesis: vasa vasorum and neovascularization of human coronary arteries. A possible role in the pathophysiology of atherosclerosis. N. Engl. J. Med. 310:175–177
Bayes-Genis A., Conover C.A., Overgaard M.T., Bailey K.R., Christiansen M., Holmes D.R., Jr., Virmani R., Oxvig C., Schwartz R.S. 2001. Pregnancy-associated plasma protein A as a marker of acute coronary syndromes. N. Engl. J. Med. 345:1022–1029
Bea F., Blessing E., Bennett B.J., Kuo C.C., Campbell L.A., Kreuzer J., Rosenfeld M.E. 2003. Chronic inhibition of cyclooxygenase-2 does not alter plaque composition in a mouse model of advanced unstable atherosclerosis. Cardiovasc. Res. 60:198–204
Beaudeux J.L., Giral P., Bruckert E., Foglietti M.J., Chapman M.J. 2004. Matrix metalloproteinases, inflammation and atherosclerosis: therapeutic perspectives. Clin. Chem. Lab. Med. 42:121–131
Bechler K. 1997. Influence of capping and polyadenylation on mRNA expression and on antisense RNA mediated inhibition of gene expression. Biochem Biophys. Res. Commun. 241:193–199
Bode W., Grams F., Reinemer P., Gomis-Ruth F.X., Baumann U., McKay D.B., Stocker W. 1996. The metzincin-superfamily of zinc-peptidases. Adv. Exp. Med. Biol. 389:1–11
Burleigh M.E., Babaev V.R., Gates J.A., Harris R.C., Gautam S., Riendeau D., Marnett L.J., Morrow J.D., Fazio S., Linton M.F. 2002. Cycloxygenase-2 promotes early atherosclerotic lesion formation in LDL receptor-deficient mice. Circulation 105:1816–1823
Burleigh M.E., Babaev V.R., Yancey P.G., Major A.S., McCaleb J.L., Gates J.A., Morrow J.D., Fazio S., Linton M.F. 2005. Cyclooxygenase-2 promotes early atherosclerotic lesion formation in ApoE-deficient and C57BL/6 mice. J. Mol. Cell. Cardiol. 21:21
Callejas N., Casado M., Diaz-Guerra M., Bosca L., Martin-Sanz P. 2001. Expression of cyclooxygenase-2 promotes the release of matrix metalloproteinase-2 and -9 in fetal rat hepatocytes. Hepatology 33:860–867
Calviello G., Di Nicuolo F., Gragnoli S., Piccioni E., Serini S., Maggiano N., Tringali G., Navarra P., Ranelletti F.O., Palozza P. 2004. n-3 PUFAs reduce VEGF expression in human colon cancer cells modulating the COX-2/PGE2 induced ERK-1 and -2 and HIF-1alpha induction pathway. Carcinogenesis 25:2303–2310
Carluccio M.A., Massaro M., Bonfrate C., Siculella L., Maffia M., Nicolardi G., Distante A., Storelli C., De Caterina R. 1999. Oleic acid inhibits endothelial activation: A direct vascular antiatherogenic mechanism of a nutritional component in the mediterranean diet. Arterioscler. Thromb. Vasc. Biol. 19:220–228
Chandrasekharan N.V., Dai H., Roos K.L., Evanson N.K., Tomsik J., Elton T.S., Simmons D.L. 2002. COX-3, a cyclooxygenase-1 variant inhibited by acetaminophen and other analgesic/antipyretic drugs: cloning, structure, and expression. Proc. Natl. Acad. Sci. U S A 99:13926–13931
Chase A.J., Bond M., Crook M.F., Newby A.C. 2002. Role of nuclear factor-kappa B activation in metalloproteinase-1, -3, and -9 secretion by human macrophages in vitro and rabbit foam cells produced in vivo. Arterioscler. Thromb. Vasc. Biol. 22:765–771
Chen H., Li D., Roberts G.J., Saldeen T., Mehta J.L. 2003. Eicosapentanoic acid inhibits hypoxia-reoxygenation-induced injury by attenuating upregulation of MMP-1 in adult rat myocytes. Cardiovasc. Res. 59:7–13
Cipollone F., Prontera C., Pini B., Marini M, Fazia M., De Cesare D., Iezzi A., Ucchino S., Boccoli G., Saba V., Chiarelli F., Cucurullo F., Mezzetti A. 2001. Overexpression of functionally coupled cyclooxygenase-2 and prostaglandin E synthase in symptomatic atherosclerotic plaque as a basis of prostaglandin E2-dependent plaque instability. Circulation 104:921–927
Cole C.N. 2000. mRNA export: the long and widing road. Nat. Cell Biol. 2:E55–E58
Collins T. 1993. Endothelial nuclear factor-κB and the initiation of the atherosclerotic lesion. Lab. Invest. 68:499–508
Collins T., Read M.A., Neish A.S., Whitley M.Z., Thanos D., Maniatis T. 1995. Transcriptional regulation of endothelial cell adhesion molecules: NF-kappa B and cytokine-inducible enhancers. FASEB J. 9:899–909
Connolly J.M., Gilhooly E.M., Rose D.P. 1999. Effects of reduced dietary linoleic acid intake, alone or combined with an algal source of docosahexaenoic acid, on MDA-MB-231 breast cancer cell growth and apoptosis in nude mice. Nutr. Cancer 35:44–49
Corcoran M., Stetler-Stevenson W., DeWitt D., Whal L. 1994. Effect of cholera toxin and pertussis toxin on prostaglandin H synthase-2, prostaglandin E2 and matrix metalloproteinase production by human monocytes. Arch. Biochem. Biophys. 310:481–488
Crawford D.L., Powers D.A. 1992. Evolutionary adaptation to different thermal environment via transcriptional regulation. Mol. Biol. Evol. 9:806–813
Culp B.R., Titus B.G., Lands W.E. 1979. Inhibition of prostaglandin biosynthesis by eicosapentaenoic acid. Prostaglandins Med. 3:269–278
Cybulsky M.J., Iiyama K., Li H., Zhu S., Chen M., Iiyama M., Davis V.M., Gutierrez-Ramos J.-C., Connelly P.W., Milstone D.S. 2001. A major role for VCAM-1, but not ICAM-1, in early atherosclerosis. J. Clin. Invest. 107:1255–1262
De Caterina R., Bernini W., Carluccio M., Liao J., Libby P. 1998. Structural requirements for inhibition of cytokine-induced endothelial activation by unsaturated fatty acids. J. Lipid Res. 39:1062–1070
De Caterina R., Cybulsky M.A., Clinton S.K., Gimbrone M.A., Jr., Libby P. 1995. Omega-3 fatty acids and endothelial leukocyte adhesion molecules. Prostagl. Leukotr. Ess. Fatty Acids. 52:191–195
De Caterina R., Cybulsky M.I., Clinton S.K., Gimbrone M.A., Jr., Libby P. 1994. The omega-3 fatty acid docosahexaenoate reduces cytokine-induced expression of pro-atherogenic and pro-inflammatory proteins in human endothelial cells. Arterioscl. Thromb. 14:1829–1836
De Caterina R., Giannessi D., Mazzone A., Bernini W., Lazzerini G., Maffei S., Cerri M., Salvatore L., Weksler B. 1990. Vascular prostacyclin is increased in patients ingesting omega-3 polyunsaturated fatty acids before coronary artery bypass surgery. Circulation 82:428–438
De Caterina, R., Gimbrone, M.A., Jr. 1995. Leukocyte-endothelial interactions and the pathogenesis of atherosclerosis. In: n-3 Fatty Acids — Prevention and Treatment in Vascular Disease. Kristensen, SD., Schmidt, EB., De Caterina, R., Endres, S., eds. pp 9–24, Springer Verlag
De Caterina R., Madonna R., Hassan J., Procopio A.D. 2001. Nutrients and gene expression. World Rev. Nutr. Diet 89:23–52
De Caterina R., Zampolli A. 2001. n-3 fatty acids:antiatherosclerotic effects. Lipids 36:S69–S78
De Caterina R., Zampolli A. 2004. From asthma to atherosclerosis — 5-lipoxygenase, leukotrienes, and inflammation. N. Engl. J. Med. 350:4–7
De Caterina, R., Zampolli, A., Del Turco, S., Madonna, R., Massaro, M. 2006. Nutritional mechanisms that influence cardiovascular disease. Am. J. Clin. Nutr. in press
Dixon D.A., Kaplan C.D., McIntyre T.M., Zimmerman G.A., Prescott S.M. 2000. Post-transcriptional control of cyclooxygenase-2 gene expression. The role of the 3′-untranslated region. J. Biol. Chem. 275:11750–11757
Dollery C.M., Owen C.A., Sukhova G.K., Krettek A., Shapiro S.D., Libby P. 2003. Neutrophil elastase in human atherosclerotic plaques: production by macrophages. Circulation 107:2829–2836
Dyerberg J., Bang H.O. 1978. Dietary fat and thrombosis. Lancet 1:152
Dyerberg J., Bang H.O., Stofferson E., Moncada S., Vane J.R. 1978. Eicosapentaenoic acid and prevention of thrombosis and atherosclerosis. Lancet 117–119
Egan K.M., Lawson J.A., Fries S., Koller B., Rader D.J., Smyth E.M., Fitzgerald G.A. 2004. COX-2-derived prostacyclin confers atheroprotection on female mice. Science 306:1954–1957
Endres S., Ghorbani R., Kelley V.E., Georgilis K., Lonneman G., van der Meer J.W.M., Cannon J.G., Rogers T.S., Klemper M.S., Weber P.C., Schaefer E.J., Woff S.M., Dinarello C.A. 1989. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N. Engl. J. Med. 320:265–271
Eritsland J., Arnesen H., Grønseth K., Field N.B., Abdelnoor M. 1994. Effect of supplementation with n-3 fatty acids on graft patency in patients undergoing coronary artery bypass operation. Results from the SHOT study. Eur Heart J. 15:29
Falk E., Shah P.K., Fuster V. 1995. Coronary plaque disruption. Circulation 92:657–671
Fischer S., von, Schacky C., Siess W., Strasser T., Weber P.C. 1984. Uptake, release and metabolism of docosahexaenoic acid (DHA, c22:6 omega 3) in human platelets and neutrophils. Biochem. Biophys. Res. Commun. 120:907–918
Fitzgerald G.A. 2004. Coxibs and cardiovascular disease. N. Engl. J. Med. 351:1709–1711
Furberg C.D., Psaty B.M., FitzGerald G.A. 2005. Parecoxib, valdecoxib, and cardiovascular risk. Circulation 111:249
Galis Z.S., Muszynski M., Sukhova G.K., Simon-Morrissey E., Libby P. 1995. Enhanced expression of vascular matrix metalloproteinases induced in vitro by cytokines and in regions of human atherosclerotic lesions. Ann. N. Y. Acad. Sci. 748:501–507
Galis Z.S., Muszynski M., Sukhova G.K., Simon-Morrissey E., Unemori E.N., Lark M.W., Amento E., Libby P. 1994. Cytokine-stimulated human vascular smooth muscle cells synthesize a complement of enzymes required for extracellular matrix digestion. Circ. Res. 75:181–189
Galis Z.S., Sukhova G.K., Kranzhofer R., Clark S., Libby P. 1995. Macrophage foam cells from experimental atheroma constitutively produce matrix-degrading proteinases. Proc. Natl. Acad. Sci. USA 92:402–406
Galis Z.S., Sukhova G.K., Lark M.W., Libby P. 1994. Increased expression of matrix metalloproteinases and matrix degrading activity in vulnerable regions of human atherosclerotic plaques. J. Clin. Invest. 94:2493–2503
Garcia-Touchard A., Henry T.D., Sangiorgi G., Spagnoli L.G., Mauriello A., Conover C., Schwartz R.S. 2005. Extracellular proteases in atherosclerosis and restenosis. Arterioscler. Thromb. Vasc. Biol. 25:1119–1127
Gartenberg M.R., Wang J.C. 1992. Positive supercoiling of DNA greatly diminishes mRNA synthesis in yeast. Proc. Natl. Acad. Sci. USA 89:11461–11465
Geiringer E. 1951. Intimal vascularization and atherosclerosis. J. Pathol. Bacteriol. 63:201–211
Gilbert M., Dalloz S., Maclouf J., Lagarde M. 1999. Differential effects of long chain n-3 fatty acids on the expression of PGH synthase isoforms in bovine aortic endothelial cells. Prostaglandins Leukot. Essent. Fatty Acids. 60:363–365
Gong B., Chen Q., Almasan A. 1998. Ionizing radiation stimulates mitochondrial gene expression and activity. Radiat. Res. 150:505–512
Guan Z., Buckman S.Y., Pentiand A.P., Templeton D.J., Morrison A.R. 1998. Induction of cyclooxygenase-2 by the activated MEKK1→SEK1/MKK4→p38 mitogen-activated protein kinase pathway. J. Biol. Chem. 273:12901–12908
Habib A., Creminon C., Frobert Y., Grassi J., Pradelles P., Maclouf J. 1993. Demonstration of an inducible cyclooxygenase in human endothelial cells using antibodies raised against the carboxyl-terminal region of the cyclooxygenase-2. J. Biol. Chem. 268:23448–23454
Halpert I., Sires U.I., Roby J.D., Potter-Perigo S., Wight T.N., Shapiro S.D., Welgus H.G., Wickline S.A., Parks W.C. 1996. Matrilysin is expressed by lipid-laden macrophages at sites of potential rupture in atherosclerotic lesions and localizes to areas of versican deposition, a proteoglycan substrate for the enzyme. Proc. Natl. Acad. Sci. USA 93:9748–9753
Harris M.A., Hansen R.A., Vidsudhiphan P., Koslo J.L., Thomas J.B., Watkins B.A., Allen K.G. 2001. Effects of conjugated linoleic acids and docosahexaenoic acid on rat liver and reproductive tissue fatty acids, prostaglandins and matrix metalloproteinase production. Prostaglandins Leukot. Essent. Fatty Acids. 65:23–29
Hennecke H. 1990. Regulation of bacterial gene expression by metal-protein complexes. Mol. Microbiol. 4:1621–1628
Herron G.S., Banda M.J., Clark E.J., Gavrilovic J., Werb Z. 1986. Secretion of metalloproteinases by stimulated capillary endothelial cells. II. Expression of collagenase and stromelysin activities is regulated by endogenous inhibitors. J. Biol. Chem. 261:2814–2818
Higgins C.F., Peltz S.W., Jacobson A. 1992. Turnover of mRNA in prokaryotes and lower eukaryotes. Curr. Opin. Genet. Dev. 2:739–747
Hirai A., Hamazaki T., Terano T. 1980. Eicosapentaenoic acid and platelet function in Japanese. Lancet 2:1132–1133
Hirai K., Ezumi Y., Nishida E., Uchiyama T., Takayama H. 1999. Comparative study of vanadate- and phorbol ester-induced cyclo-oxygenase-2 expression in human endothelial cells. Thromb. Haemost. 82:1545–1552
Hong B.K., Kwon H.M., Lee B.K., Kim D., Kim I.J., Kang S.M., Jang Y., Cho S.H., Kim H.K., Jang B.C., Cho S.Y., Kim H.S., Kim M.S., Kwon H.C., Lee K. 2000. Coexpression of cyclooxygenase-2 and matrix metalloproteinases in human aortic atherosclerotic lesions. Yonsei Med. J. 41:82–88
Inaba T., Nagano Y., Reid J.B., Sasaki Y. 2000. A 12 bp cis-regulatory element sufficient to confer dark-inducible and light down expression to a minimal promoter in pea. J. Biol. Chem. 88:22056–22060
Jones D.A., Carlton D.P., McIntyre T.M., Zimmerman G.A., Prescott S.M. 1993. Molecular cloning of human prostaglandin endoperoxide synthase type II and demonstration of expression in response to cytokines. J. Biol. Chem. 268:9049–9054
Jones M.K., Wang H., Peskar B.M., Levin E, Itani R.M., Sarfeh I.J., Tarnawski A.S. 1999. Inhibition of angiogenesis by nonsteroidal anti-inflammatory drugs: insight into mechanisms and implications for cancer growth and ulcer healing. Nat. Med. 5:1418–1423
Kagawa Y., Nishizawa M., Suzuki M. 1982. Eicosapolyenoic acid of serum lipids of Japanese islanders with low incidence of cardiovascular diseases. J. Nutr. Sci. Vitaminol. (Tokyo). 28:441–453
Kanayasu T., Morita I., Nakao-Hayashi J., Asuwa N., Fujisawa C., Ishii T., Ito H., Murota S. 1991. Eicosapentaenoic acid inhibits tube formation of vascular endothelial cells in vitro. Lipids. 26:271–276
Kromann N., Green A. 1980. Epidemiological studies in the Upernavik district, Greenland. Acta Med. Scand. 208:401–406
Kromhout D., Bosschieter E.B., De Lezenne Coulander C. 1985. The inverse relation between fish consumption and 20-year mortality from coronary heart disease. N. Engl. J. Med. 312:1205–1209
Kumamoto M., Nakashima Y., Sueishi K. 1995. Intimal neovascularization in human coronary atherosclerosis: its origin and pathophysiologieal significance. Hum. Pathol. 26:450–456
Kuwano T., Nakao S., Yamamoto H., Tsuneyoshi M., Yamamoto T., Kuwano M., Ono M. 2004. Cyclooxygenase 2 is a key enzyme for inflammatory cytokine-induced angiogenesis. FASEB J. 18:300–310
Lee J.Y., Plakidas A., Lee W.H., Heikkinen A., Chanmugam P., Bray G., Hwang D.H. 2003. Differential modulation of Toll-like receptors by fatty acids: preferential inhibition by n-3 polyunsaturated fatty acids. J. Lipid Res. 44:479–486
Lee J.Y., Ye J., Gao Z., Youn H.S., Lee W.H., Zhao L., Sizemore N., Hwang D.H. 2003. Reciprocal modulation of Toll-like receptor-4 signaling pathways involving MyD88 and phosphatidylinositol 3-kinase/AKT by saturated and polyunsaturated fatty acids. J. Biol. Chem. 278:37041–37051
Lewin B. 1997. Genes VI. Oxford University Press, Oxford, New York, Tokyo
Lewis R.A., Austen K.F., Soberman R.J. 1990. Leukotrienes and other products of the 5-lipoxygenase pathway. Biochemistry and relation to pathobiology in human diseases. N. Engl. J. Med. 323:645–655
Li Q., Wrange O., Eriksson P. 1997. The role of chromatin in transcriptional regulation. Int. J. Biochem. Cell Biol. 29:731–742
Madri J.A., Graesser D., Haas T. 1996. The roles of adhesion molecules and proteinases in lymphocyte transendothelial migration. Biochem. Cell Biol. 74:749–757
Manger I.D., Relman D.A. 2000. How the host ‘sees’ pathogen: global gene expression responses to infection. Curr. Opin. Immunol. 12:215–218
McCarthy M.J., Loftus I.M., Thompson M.M., Jones L., London N.J., Bell P.R., Naylor A.R., Brindle N.P. 1999. Angiogenesis and the atherosclerotic carotid plaque: an association between symptomatology and plaque morphology. J. Vasc. Surg. 30:261–268
McMillan W.D., Patterson B.K., Keen R.R., Shively V.P., Cipollone M., Pearce W.H. 1995. In situ localization and quantification of mRNA for 92-kD type IV collagenase and its inhibitor in aneurysmal, occlusive, and normal aorta. Arterioseler.Thromb. Vasc. Biol. 15:1139–1144
Middaugh J.P. 1990. Cardiovascular deaths among Alaskan natives 1980–1986. Am. J. Public Health 80:282–285
Mofidi R., Crotty T.B., McCarthy P., Sheehan S.J., Mehigan D., Keaveny T.V. 2001. Association between plaque instability, angiogenesis and symptomatic carotid occlusive disease. Br. J. Surg. 88:945–950
Moulton K.S. 2002. Plaque angiogenesis: its functions and regulation. Cold Spring Harb. Symp. Quant. Biol. 67:471–482
Nagase H. 1997. Activation mechanisms of matrix metalloproteinases. Biol. Chem. 378:151–160
Nagase H., Woessner J.F. Jr. 1999. Matrix metalloproteinases. J. Biol. Chem. 274:21491–21494
Nakagawa K., Chen Y.X., Ishibashi H., Yonemitsu Y., Murata T., Hata Y., Nakashima Y., Sueishi K. 2000. Angiogenesis and its regulation: roles of vascular endothelial cell growth factor. Semin. Thromb. Hemost. 26:61–66
Newell-Price J., Clark A.J., King P. 2000. DNA methylation and silencing of gene expression. Trends Endocrinol. Metab. 11:142–148
Newman W.P., Middaugh J.P., Propst M.T., Rogers D.R. 1993. Atherosclerosis in Alaska natives and non-natives. Lancet 341:1056–1057
O’Brien K.D., Alien M.D., McDonald T.O., Chait A., Harlan J.M., Fishbein D., McCarty J., Ferguson M., Hudkins K., Benjamin C.D., et al. 1993. Vascular cell adhesion molecule-1 is expressed in human coronary atherosclerotic plaques. Implications for the mode of progression of advanced coronary atherosclerosis. J. Clin. Invest. 92:945–951
O’Brien K.D., McDonald T.O., Chait A., Alien M.D., Alpers C.E. 1996. Neovascular expression of E-selectin, intercellular adhesion molecule-1, and vascular cell adhesion molecule-1 in human atherosclerosis and their relation to intimal leukocyte content. Circulation 93:672–682
Okie S. 2005. Raising the safety bar—the FDA’s coxib meeting. N. Engl. J. Med. 352:1283–1285
Pratico D., Tillmann C., Zhang Z.B., Li H., FitzGerald G.A. 2001. Acceleration of atherogenesis by COX-1-dependent prostanoid formation in low density lipoprotein receptor knockout mice. Proc. Natl. Acad. Sci. USA 98:3358–33631
Rajavashisth T.B., Liao J.K., Galis Z.S., Tripathi S., Laufs U., Tripathi J., Chai N.N., Xu X.P., Jovinge S., Shah P.K., Libby P. 1999. Inflammatory cytokines and oxidized low density lipoproteins increase endothelial cell expression of membrane type 1-matrix metalloproteinase. J. Biol. Chem. 274:11924–11929
Rajavashisth T.B., Xu X.P., Jovinge S., Meisel S., Xu X.O., Chai N.N., Fishbein M.C., Kaul S., Cercek B., Sharifi B., Shah P.K. 1999. Membrane type 1 matrix metalloproteinase expression in human atherosclerotic plaques: evidence for activation by proinflammatory mediators. Circulation 99:3103–3109
Rikitake Y., Hirata K., Kawashima S., Takeuchi S., Shimokawa Y., Kojima Y., Inoue N., Yokoyama M. 2001. Signaling mechanism underlying COX-2 induction by lysophosphatidylcholine. Biochem. Biophys. Res. Commun. 281:1291–1297
Romanic A.M., Madri J.A. 1994. The induction of 72-kD gelatinase in T cells upon adhesion to endothelial cells is VCAM-1 dependent. J. Cell Biol. 125:1165–1178
Rose D.P., Connolly J.M. 1999. Antiangiogenicity of docosahexaenoic acid and its role in the suppression of breast cancer cell growth in nude mice. Int. J. Oncol. 15:1011–1015
Rose D.P., Connolly J.M., Coleman M. 1996. Effect of omega-3 fatty acids on the progression of metastases after the surgical excision of human breast cancer cell solid tumors growing in nude mice. Clin. Cancer Res. 2:1751–1756
Rott D., Zhu J., Burnett M.S., Zhou Y.F., Zalles-Ganley A., Ogunmakinwa J., Epstein S.E. 2003. Effects of MF-tricyclic, a selective cyclooxygenase-2 inhibitor, on atherosclerosis progression and susceptibility to cytomegalovirus replication in apolipoprotein-E knockout mice. J. Am. Coll. Cardiol. 41:1812–1819
Rueckschloss U., Duerrschmidt N., Morawietz H. 2003. NADPH oxidase in endothelial cells: impact on atherosclerosis. Antioxid. Redox Signal 5:171–180
Schnaper H.W., Grant D.S., Stetler-Stevenson W.G., Fridman R., D’Orazi G., Murphy A.N., Bird R.E., Hoythya M., Fuerst T.R., French D.L., et al. 1993. Type IV collagenase(s) and TIMPs modulate endothelial cell morphogenesis in vitro. J. Cell Physiol. 156:235–246
Schonbeck U., Mach F., Sukhova G.K., Atkinson E., Levesque E., Herman M., Graber P., Basset P., Libby P. 1999. Expression of stromelysin-3 in atherosclerotic lesions: regulation via CD40-CD40 ligand signaling in vitro and in vivo. J. Exp. Med. 189:843–853
Schonbeck U., Sukhova G.K., Graber P., Coulter S., Libby P. 1999. Augmented expression of cyclooxygenase-2 in human atherosclerotic lesions. Am. J. Pathol. 155:1281–1291
Seals D.F., Courtneidge S.A. 2003. The ADAMs family of metalloproteases: multidomain proteins with multiple functions. Genes Dev. 17:7–30
Shah P.K., Falk E., Badimon J.J., Fernandez-Ortiz A., Mailhac A., Villareal-Levy G., Fallon J.T., Regnstrom J., Fuster V. 1995. Human monocyte-derived macrophages induce collagen breakdown in fibrous caps of atherosclerotic plaques. Potential role of matrix-degrading metalloproteinases and implications for plaque rupture. Circulation 92:1565–1569
Shekelle R., Missel L., Paul O., MacMillan-Schryock A., Stamler J. 1985. Fish consumption and mortality from coronary heart disease (Letter). N. Engl. J. Med. 313:820
Shtivelband M.I., Juneja H.S., Lee S., Wu K.K. 2003. Aspirin and salicylate inhibit colon cancer medium- and VEGF-induced endothelial tube formation: correlation with suppression of cyclooxygenase-2 expression. J. Thromb. Haemost. 1:2225–2233
Smith G.R. 1981. DNA supercoiling: another level for regulating gene expression. Cell. 24:599–600
Smith W.L., Garavito R.M., DeWitt D.L. 1996. Prostaglandin endoperoxide H synthases (cyclooxygenases)-1 and -2. J. Biol Chem. 271:33157–33160
Soei, L., Lamers, J., Sassen, L., van Tol, A., Scheek, L., Dekkers, D., van Meegen, J., Verdouw, P. Fish oil: a modulator of experimental atherosclerosis in animals. In: n-3 Fatty Acids: Prevention and Treatment in Vascular Disease. Kristensen, S., Endres, S., De Caterina, R., Schmidt, E., eds. pp 77–84. Springer Verlag
Spector A.A., Kaduce T.L., Figard P.H., Norton K.C., Hoak J.C., Czervionke R.L. 1983. Eicosapentaenoic acid and prostacyclin production by cultured human endothelial cells. J. Lipid Res. 24:1595–1604
Stary H.C., Chandler A.B., Dinsmore R.E., Fuster V., Glagov S., Insull W., Jr., Rosenfeld M.E., Schwartz C.J., Wagner W.D., Wissler R.W. 1995. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 92:1355–1374
Stemme V., Swedenborg J., Claesson H., Hansson G.K. 2000. Expression of cyclo-oxygenase-2 in human atherosclerotic carotid arteries. Eur. J. Vasc. Endovasc. Surg. 20:146–152
Stocker V., Grams F., Baumann U., Reinemer P., Gomis-Ruth F.X., McKay D.B., Bode W. 1995. The metzincins—topological and sequential relations between the astacins, adamalysins, serralysins, and matrixins (collagenases) define a superfamily of zinc-peptidases. Protein Sci. 4:823–840
Sukhova G.K., Schonbeck U., Rabkin E., Schoen F.J., Poole A.R., Billinghurst R.C., Libby P. 1999. Evidence for increased collagenolysis by interstitial collagenases-1 and -3 in vulnerable human atheromatous plaques. Circulation 99:2503–2509
Suzuki I., Iigo M., Ishikawa C., Kuhara T., Asamoto M., Kunimoto T., Moore M.A., Yazawa K., Araki E., Tsuda H. 1997. Inhibitory effects of oleic and docosahexaenoic acids on lung metastasis by colon-carcinoma-26 cells are associated with reduced matrix metalloproteinase-2 and -9 activities. Int. J. Cancer. 73:607–612
Tanabe T., Tohnai N. 2002. Cyclooxygenase isozymes and their gene structures and expression. Prostaglandins Other Lipid Mediat. 68–69:95–114
The GISSI-Prevenzione Investigators: 1999. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamins E after myocardial infarction: results of the GISSI-Prevenzione trial. Lancet 354:447–455
Thies F., Garry J.M., Yaqoob P., Rerkasem K., Williams J., Shearman C.P., Gallagher P.J., Calder P.C., Grimble R.F. 2003. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: a randomised controlled trial. Lancet 361:477–485
Topol E.J. 2004. Failing the public health—rofecoxib, Merck, and the FDA. N. Engl. J. Med. 351:1707–1709
Tsuji M., Murota S.I., Morita I. 2003. Docosapentaenoic acid (22:5, n-3) suppressed tube-forming activity in endothelial cells induced by vascular endothelial growth factor. Prostaglandins Leukot. Essent. Fatty Acids 68:337–342
van der Wal A.C., Decker A.E., van der Loos C.M., Das P.K. 1994. Site of intimal rupture or erosion of thrombosed coronary atherosclerotic plaques is characterized by an inflammatory process irrespective of the dominant plaque morphology. Circulation 89:36–44
Verderber E., Lucast L.J., Van Dehy J.A., Cozart P., Etter J.B. 1997. Role of the hemeA gene product and delta aminolevulinic acid in regulation of Escherichia Coli heme synthasis. J. Bacteriol. 179:4583–4590
Weber C., Erl W., Pietsch A., Danesch U., Weber P. 1995. Docosahexaenoic acid selectively attenuates induction of vascular cell adhesion molecule-1 and subsequent monocytic cell adhesion to human endothelial cells stimulated by tumor necrosis factor-alpha. Arterioscler. Thromb. Vasc. Biol. 15:622–628
Winternitz, M., Thomas, R., LeCompte, P. 1938. The Biology of Atherosclerosis. Thomas, C.C., ed. Springfield
Yang S.P., Morita I., Murota S.I. 1998. Eicosapentaenoic acid attenuates vascular endothelial growth factor-induced proliferation via inhibiting Flk-1 receptor expression in bovine carotid artery endothelial cells. J. Cell Physiol. 176:342–349
Zhang Y., Cliff W.J., Schoefl G.I., Higgins G. 1993. Immunohistochemical study of intimal microvessels in coronary atherosclerosis. Am. J. Pathol. 143:164–172
Zucker S., Mirza H., Conner C.E., Lorenz A.F., Drews M.H., Bahou W.F., Jesty J. 1998. Vascular endothelial growth factor induces tissue factor and matrix metalloproteinase production in endothelial cells: conversion of prothrombin to thrombin results in progelatinase A activation and cell proliferation. Int. J. Cancer. 75:780–786
Acknowledgment
We are indebted to all our former and current collaborators at the Laboratory for Thrombosis and Vascular Research at the CNR Institute of Clinical Physiology in Pisa, the Laboratory for Atherosclerosis Research at the CNR Institute of Clinical Physiology in Lecce, and the Experimental Cardiology Laboratory at the “G. d’Annunzio” Foundation in Chieti, for the experimental data on which most of the considerations here highlighted are based.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Caterina, R., Massaro, M. Omega-3 Fatty Acids and the Regulation of Expression of Endothelial Pro-Atherogenic and Pro-Inflammatory Genes. J Membrane Biol 206, 103–116 (2005). https://doi.org/10.1007/s00232-005-0783-2
Received:
Issue Date:
DOI: https://doi.org/10.1007/s00232-005-0783-2