A 59-year-old man presented to the emergency department with fever and chills that began 6 weeks earlier, when a symptomatic deep vein thrombosis (DVT) of the right posterior tibial vein had been diagnosed. The patient had also noticed word-finding difficulties and mild headache for the week before the current presentation. He did not have any chest pain, nausea or loss of consciousness. The patient was receiving anticoagulation treatment for his distal DVT with rivaroxaban (20 mg daily) because of his history of a squamous cell carcinoma of the tongue, which had been treated 2 years earlier.
On physical examination, the patient’s body temperature was 38.1°C, with otherwise normal vital signs. Cardiac auscultation found a 3/6 holosystolic murmur heard best over the apex. Neurologic examination was normal except for a subtle expressive aphasia. Vascular examination of the extremities was normal. Initial laboratory tests reported a leukocyte count of 4.4 (normal 4.8–10.8) × 109/L, a hemoglobin level of 131 (normal 140–175) g/L, an erythrocyte sedimentation rate of 17 (normal < 10) mm/h and a creatinine level of 92 (normal 49–93) μmol/L. The patient’s liver enzyme and electrolyte levels were normal. Two blood culture sets were drawn.
Which neurologic diagnosis needs to be ruled out in this patient?
Hemorrhagic stroke
Cardiogenic or paradoxical embolic stroke
Infection of the central nervous system
Cerebral metastasis
All of the above
The answer is (e). Our patient’s history of cancer and recent DVT raised concerns about cerebral metastasis and paradoxical embolic stroke. In addition, we were concerned about infective endocarditis, given the combination of fever, a new heart murmur and aphasia. Septic emboli can lead to ischemic or hemorrhagic strokes, meningitis or cerebral abscesses, and would not be prevented by the anticoagulation treatment for DVT.
What additional investigations should be ordered urgently?
Lumbar puncture
Computed tomography (CT) of the head, with and without contrast
Two additional blood cultures drawn at least an hour later
Transthoracic echocardiography
Transesophageal echocardiography
The answer is (b). Although (c), (d) and (e) were all indicated investigations, a CT of the head was most urgent because our patient had acute neurologic signs while taking anticoagulants. Computed tomography angiography (CTA) can identify a proximal arterial occlusion or an intracranial mycotic aneurysm.1,2 The American Heart Association (AHA) 2015 guideline recommends cerebrovascular imaging in all patients with a suspected diagnosis of infective endocarditis presenting with neurologic symptoms.1 Computed tomography angiography, magnetic resonance angiography or digital subtraction angiography may be used when intracranial aneurysms are suspected and should also be considered with left-sided infective endocarditis for early detection of mycotic aneurysms, even in the absence of neurologic signs.1
When infective endocarditis is suspected, the AHA guideline recommends at least 3 sets of blood cultures, obtained from different venipuncture sites, with first and last samples drawn at least 1 hour apart.1 Additional blood cultures should therefore be obtained without delay because the possibility of sepsis warrants prompt initiation of antibiotic treatment. We began empirical therapy with intravenous ceftriaxone (2 g every 12 h) and vancomycin (1 g every 8 h) to cover the most common pathogens associated with subacute community-acquired native valve endocarditis (i.e., staphylococci, streptococci and enterococci).1,2 Because we suspected cerebral septic emboli, meningeal doses were given.
Transthoracic echocardiography should also be performed as soon as endocarditis is suspected. Transesophageal echocardiography is more sensitive and is indicated in patients with a negative or nondiagnostic transthoracic echocardiograph and a high index of suspicion for endocarditis. Transesophageal echocardiography is also mandatory when clinicians suspect intracardiac complications, such as a paravalvular abscess, a pseudoaneurysm or a fistula, and may be considered in all patients after a positive result on transthoracic echocardiography. 1,2 Lumbar puncture was contraindicated in our patient because of oral anticoagulation treatment.
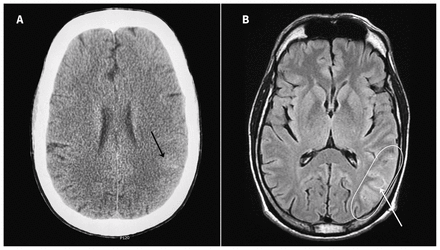
Computed tomography and magnetic resonance imaging of our patient’s head showed a left parietal–temporal subacute ischemic stroke with signs of localized adjacent nontraumatic convexal subarachnoid hemorrhage, without arterial abnormalities (Figure 1). Rivaroxaban was immediately suspended. Both types of echocardiography confirmed a new-onset severe mitral valve regurgitation and multiple vegetations (≤ 5 mm) on its anterior leaflet. No patent foramen ovale was identified by bubble studies using agitated saline.
Axial views of (A) computed tomography and (B) fluid-attenuated inversion recovery magnetic resonance imaging of the head showing left parietal–temporal acute ischemic stroke (oval) with signs of localized adjacent convexal subarachnoid hemorrhage (black and white arrows).
Four blood cultures identified the presence of Enterococcus faecalis susceptible to penicillin, ampicillin, vancomycin and gentamicin (our interpretation was based on the Clinical and Laboratory Standards Institute guideline [https://clsi.org/] and reported as categorical results). Our patient’s echocardiographic and cerebral imaging findings met the Duke diagnostic criteria for infective endocarditis with probable cerebral emboli.1 We adjusted our patient’s treatment with antibiotics to intravenous penicillin G (4 million units every 4 h), and we added gentamicin (3 mg/kg divided in 2 doses) for its synergistic effect. Ceftriaxone plus ampicillin would have been an appropriate alternative regimen, especially in the presence of chronic kidney disease or frailty, where the risk of aminoglycoside toxicity is especially high.1
Endocarditis related to E. faecalis is usually associated with genitourinary procedures or bacteriuria, neither of which applied to our patient.3 Other potential sources include colorectal neoplasms, and inflammatory lesions of the gut and biliary tree. For this reason, we arranged for CT of the abdomen and pelvis, and a colonoscopy, all of which were normal.3,4 These investigations, combined with a thorough history and physical examination, completed our patient’s age-appropriate cancer screening, which is indicated in the context of idiopathic DVT.5 Because of our patient’s history of tongue carcinoma, we also investigated cancer recurrence with nasolaryngoscopy and CT of the neck, both of which were negative. A dental examination was also normal.
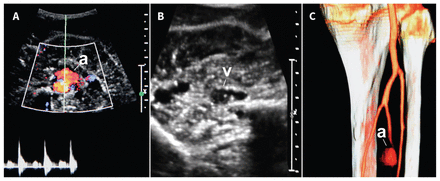
We ordered venous Doppler ultrasonography to reassess the DVT in our patient’s right tibial vein. It showed an unchanged distal DVT, but the vein appeared compressed by a vascular structure with arterial Doppler flow (Figure 2A, Figure 2B). An abdominal and lower extremity CTA confirmed a right fibular artery aneurysm (Figure 2C).
(A) Transverse view of a right fibular artery aneurysm on 1 cm–scaled ultrasonography. Colour Doppler ultrasonography showing arterial flow within a 2 cm aneurysm (a). (B) Doppler ultrasonography of the leg showing that the adjacent tibial vein is compressed by the aneurysm and thrombosed distally (v). (C) Three-dimensional reconstruction of the distal right leg arteries by computed tomography angiography showing a 2 cm aneurysm (a) of the right fibular artery.
What is the next step in management?
Observe and repeat CTA of the right leg in 6 weeks
Restart anticoagulation therapy with intravenous heparin
Surgically excise the aneurysm and ligate the right fibular artery
Proceed with emergent arterial embolization of the right fibular artery
Proceed with filter placement in the inferior vena cava
The answer is (c). In the context of infective endocarditis, we suspected a mycotic aneurysm of the right fibular artery that originated from a septic embolus. Rapid initiation of appropriate treatment with intravenous antibiotics is essential in controlling sepsis and lowers the risk of recurrent embolization.1 Prompt surgical intervention is required because of the risk of rupture with potential hemorrhage or loss of limb.6
There is inadequate evidence to suggest endovascular treatment of a peripheral mycotic aneurysm.7 We decided against using an inferior vena cava filter, given the distal localization of the DVT, the associated bacteremia and a treatable compressive cause.
Discussion
Our patient was a cancer survivor who was treated for a distal DVT, which was presumed idiopathic at the time. He subsequently developed infective endocarditis that was complicated by an embolic stroke and we diagnosed a peripheral mycotic aneurysm adjacent to the DVT. Persistent fever over weeks is unusual for a treated DVT and warrants investigation for other causes of fever, including bacteremia. Although associated with prosthetic valves, endocarditis affects native valves in 80% of cases. Our patient’s enterococcal endocarditis began insidiously. His leukocyte count was within the reference range, as was found in 53% of patients in a retrospective cohort study.8 We could not establish the source of infection, as is the situation for most cases (70%) of enterococcal endocarditis.4
Our patient’s fibular aneurysm was caused by a septic embolus that created a focus of infection in the fibular artery wall, and it is possible that a small mycotic aneurysm was missed by the first ultrasonography. The local infection and inflammation may have caused a DVT in the contiguous posterior tibial vein. Compression by the growing mycotic aneurysm, as reported in previous case series, could explain the persistence of the DVT despite anticoagulation treatment.9
Mycotic aneurysm is reported in 2%–4% of cases of infective endocarditis and arises from septic embolism, most frequently from mitral valve vegetations.6 Most mycotic aneurysms are caused by Staphylococcus and Salmonella species but may also be caused by Streptococcus and Enterococcus species and gram-negative organisms. 10 They can affect intracranial, peripheral or thoracoabdominal arteries.6 Patients often present with an erythematous and painful pulsatile mass, but other presentations include acute limb ischemia, constitutional symptoms and coexisting local soft tissue infection. The AHA mycotic aneurysm guideline recommends Doppler ultrasonography, CTA or both to diagnose peripheral aneurysms.6
Management of peripheral infected aneurysms is mainly surgical. 6 Different procedures, such as ligation, excision and in situ reconstruction, or excision with extra-anatomic revascularization, are supported by retrospective studies.6 A 2018 study involving 28 patients with peripheral mycotic aneurysms found no difference in limb-threatening ischemia when comparing revascularization to resection and ligation only.11 In the absence of randomized prospective data, the AHA advocates for individualized management.6 The results of a 2017 systematic review involving patients with infected iliofemoral arterial pseudoaneurysms showed that endovascular stents for iliofemoral mycotic aneurysms, combined with surgical débridement, favoured low complication and high patency rates.7 This option might be offered to patients who are at high surgical risk, although evidence is insufficient to suggest it. Intravenous antibiotics are usually continued up to at least 6 weeks after surgery.6
Case revisited
Our patient underwent resection of the aneurysm with ligation of the right fibular artery. Collateral tibial arteries were sufficient for distal perfusion. No intracranial or extracranial aneurysms were found in a subsequent CTA. Replacement of the mitral valve was delayed until 4 weeks had passed after the diagnosis of hemorrhagic stroke, in part because of the necessity for anticoagulation treatment during heart surgery. The patient remained hemodynamically and neurologically stable before and after the valve replacement, and made a complete recovery.
CMAJ invites submissions to “What is your call?” Clinical details (including images) are presented with a multiple-choice question about the diagnosis. The answer and a brief discussion of the condition follow. We specifically invite submissions illustrating common or important radiographic and electrocardiographic diagnoses of appeal to a general audience. We require authors to obtain consent from the patient for publication of his or her story. Submit manuscripts online at http://mc.manuscriptcentral.com/cmaj
Acknowledgement
The authors thank Dr. André Lamarre, interventional radiologist at CHU de Québec-Université Laval, for his support.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
The authors have obtained patient consent.
Contributors: All of the authors contributed to the conception and design of the work, drafted the manuscript, revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: Benoit M.-Labbé is supported by the Quebec Heart and Lung Institute – Université Laval Chair in Knowledge Translation, Education and Prevention in Respiratory and Cardiovascular Health. Alexandre Lafleur is supported by the CMA-MD Educational Leadership Chair in Health Professions Education.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/