A six-year-old boy with autism spectrum disorder presented to the emergency department with a one-week history of left hip pain and limping. His associated symptoms included fever, rhinorrhea and cough. Bilateral hip and knee radiographs were normal, and he was discharged home with a diagnosis of transient synovitis.
The patient’s fever and infectious symptoms resolved, but the pain in his left hip worsened. He returned to the emergency department seven weeks later after he had been unable to bear weight for one week. There were no constitutional symptoms (e.g., fever, night sweats or weight loss). He did not have other joint involvement. He had one soft bowel movement daily, with no blood or mucus, and had no abdominal pain. There were no notable findings on review of systems other than the parents noting that he had “red dots” on his arms and legs. He was not on any medications, and was fully immunized.
Repeat radiographs of the patient’s hips showed periarticular osteopenia bilaterally, most marked in the right hip (Figure 1).
Anteroposterior radiograph of the hips of a six-year-old boy with autism spectrum disorder and left hip pain, showing bilateral osteopenia of the femoral heads, which is most marked in the right hip.
What diagnoses should be ruled out in this patient?
Slipped capital femoral epiphysis
Septic arthritis
Osteomyelitis
Malignant disease
All of the above
The answer is (e). Multiple conditions may cause hip pain in children, but it is essential to rule out serious conditions, such as those listed above. The radiograph did not show evidence of slipped capital femoral epiphysis, which typically occurs in older children. Legg–Calve–Perthes disease is also an important cause of hip pain, but the presentation is usually more insidious. There was also no avascular necrosis seen on the radiographs, although initial radiographs can appear normal.
Septic arthritis should always be considered in a patient with joint pain, although it is typically more acute in onset and less likely without fever. Osteomyelitis is also less probable in a patient who is afebrile. Malignant disease should be considered in any case of bony pain; about 25% of children with newly diagnosed leukemia present with a complaint of severe bone pain. The patient had no constitutional symptoms, but the parents described “red dots,” which could be indicative of thrombocytopenia in the setting of leukemia.
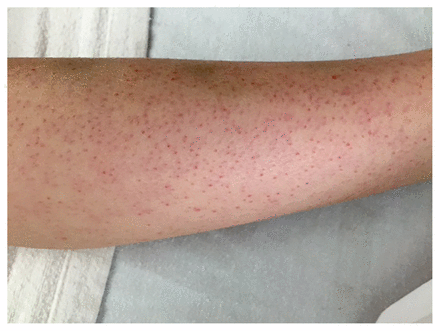
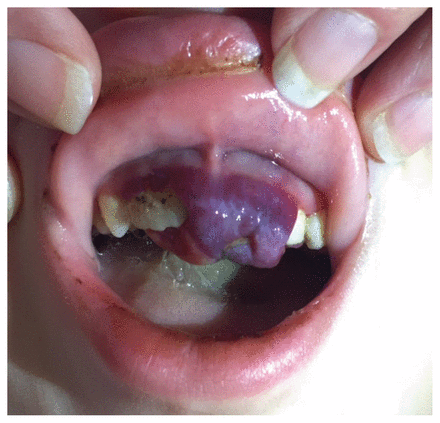
On physical examination, the patient’s weight was 21.2 kg (25th percentile) and his height was 121 cm (50th percentile). He was afebrile, and vital signs were within normal limits. He appeared pale and sat with his left hip flexed and externally rotated, with flexion of the left knee. He was unable to fully extend his left knee and had discomfort on internal rotation of his left hip. There was no evidence of erythema or warmth, and he had no discomfort on palpation. The range of motion of his right hip and knee were normal, and there were no joint effusions. Hepatosplenomegaly and lymphadenopathy were absent. Dermatologic examination showed palpable petechiae on his arms and legs bilaterally (Figure 2) and some ecchymoses on his legs. The patient had swollen, purple gingiva with blood-filled blisters (Figure 3), which bled spontaneously on examination. His parents reported that these lesions had begun about three months earlier.
Palpable petechiae on the patient’s arms and legs, shown here on his left leg.
Swollen, purple gingiva with blood-filled blisters.
Results of initial laboratory tests are shown in Box 1.
| Laboratory test | Value | Reference range* |
|---|---|---|
| Leukocyte count, × 109/L | 6.3 | 4.0–10.0 |
| Platelet count, × 109/L | 250 | 150–400 |
| Hemoglobin, g/L | 74 | 120–160 |
| Mean corpuscular volume, fL | 79 | 77–96 |
| Reticulocyte count, × 109/L | 153 (5.36%) | 10–100 |
| Blood smear | Moderate polychromasia | |
| Erythrocyte sedimentation rate, mm/h | 103 | 2–34 |
| C-reactive protein, mg/L | 31.1 | 0.1–1.0 |
| International normalized ratio | 1.0 | 0.80–1.20 |
| Partial thromboplastin time, s | 32.0 | 24.0–36.0 |
↵* Reference ranges for age.
What is the most likely diagnosis?
Leukemia
Henoch–Schönlein purpura
Bleeding disorder
Nonaccidental injury
Dietary deficiencies
The answer is (e). Given the patient’s normal leukocyte and platelet counts, and the absence of blasts on peripheral smear, leukemia was not likely. In addition, although the hemoglobin concentration was low, the reticulocyte count was appropriately elevated, which indicated adequate bone marrow function. Henoch–Schönlein purpura can cause both joint pain and palpable purpura without thrombocytopenia; however, the joint pain is typically more acute in onset. In addition, mucosal bleeding and anemia are not characteristic. A bleeding disorder would be less likely in the context of a normal platelet count, and normal international normalized ratio and partial thromboplastin time. Nonaccidental injury should be considered in any case of bony pain and unexplained bleeding, but it was not felt to be the cause in view of the other findings.
Dietary deficiencies should be considered in children presenting with anemia, gingival disease and osteopenia, especially those with autism spectrum disorder, who are more likely to have restrictive diets. A 24-hour dietary recall interview was conducted. We established the percentage of recommended daily intake of vitamins and minerals by looking at the specific brands purchased.
Our patient had a highly restrictive diet, with complete absence of vitamins C and D and only minimal iron intake (Box 2). He had not consumed fruits (other than a few apples) or vegetables in more than three years and was not taking vitamin supplements. He ate the same foods every day, which meant the 24-hour dietary recall reflected his entire diet. At the time of our examination, we learned that a dietitian had started working with the patient to reintroduce fruits and vegetables. Apples were the first fruit reintroduced, several months earlier, but the patient had stopped eating them because of gingival pain.
| Meal | Food | Vitamin/mineral; % RDI | ||
|---|---|---|---|---|
| Vitamin C | Vitamin D | Iron | ||
| Breakfast |
| 0 | 0 | 6 |
| Snack | Plain Greek yogurt (3 spoonfuls) | 0 | 0 | 0 |
| Lunch |
| 0 | 0 | 4 |
| Snack | Nacho cheese-flavoured tortilla chips (small pack) | 0 | 0 | 0 |
| Dinner |
| 0 | 0 | 10 |
| Snack | Microwave popcorn | 0 | 0 | 0 |
| Total | 0 | 0 | 20 | |
Note: RDI = recommended daily intake.
The combination of bony pain, perifollicular petechial rash, bleeding gums, anemia and no dietary vitamin C is classic of scurvy. To confirm the diagnosis, ascorbic acid measurement was done, as well as iron studies and measurement of vitamins B12 and D (Box 3). Zinc levels were not measured at the time of diagnosis.
| Laboratory test | Initial value | Value at 8-wk follow-up | Reference range* |
|---|---|---|---|
| Ascorbic acid, μmol/L | < 5 | NA | > 24 |
| Hemoglobin, g/L | 74 | 138 | 120–160 |
| Iron, μmol/L | 3 | 16 | 5–27 |
| Ferritin, μg/L | 110 | 140 | 14–80 |
| Transferrin, g/L | 2.19 | NA | 2.16–3.30 |
| Cyanocobalamin (vitamin B12), pmol/L | 360 | 425 | 220–1300 |
| Folate, nmol/L | NA | 41 | > 30 |
| 25-hydroxyvitamin D, nmol/L | 17 | 109 | 70–250 |
| Alkaline phosphatase, U/L | 92 | 138 | 151–342 |
| Erythrocyte sedimentation rate, mm/h | 103 | 30 | 2–34 |
Note: NA = not available.
↵* Reference ranges for age.
Although the patient’s dietary history suggested micronutrient deficiency secondary to restriction, deficiency from malabsorption was also considered. This was thought to be less likely considering the degree of dietary restriction and the absence of gastrointestinal symptoms. The decision was therefore made to start vitamin and mineral supplementation (250 mg of vitamin C and 800 IU of vitamin D once daily, 40 mg of elemental iron and 250 mg of calcium carbonate three times daily, and a multivitamin once daily) and to monitor the patient closely through follow-up appointments to determine the need for further investigations into malabsorption.
Within two weeks, the patient’s hip pain had resolved and his gingival bleeding had greatly improved. Results of repeat laboratory investigations done eight weeks later showed normalization of the erythrocyte sedimentation rate, hemoglobin concentration and vitamin D level (Box 3). Because of laboratory error, the repeat ascorbic acid level is not reported here. The rapid resolution of symptoms and normalization of laboratory values following vitamin and mineral supplementation further indicated that the micronutrient deficiencies were secondary to dietary restriction rather than to malabsorption. No further investigations were pursued.
Discussion
Although scurvy is thought of as a disease of the past, it still occurs today. The incidence of clinical scurvy in North America is unknown, but the overall prevalence of vitamin C deficiency in individuals more than six years old in the United States is 3.6%, based on data from the National Health and Nutrition Examination Survey.1
Vitamin C, or ascorbic acid, plays a vital role in the synthesis and cross-linking of collagen.2 Many of the clinical manifestations of scurvy can be attributed to capillary fragility, secondary to depletion of pericapillary collagen.3
The most common radiographic finding is osteopenia, 3 which is nonspecific, and in our case may have been magnified by concomitant vitamin D deficiency, in addition to zinc deficiency. Less common but more specific are radiographic findings that were not seen in our patient, including calcification zones with adjacent lucency in the distal metaphyses, known as the “scurvy zone,” which can be the site of fractures.3 Healing fractures in this zone can lead to spur formation, known as Pelkan spurs.3 In addition, the border of the epiphyses can be increased in density, known as a Wimberger ring.3 These findings are often unfamiliar to radiologists because of the rarity of scurvy.3
Restrictive diets are not uncommon in children with autism spectrum disorder. In a study comparing food selectivity in 111 children, Bandini and colleagues4 found that typically developing children refused 19% of foods offered, whereas those with autism spectrum disorder refused 42%. Hyman and colleagues5 found that reported intakes of vitamin A, vitamin C and zinc were lower among children four to eight years of age with autism spectrum disorder than among matched controls. In addition, although it is often thought that children outgrow their picky eating habits, the proportion of children with nutrient intake less than the estimated average requirement increased with age for certain nutrients, including vitamins A, C, E, B12 and folate, and the minerals zinc and magnesium.5
Scurvy has been reported in children with restrictive diets associated with varying degrees of developmental delay.3 It has also been reported in an infant fed exclusively almond beverages6 and in a child with end-stage renal disease whose loss of ascorbic acid was linked to peritoneal dialysis. 7 In adults, scurvy has been reported in patients with restrictive diets and eating disorders,8 patients with either suspected or perceived ascorbic acid allergy9 and patients with diseases causing malabsorption, such as Whipple disease10 and Crohn disease.8
Clinicians involved in the care of people with autism spectrum disorder should be prepared to counsel families regarding the risks of restrictive diets and the importance of vitamin supplementation when necessary. They should consider the possibility of insufficient dietary intake in these patients, look for signs and symptoms of nutritional deficiencies and refer to a dietitian for assessment if the patient appears to have a restrictive diet or signs and symptoms associated with nutritional deficiency.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
The authors have obtained patient consent.
Contributors: Both authors contributed equally to the work, drafted and critically revised the manuscript, approved the final version to be published and agreed to act as guarantors of the work.