Abstract
Background: It is uncertain whether metformin use is associated with reduced risk of joint replacement in patients with type 2 diabetes mellitus. We aimed to establish whether metformin use was associated with a reduced risk of total knee replacement (TKR) or total hip replacement (THR) among these patients.
Methods: We selected patients with type 2 diabetes mellitus that was diagnosed between 2000 and 2012 from the Taiwan National Health Insurance Research Database. We used prescription time-distribution matching and propensity-score matching to balance potential confounders between metformin users and nonusers. We assessed the risks of TKR or THR using Cox proportional hazards regression.
Results: We included 20 347 participants who were not treated with metformin and 20 347 who were treated with metformin, for a total of 40 694 participants (mean age 63 yr, standard deviation 11 yr; 49.8% were women) after prescription time-distribution matching. Compared with participants who did not use metformin, those who used metformin had lower risks of TKR or THR (adjusted hazard ratio [HR] 0.70, 95% confidence interval [CI] 0.60–0.81 for TKR or THR; adjusted HR 0.71, 95% CI 0.61–0.84 for TKR; adjusted HR 0.61, 95% CI 0.41–0.92 for THR) after adjustment for covariates. Propensity-score matching analyses (10 163 participants not treated with metformin v. 10 163 treated with metformin) and sensitivity analyses using inverse probability of treatment weighting and competing risk regression showed similar results.
Interpretation: Metformin use in patients with type 2 diabetes mellitus was associated with a significantly reduced risk of total joint replacement. Randomized controlled clinical trials in patients with osteoarthritis are warranted to determine whether metformin is effective in decreasing the need for joint replacement.
Osteoarthritis (OA) is a chronic joint disorder and a leading cause of pain, loss of function and disability among older adults.1 It is a heterogeneous disease characterized by multifactorial causes and complex phenotypes, including synovial inflammation, osteoporotic, articular cartilage degradation and metabolic phenotypes. 2,3 Total joint replacements are often required in patients with advanced OA.4 The demand for total knee replacement (TKR) and total hip replacement (THR) in the United States is expected to reach 3.5 million and 572 000 annually by 2030, respectively.5,6 No drugs can prevent or reverse the progression of OA disease.7
Diabetes mellitus has been identified as a risk factor for OA, independent of body mass index,8 and studies have reported that insulin resistance contributes to the development of OA.9 Metformin is often the first drug used to treat diabetes mellitus and is the most commonly prescribed drug worldwide for patients with this condition because of its glucose-lowering effect and excellent safety profile.10 Metformin use has also been associated with a reduced risk of cardiovascular11 and inflammatory diseases,12 as well as all-cause mortality.13 Metformin does not affect weight,14 improves insulin resistance15 and has anti-inflammatory and chondroprotective effects.16 Recent animal studies have shown that, when administered shortly after joint injury, metformin delayed the development of OA in an injury-induced OA model;17 intragastric or intra-articular metformin attenuated cartilage degradation and modulated pain in 2 OA mouse models.18,19
Observational studies examining the association of metformin use with OA-related outcomes are uncommon and inconclusive. Although 2 studies reported that metformin use was associated with reduced annual loss of medial cartilage volume20 or joint replacement surgery21 in obese people with knee OA and people with diabetes and OA, another study reported no significant association between metformin use and the risk of OA among patients with type 2 diabetes mellitus.22 Recently, the authors of a retrospective cohort study with a 4-year follow-up among patients with diabetes found that, compared with patients who did not use metformin, those who used metformin regularly had a 19% lower hazard of TKR.23
We sought to determine whether metformin use was associated with the risk of TKR or THR among patients with type 2 diabetes mellitus.
Methods
Data sources
We used data from Taiwan’s National Health Insurance Research Database (NHIRD), which is maintained by Taiwan’s National Research Agency for research purposes. It contains all registration files and original claims data from National Health Insurance beneficiaries, including inpatient care, outpatient visits, diagnosis codes, medication prescriptions and personal information. The National Health Insurance is a mandatory health insurance program that covers 99% of Taiwan’s population of 23 million. Taiwan’s National Health Research Institutes have restricted the proportion of data given to researchers to 10% or less of Taiwan’s population. We used the Longitudinal Health Insurance Database 2000, a subset of NHIRD, comprising 1 million people who were randomly sampled from the 2000 Registry for Beneficiaries of the NHIRD and which included their 1997–2013 medical claim records. All data were validated by the Bureau of National Health Insurance to ensure the accuracy of exposures, outcomes and disease diagnoses.
Study participants
We identified patients who received diagnoses of diabetes mellitus between January 2000 and December 2012 using the International Classification of Diseases, Ninth Revision (ICD-9) codes 250.xx. A sensitivity of 90.9% and positive predictive value of 92.0% has been reported for the diagnosis of diabetes mellitus.24 We defined diseases using ICD-9 codes. We excluded patients with type 1 diabetes, those younger than 45 years at the time diabetes mellitus was diagnosed, those with an index date after June 2013, those who died or had a joint replacement before the index date, those admitted for knee or hip OA before the index date and those who were prescribed metformin more than 180 days before the diagnosis of diabetes mellitus.
Exposure
We defined the exposure as metformin use within 180 days after or before a new diagnosis of type 2 diabetes mellitus between January 2000 and December 2012. For participants treated with metformin, we defined the index date as 3 months after the first prescription of metformin. To deal with immortal time bias, we individually assigned index dates to metformin nonusers that corresponded to the index date for metformin users — matched by age (within 3 yr), sex and time of physician visit for first diagnosis of type 2 diabetes mellitus (within 180 d) — using prescription time-distribution matching (PTDM).25 We defined the time from 9 months before the index date to the index date as the baseline period; the follow-up points of interest were the index date, and 12 months and 24 months after the first metformin prescription (Appendix 1, Supplementary Figure 1, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.220952/tab-related-content). We calculated the daily dosages of metformin use from the first prescription to the 3 time points.
Outcomes
We defined the main outcomes as a TKR or THR after the index date. The accuracy of the data on TKR and THR was high because all reimbursements were peer-reviewed, given their substantial cost.26 We calculated observed person-months from the sum of the follow-up time for each participant from the index date until the time of total joint replacement or censoring point (death or end of study), whichever came first.
Covariates
We included demographics, diabetes severity, comorbidities, medications for pain management (systemic corticosteroids, nonsteroidal anti-inflammatory drugs, glucosamine and opioids), other oral antihyperglycemic agents for diabetes mellitus (sulfonylureas, α-glucosidase inhibitors, thiazolidinediones and inhibitors of dipeptidyl peptidase-4) and insulin as covariates. Comorbidities included OA (ICD-9 code 715), obesity (ICD-9 code 278), hypertension (ICD-9 code 401), hyperlipidemia (ICD-9 code 272), depression (ICD-9 code 296), chronic obstructive pulmonary diseases (ICD-9 code 490–492, 493–496), gout (ICD-9 code 274), rheumatoid arthritis (ICD-9 code 714.0) and osteoporosis (ICD-9 code 733). To include these comorbidities, the ICD-9 code had to be recorded in at least 2 outpatient records or 1 admission record during the baseline period. We quantified diabetes severity using an adaption of the Diabetes Complication Severity Index.27
Statistical analysis
In addition to PTDM, we used propensity-score matching (PSM) to further balance potential confounders.28 We calculated the propensity score (probability) of metformin use by logistic regression with age, gender, year of diabetes diagnosis, insurance type, diabetes severity, comorbidities, pain medications, other oral antihyperglycemic agents and insulin at baseline as predictors. We then matched each metformin nonuser 1:1 to a participant who did use metformin with a similar propensity score using the nearest neighbour greedy algorithm with the caliper of 0.01. We considered an absolute standardized mean difference of less than 0.1 between the 2 groups after PSM as well balanced.29
We calculated the incidence rates of TKR or THR in the PTDM and PSM cohorts. We used univariable and multivariable Cox proportional hazards models in these cohorts to estimate crude and adjusted hazard ratios (HRs) with Wald confidence intervals (CIs). We reported cumulative incidence curves estimated by the Kaplan–Meier method, with a comparison between groups by the log-rank test. We also used a time-dependent Cox model to explore the effects of time-varying exposure of metformin after adjustment for the time-dependent covariates.
To verify the results, we conducted sensitivity analyses using the inverse probability of treatment weighting method in adjusted multivariable Cox proportional hazards regression to balance and control for covariates across study groups.30 We used a Fine and Gray competing risk regression model to estimate the adjusted subdistribution HRs in another sensitivity analysis, where we assumed that the competing events — including death, admission for lower limb fracture (ICD-9 code 820–829) or amputation (ICD-9 code 896, 897 or ICD-9 operation code 84.12–84.19) — precluded future TKR or THR.31 In addition, we explored the diseases that resulted in TKR or THR, and we performed sensitivity analyses by using OA-related TKR or THR as outcomes. Furthermore, we conducted the analyses with the alternative outcome of hospital admission for knee or hip OA regardless of the type of treatment.
To examine the potential dose–response relationship between metformin use and risk of total joint replacement, we categorized participants into 3 groups based on daily dosage of metformin (0 g, < 1.0 g and ≥ 1.0 g) at 3, 12 and 24 months after the first prescription. We also categorized participants treated with metformin as either continuing users (defined as participants who had ever used metformin, including within 9 months before index date), and previous users (defined as participants who had previously used metformin, but not within 9 months before index date) at 12 and 24 months after first prescription of metformin. We evaluated dose–response effects using adjusted Cox proportional hazards models. Given very few missing data, we did not perform multiple imputation.
We considered a 2-tailed test with a p value less than 0.05 to be statistically significant, without adjustment for multiplicity. We performed statistical analysis using SAS version 9.4 (SAS Institute) with propensity-score estimation and matching performed by the PSMATCH procedure.
Ethics approval
Ethics approval for the study was granted by the Chung Shan Medical University Hospital Human Research Ethics Committee (Institutional Review Board approval no. CS15134). We used anonymous data sets. Informed consent was waived owing to the retrospective nature of this observational study.
Results
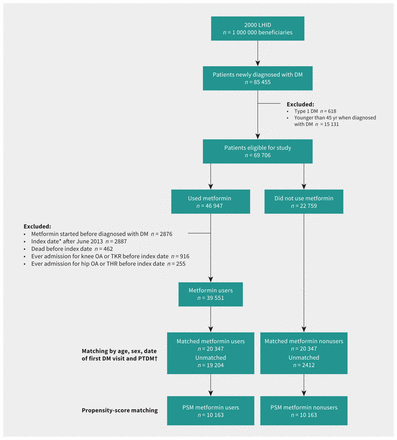
We identified 85 455 participants who had new diagnoses of diabetes mellitus during the study period. After ineligibility exclusions and 1:1 matching by age, sex and date of first visit for diagnosis of diabetes mellitus using PTDM, 20 347 participants were in each of the metformin user and nonuser groups (Figure 1). A total of 837 patients underwent TKR and 148 underwent THR in the PTDM cohorts. An average of 90% of total joint replacements were related to OA (97.5% for TKR, 50.7% for THR) (Appendix 1, Supplementary Table 1). Detailed information about the diseases associated with total joint replacement are shown in Appendix 1, Supplementary Table 2 and Supplementary Table 3.
Study flowchart. Note: DM = diabetes mellitus, LHID = the Longitudinal Health Insurance Database, OA = osteoarthritis, PSM = propensity-score matching, PTDM = prescription time-distribution matching, THR = total hip replacement, TKR = total knee replacement. *Index date for metformin users was 3 months after first prescription of metformin. †Participants who did not use metformin were individually matched with those who did use metformin by PTDM.
The characteristics of participants at baseline are shown in Table 1. After PTDM, the 2 groups were similar in age, sex, year of diagnosis of diabetes mellitus, urbanization status, pain relief medications, other oral antihyperglycemic agents and insulin at baseline, but had different types of insurance. Compared with nonusers, participants who did use metformin had more severe diabetes (i.e., a higher level of adapted Diabetes Complication Severity Index), had a higher prevalence of hypertension and hyperlipidemia, had a lower prevalence of depression and were more likely to be prescribed other oral antihyperglycemic agents or insulin at baseline. After PSM, the 10 163 participants remaining in each group were well matched (all absolute standardized mean differences < 0.1).
Baseline characteristics among participants who did or did not use metformin
Association of metformin use with incidence of total joint replacement
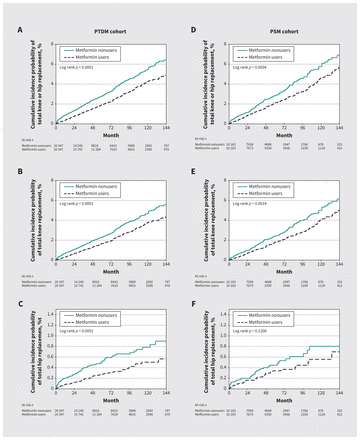
Use of metformin was associated with a lower cumulative incidence probability of TKR, THR or either total joint replacement (Figure 2). In the PTDM cohort, the incidence of hip or knee joint replacement over the study period was 4.99 per 10 000 person-months among nonusers and 3.40 per 10 000 person-months among participants who did use metformin. The crude HR was 0.69 (95% CI 0.60–0.78), comparing those who used metformin with those who did not, and the HR was 0.70 (95% CI 0.60–0.81) after adjustment for baseline covariates (Table 2).
Cumulative probability of (A) total hip or knee replacement, (B) total knee replacement and (C) total hip replacement in a prescription time-distribution matched (PTDM) cohort of participants who did or did not use metformin, and of (D) total hip or knee replacement, (E) total knee replacement and (F) total hip replacement in a propensity-score matched (PSM) cohort of participants who did or did not use metformin. We conducted log-rank tests to compare the distributions of cumulative incidence curves, estimated by the Kaplan–Meier method, between the 2 groups (metformin nonusers v. metformin users).
Association of metformin use with the incidence of total joint replacement at 3 months after index date
The incidence of TKR among metformin nonusers and users was 4.15 per 10 000 person-months and 2.96 per 10 000 person-months, respectively; and the incidence of THR was 0.83 and 0.44 per 10 000 person-months, respectively. Participants who used metformin had significantly lower risks of TKR (adjusted HR 0.71, 95% CI 0.61–0.84) and THR (adjusted HR 0.61, 95% CI 0.41–0.92) than nonusers, after adjustment for covariates (Table 2).
In the PSM cohort, the crude HR of total joint replacement was 0.73 (95% CI 0.61–0.87), and the adjusted HR was 0.75 (95% CI 0.62–0.89), comparing participants who did use metformin with those who did not. Metformin users had a significantly lower risk of TKR (adjusted HR 0.76, 95% CI 0.62–0.92) than nonusers, after adjustment for covariates. The estimated association between metformin use and incidence of THR was similar but not statistically significant (adjusted HR 0.71, 95% CI 0.44–1.13) in the PSM cohort (Table 2).
Dose–response relationships
Compared with metformin nonusers, the risk of joint replacement was significantly lower among those taking less than 1.0 g metformin per day and among those taking 1.0 g per day or more, after adjustment for covariates, although the results were statistically significant only when the daily dose was calculated from first prescription to the 3-month point (Table 3 and Appendix 1, Supplementary Figure 2). The dose–response relationships between metformin use and total joint replacements were inconsistent.
Association of metformin dosage at points 3, 12 and 24 months after first prescription with incidence of total joint replacement
Compared with nonusers, the risk of total joint replacement was lower among those who had previously used metformin (adjusted HR 0.83, 95% CI 0.52–1.35) and continuing users (adjusted HR 0.72, 95% CI 0.48–1.09), after adjustment for covariates, at the 12-month mark, although this was not statistically significant. At the 24-month mark, compared with nonusers, continuing users of metformin had a significantly lower risk of total joint replacement (adjusted HR 0.72, 95% CI 0.57–0.92) and TKR (adjusted HR 0.75, 95% CI 0.58–0.96), after adjustment for covariates (Table 3).
Sensitivity analyses
The results of sensitivity analyses for risk of TKR or THR using inverse probability of treatment weighting and competing risk regression were consistent with the primary analysis (Table 2). Sensitivity analyses including only OA-related TKR or THR as the outcomes also generated similar results as the primary analysis (primary analysis: adjusted HR 0.66–0.70; sensitivity analysis: adjusted HR 0.67–0.81) (Appendix 1, Supplementary Table 1). The time-dependent Cox model generated similar results (THR or TKR: adjusted HR 0.81, 95% CI 0.70–0.95; TKR: adjusted HR 0.82, 95% CI 0.69–0.97; THR: adjusted HR 0.80, 95% CI 0.54–1.21) (Table 2). Further, the sensitivity analysis using the alternative outcome of the incidence of hospital admission for knee or hip OA showed that metformin users had significantly lower risks than nonusers (Appendix 1, Supplementary Table 4).
Interpretation
We conducted a large, population-based cohort study with a 14-year follow-up period, and found that, among patients with type 2 diabetes mellitus, metformin use was consistently associated with a reduced risk of TKR and THR compared with not using metformin. The findings were consistent using PTDM, PSM, inverse probability of treatment weighting and competing risk regression models. Sensitivity analyses using secondary outcomes such as OA-related total joint replacements and hospital admission for knee or hip OA generated similar results.
Other studies have evaluated the association between metformin use and OA in patients with diabetes. A cohort study using electronic health records reported no statistically significant association (adjusted HR 1.02, 95% CI 0.91–1.15) between metformin and occurrence of OA in patients with type 2 diabetes mellitus, but this study’s limitations included variability among clinicians in diagnosing and recording OA, no assessment of specific joint OA and no adjustment for some important covariates.22 Another retrospective cohort study reported that, among 968 patients who had OA and type 2 diabetes mellitus, those who were prescribed cyclooxygenase-2 (COX-2) inhibitors and metformin had lower rates of joint replacement than those treated with COX-2 inhibitors alone (adjusted HR 0.74, 95% CI 0.60–0.92).21 The authors did not control for immortal time bias, and potential confounding effects were not adjusted for using propensity-score weighting or matching. Most recently, a large retrospective study conducted among 93 330 patients with type 2 diabetes mellitus that used PSM found that regular metformin users had a 19% lower hazard of TKR than nonusers over their 4-year follow-up period, consistent with our findings.23
Diabetes and metabolic syndrome have been shown to be associated with OA.32,33 The biological mechanisms linking metformin and OA are yet to be clarified. Metformin could decrease the risk of total joint replacement among patients with diabetes mellitus by multiple mechanisms, including by reducing inflammation, by sustaining adenosine 5′-monophosphate–activated protein kinase activity in chondrocytes and by regulating metabolism. 16,34,35 Given our findings and those of a previous observational study,23 a randomized trial of the efficacy of metformin in patients with OA is indicated. Metformin has been studied in patients without diabetes mellitus and was found to be safe.36 Trials of metformin for the prevention of diabetes mellitus and for weight reduction among patients with obesity and without diabetes mellitus showed that doses of 1.7–2.5 g/d could be prescribed without increases in incidence of hypoglycemia or lactic acidosis.37,38
Limitations
Data on HbA1c levels were not available, so we could not adjust for degree of blood glucose control, although we did adjust for severity of diabetes mellitus using the adapted DCSI. Despite the multiple strategies that we employed to control for potential confounders, unmeasured confounding factors could yet bias the results. We conducted our study among a population with type 2 diabetes mellitus in Taiwan; generalizability of our findings to other populations and to patients with OA but without diabetes mellitus is unknown. Finally, our research data sets were collected before 2014, and oral antihyperglycemic agents that have been approved more recently were not used as frequently as they are now. However, metformin continues to be used as first-line treatment for type 2 diabetes mellitus in Taiwan and many other jurisdictions.36
Conclusion
We found that metformin use in patients with type 2 diabetes mellitus was associated with a significantly reduced risk of joint replacement, suggesting a potential therapeutic effect of metformin in patients with OA. Randomized controlled clinical trials are warranted to determine whether metformin is effective in patients with OA.
Acknowledgements
The authors thank all of the participants and staff who made this study possible, especially participants who have contributed data to the National Health Insurance Research Database. They also thank Dr. Danny Chan from the University of Hong Kong for commenting on the manuscript.
Footnotes
Competing interests: David Hunter receives consulting fees from Merck Serono, TLC Bio, Tissuegene, Lilly and Pfizer for scientific advisory roles related to osteoarthritis. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Zhaohua Zhu, Jing-Yang Huang, Guangfeng Ruan, Shibo Chen, David Hunter, James Wei and Changhai Ding conceived and designed the study. Jing-Yang Huang, Guangfeng Ruan, Peihua Cao, Yan Zhang, Weiyu Han, Tianyu Chen, Xiaoyan Cai, Jia Liu, Yujin Tang, Na Yu and Qian Wang managed and analyzed the data. All authors contributed to data interpretation. Zhaohua Zhu drafted the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work. Zhaohua Zhu, Jing-Yang Huang, Guangfeng Ruan, Peihua Cao and James Wei contributed equally to this work.
Funding: The study was supported by the National Natural Science Foundation of China (32000925, 81974342), Guangzhou Science and Technology Program (202002030481), Guangdong Basic and Applied Basic Research Foundation (2019A1515111169, 2019A1515110620) and Wu Jieping Medical Foundation Program (3206750.2020-03-12).
Data sharing: No data are available for sharing. Although we are unable to share the data sets, code lists are available on request to corresponding authors.
- Accepted October 28, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/