Abstract
BACKGROUND: Inflammatory bowel disease (IBD) increases the risk of venous thromboembolism (VTE) by 2 to 3 times. We compared the reduction in risk of incident VTE associated with use of tumour necrosis factor-α (TNF-α) inhibitors versus nonbiologic immunomodulatory agents in patients with IBD.
METHODS: This observational cohort study used data from public (Medicaid, 2000–2010; Medicare, 2007–2013) and private (Optum Clinformatics, 2004–2013) health insurance programs in the United States. We included a total of 21 671 patients who had IBD without a prior diagnosis of cancer or VTE. The exposure of interest was treatment initiation with TNF-α inhibitor or nonbiologic (azathioprine, mercaptopurine, methotrexate, cyclosporine). The outcome of interest was admission to hospital with VTE as the principal diagnosis. We used Cox proportional hazard regression models to estimate hazard ratios (HRs) separately for each database after risk adjustment for more than 50 covariables using propensity score fine stratification. We used inverse variance meta-analytic methods to pool the adjusted HRs across the 3 databases.
RESULTS: We included a total of 5173 patients who started TNF-α inhibitor therapy (1439 in the Medicaid database, 1480 in Medicare and 2254 in Optum Clinformatics) and 16 498 who initiated a nonbiologic agent (5041 in Medicaid, 5166 in Medicare, 6291 in Optum Clinformatics). The adjusted pooled HR for VTE risk with TNF-α inhibitor versus a nonbiologic agent was 0.78 (95% confidence interval [CI] 0.60 to 1.02). The HR was lower in patients with Crohn disease (pooled HR 0.62, 95% CI 0.44 to 0.86) and younger patients (18–44 yr; pooled HR 0.55, 95% CI 0.34 to 0.87).
INTERPRETATION: We did not find a statistically significant association between risk of VTE and use of TNF-α inhibitors, relative to nonbiologics, in patients with IBD overall. However, an association was evident for patients younger than 45 years and those with Crohn disease.
Venous thromboembolism (VTE), which includes deep vein thrombosis and pulmonary embolism, has been identified as a major public health challenge in the United States owing to its common occurrence,1 high case-fatality rates2 and large annual economic burden (US$7 billion to US$10 billion).3 The incidence rate of VTE is between 1 and 1.5 per 1000 person-years in Western populations;4,5 however, the incidence of VTE is reported to be 2 to 3 times higher among patients with inflammatory bowel disease (IBD), including Crohn disease and ulcerative colitis, than among age- and sex-matched patients without IBD.6–8 This observation led to the recommendation in current practice guidelines to use VTE prophylaxis for patients with IBD who have been admitted to hospital.9,10
Although the precise mechanism for increased VTE risk in IBD is unknown, a leading hypothesis is that the chronic activity of inflammatory pathways in patients with IBD may trigger procoagulant states, resulting in a heightened risk of clot formation. 11 Tumour necrosis factor-α (TNF-α), a pro-inflammatory cytokine involved in the pathogenesis of IBD, activates coagulation pathways.12 Blockade of TNF-α with monoclonal antibodies has been shown to reduce markers of coagulation and fibrinolysis and to lead to reduced clot formation and normalization of clot lysis profiles.13,14 However, it is not known whether use of TNF-α inhibitors reduces major VTE events in patients with IBD to a greater extent than use of other (nonbiologic) immunomodulatory treatments.
Therefore, we compared the risk of incident VTE in patients with IBD who initiated treatment with a TNF-α inhibitor or a nonbiologic agent, using data from 3 large health insurance databases in the US. We hypothesized that the risk of VTE in these patients would be lower with the use of TNF-α inhibitors than with nonbiologic agents.
Methods
Data sources and study design
We conducted an observational cohort study using health insurance claims data from Medicaid (2000–2010), Optum Clinformatics (2004–2013) and Medicare (parts A, B and D; 2007–2013) databases. These data sources contain comprehensive longitudinal information on patient demographic characteristics, coded inpatient and outpatient diagnoses and procedures, and outpatient prescription dispensings for their enrollees.15
Study cohorts
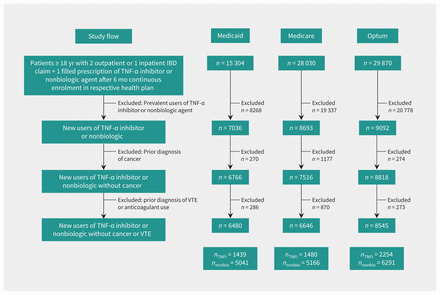
We identified patients with IBD from each of the 3 data sources after 180 days of continuous enrolment, based on at least 2 outpatient diagnoses or 1 inpatient diagnosis of Crohn disease (International Classification of Diseases, 9th Revision [ICD-9] code 555.xx) or ulcerative colitis (ICD-9 code 556.xx) and at least 1 filled prescription for either a TNF-α inhibitor (infliximab, adalimumab, certolizumab or golimumab) or a nonbiologic agent (mercaptopurine, azathioprine, methotrexate or cyclosporine) within 365 days after diagnosis of IBD.16 We did not include patients taking steroids in the nonbiologic group, because steroid treatment is more likely to be used over the short term to induce remission in patients with actively flaring IBD, whereas other nonbiologics are more likely to be used throughout the course of IBD, similar to the use of TNF-α inhibitors. We included only patients who filled a new prescription for TNF-α inhibitor or a nonbiologic agent after 180 days of not using either class of medication.17 The date of filling this new prescription was defined as the index date. We excluded patients who started an agent from both exposure groups on the same index date, to prevent mixing of exposure effects on the risk of VTE. We further required that patients identified as having IBD have no recorded cancer or VTE diagnosis any time before the index date and no anticoagulant use in the 180-day period preceding the index date, to allow us to focus on IBD-related incident VTE events. Appendix 1 (available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161485/-/DC1) summarizes the study design and provides the number of patients who initiated each study drug.
Drug exposure and follow-up
Patients were classified as having initiated a TNF-α inhibitor or a nonbiologic agent on the basis of the index prescription. The follow-up for VTE began on the day after the index date. For the primary analysis, we used an “as-treated” follow-up approach, whereby follow-up was stopped for patients who either filled a prescription for a drug in the other exposure group or discontinued the index exposure. Discontinuation was defined as no record of a subsequent prescription of the index medication class for 3 successive months, after accounting for the number of days of exposure provided by the most recent prescription. As a secondary approach, we conducted an intention-to-treat analysis in which patients were followed in their index exposure group, regardless of treatment change or discontinuation. With both the as-treated and intention-to-treat approaches, we truncated follow-up at the first of outcome occurrence, health insurance disenrolment, death or the most recent date of data availability. The intention-to-treat analysis was considered a secondary follow-up approach, because it is prone to exposed person-time misclassification in observational studies of patients receiving routine care, for whom treatment discontinuations are frequent.18
Outcome measurement
The outcome of VTE was defined as a composite end point of incident pulmonary embolism or deep vein thrombosis recorded on hospitalization claims as the principal discharge diagnosis. In a secondary approach, we defined VTE based on primary diagnosis codes from inpatient or outpatient claims as also including mild cases. The ICD-9 codes used to identify VTE (summarized in Appendix 2, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161485/-/DC1) have had high positive predictive values in administrative claims databases (in the range of 65% to 95%).19 We used a probabilistic sensitivity analysis to evaluate the impact of potential misclassification of the outcomes on our results20 (described in detail in Appendix 3, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161485/-/DC1).
Covariables
We identified a large number of covariables, which were measured in the 180-day baseline period preceding each patient’s index date, unless otherwise specified. These consisted of demographic characteristics (age, sex, geographic region and race [not available in the Optum Clinformatics database]), variables related to IBD severity, other VTE risk factors, comorbidities, concomitant use of other drugs and markers of intensity of health care use. Table 1 contains the full list of covariables considered in this analysis. Because we did not have direct information available regarding IBD activity, such as the Crohn’s Disease Activity Index or Simple Clinical Colitis Activity Index (for ulcerative colitis), we undertook a sensitivity analysis to evaluate the effect of unmeasured confounding on our reported estimates21 (described in detail in Appendix 4, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161485/-/DC1).
Baseline characteristics (crude data)* of patients with IBD initiating TNF-α inhibitor or nonbiologic agent
Statistical analysis
We stratified all of the statistical analyses by data source. We report crude incidence rates of VTE for both exposure groups, crude differences in incidence rate and crude incidence rate ratios, along with 95% confidence intervals (CIs), comparing the unadjusted rates of VTE in the TNF-α inhibitor group and the nonbiologic group.
We used propensity score methods to account for potential confounding. Propensity scores, which were defined as the predicted probability of receiving a TNF-α inhibitor conditional upon patients’ covariable constellations, were calculated separately for each data source using multivariable logistic regression models that included the covariables described above and the calendar year of the index date as independent variables. We trimmed the non-overlapping portions of the propensity score distributions to exclude noncomparable patients and created 50 propensity score strata, based on the distribution of propensity scores in the exposed patients.22 We then weighted unexposed patients proportional to the distribution of exposed patients in the stratum into which they fell. Balance achieved after weighting was shown with standardized differences.23 We used weighted Cox proportional hazard models to estimate the adjusted hazard ratios (HRs) between TNF-α inhibitor use and the risk of VTE. We also determined adjusted risk differences between the 2 treatment groups at 12 months,24 along with 95% CIs constructed using nonparametric bootstrap sampling with 500 samples. The proportional hazards assumption was tested using Schoenfeld residuals for the treatment variable.25 All analyses were conducted in SAS software, version 9.4 (SAS Institute).
After conducting individual patient-level analysis in each database, we pooled the adjusted HRs for TNF-α inhibitors versus nonbiologics across the 3 data sources using inverse variance-weighted random-effects meta-analytic methods (with Cochrane Manager R [RevMan] software) to provide a summary estimate. As a sensitivity analysis, we also determined pooled estimates after excluding 1 data source at a time, to rule out the possibility of estimates from 1 of the 3 data sets highly influencing the pooled estimates.
Subgroup analysis
We defined several subgroups of interest: patients with active IBD (defined as ongoing use of steroids or a hospital admission during the baseline period with IBD as the principal diagnosis),6 2 strata by IBD subtype (Crohn disease and ulcerative colitis) and 3 strata defined by patient age (18–44 yr, 45–64 yr, ≥ 65 yr).26 To account for confounding in these subgroup analyses, we used fine stratification based on the propensity score, with models built separately within each subgroup in each database.
Ethics approval
The use of the de-identified databases described above was approved by the Institutional Review Board of Brigham and Women’s Hospital.
Results
Study cohort selection and patient characteristics
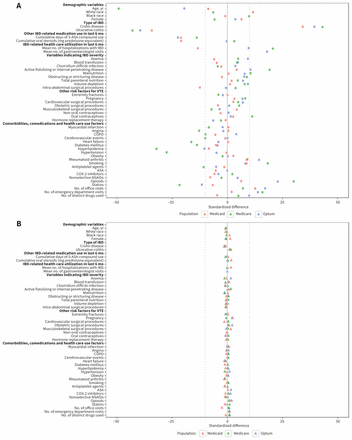
Across the 3 data sources, we identified a total of 21 671 patients with IBD who initiated treatment with either a TNF-α inhibitor (5173 or 23.9%) or a nonbiologic agent (16 498 or 76.1%) who did not have a prior cancer diagnosis, VTE diagnosis or anticoagulant use in the baseline period (Figure 1). In all 3 data sources, a majority of the patients were women, with the Optum Clinformatics cohort having a lower proportion of women than the Medicare and Medicaid cohorts (4450/8545 [52.1%] v. 4306/6646 [64.8%] and 4334/6480 [66.9%], respectively). The average age was lowest in the Medicaid cohort (33.6 yr, standard deviation [SD] 12.7 yr) and highest in the Medicare cohort (59.5 [SD 15.2] yr). The proportion of patients with a diagnosis of Crohn disease was higher in the Medicaid cohort than in the Medicare and Optum Clinformatics cohorts (5012/6480 [77.3%] v. 4112/6646 [61.9%] and 5363/8545 [62.8%], respectively). There were some appreciable differences in the characteristics of the 2 treatment groups among the 3 data sources (Table 1 and Appendix 5 [available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161485/-/DC1]). For example, the diagnosis of ulcerative colitis, the use of acetylsalicylic acid medications and mean age were higher among patients initiating nonbiologic agents, whereas the diagnosis of Crohn disease was more common among those initiating TNF-α inhibitor. The propensity score stratification and weighting approach rendered the 2 treatment groups comparable by eliminating imbalances in all of the measured covariables (see Figure 2 for standardized differences before and after propensity score weighting; the propensity score distributions are shown in Appendices 6–8, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161485/-/DC1).
Flow chart for patient selection. Note: IBD = inflammatory bowel disease, TNF = tumour necrosis factor, TNFi = tumour necrosis factor-α inhibitor, VTE = venous thromboembolism.
Balance in baseline characteristics before (A) and after (B) propensity score weighting among patients with inflammatory bowel disease (IBD) initiating tumour necrosis factor-α inhibitor or nonbiologic agent. The dotted vertical lines mark standardized differences of −10 or +10, which were suggested by Austin23 as boundaries when evaluating balance. Note: ASA = acetylsalicylic acid, COPD = chronic obstructive pulmonary disease, COX = cyclo-oxygenase, NSAID = nonsteroidal anti-inflammatory drug, VTE = venous thromboembolism.
Risk of VTE
Crude incidence rates (reported per 100 person-years) for VTE were similar for the 2 treatment groups in the Medicare and Optum Clinformatics cohorts (2.52 [95% CI 1.86 to 3.36] and 2.58 [95% CI 2.19 to 3.04] in the Medicare cohort and 0.66 [95% CI 0.37 to 1.10] and 0.70 [95% CI 0.51 to 0.95] in the Optum Clinformatics cohort for TNF-α inhibitor and nonbiologic groups, respectively), whereas in the Medicaid cohort, the rate was somewhat lower for the TNF-α inhibitor group (0.79 [95% CI 0.34 to 1.55] and 1.31 [95% CI 0.98 to 1.72], respectively) (Table 2).
Crude estimates for absolute and relative measures of VTE incidence in patients with inflammatory bowel disease who started treatment with a TNF-α inhibitor or nonbiologic agent
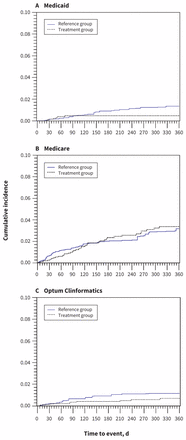
Figure 3 shows the cumulative incidence of VTE in the 2 treatment groups adjusted for baseline covariables using propensity score weighting. At 1 year, the adjusted risk differences between TNF-α inhibitor and nonbiologic groups were −0.93% (95% CI −1.77% to −0.25%) in the Medicaid cohort, −0.09% (95% CI −1.31% to 1.72%) in the Medicare cohort and −0.51% (95% CI −1.42% to 0.20%) in the Optum Clinformatics cohort.
Cumulative incidence of venous thromboembolism among patients with inflammatory bowel disease initiating tumour necrosis factor (TNF)-α inhibitor or nonbiologic agent, adjusted for baseline characteristics through propensity score weighting. Patients initiating a TNF-α inhibitor constituted the treatment group; those initiating a nonbiologic agent constituted the reference group.
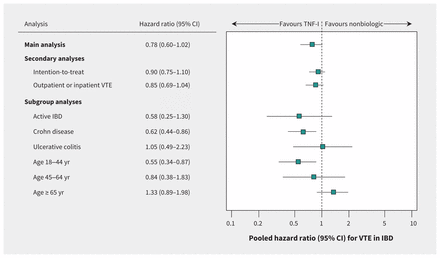
The adjusted HRs for use of TNF-α inhibitor versus nonbiologic agent were 0.58 (95% CI 0.27 to 1.23) in the Medicaid cohort, 0.87 (0.63 to 1.22) in the Medicare cohort and 0.66 (95% CI 0.37 to 1.17) in the Optum Clinformatics cohort, with a pooled HR of 0.78 (95% CI 0.60 to 1.02) (Figure 4). Although the p values for the correlation between Schoenfeld residuals and ranked event times did not indicate violations of the proportional hazards assumption, the cumulative incidence plots (Figure 3) indicated that hazards of VTE for the 2 treatment groups crossed for the Medicaid and Medicare cohorts early during follow-up (at 3.5 and 5 months, respectively). Therefore, we used an extended Cox model with a Heaviside function to provide separate HRs in the early and late follow-up periods. The pooled HRs from this analysis were less precisely estimated but were qualitatively similar to the main analysis (0.85 [95% CI 0.59 to 1.21] and 0.73 [95% CI 0.47 to 1.15] for the early and late periods, respectively; Appendix 9, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161485/-/DC1). The pooled HR was 0.90 (95% CI 0.75 to 1.10) for the intention-to-treat follow-up approach and 0.85 (95% CI 0.69 to 1.04) for VTE defined on the basis of inpatient or outpatient diagnosis (Figure 5).
Comparative risk of incident venous thromboembolism between patients with inflammatory bowel disease initiating tumour necrosis factor-α inhibitor (TNF-I) and those initiating a nonbiologic agent. Note: CI = confidence interval.
Secondary and subgroup analyses for the comparative risk of incident venous thromboembolism (VTE) among patients with inflammatory bowel disease (IBD) initiating tumour necrosis factor-α inhibitor (TNF-I) or nonbiologic agent. Hazard ratios for all subgroups were pooled after individual analysis of the Medicaid, Medicare and Optum Clinformatics cohorts, except for the subgroup analysis involving patients 65 years of age or older; for that analysis, only the Medicare cohort was included because the numbers of cases in the Medicaid and Optum Clinformatics cohorts were insufficient to conduct comparisons. Note: CI = confidence interval.
Subgroup analyses
In the subgroups of patients with Crohn disease and younger age (18–44 yr), we found statistically significant reductions in VTE risk with TNF-α inhibitor treatment (pooled HR 0.62 [95% CI 0.44 to 0.86] and 0.55 [95% CI 0.34 to 0.87], respectively). For other subgroups, the differences were not statistically significant (Figure 5).
Sensitivity analyses
The corrected relative risk estimates from the sensitivity analysis evaluating the impact of outcome misclassification were in the range of 0.53 to 0.61, suggesting that our main analysis may have underestimated the true effect of TNF-α inhibitor use on VTE risk (Appendix 3). In a second set of sensitivity analyses, we observed that unmeasured confounding by disease activity could have altered our findings only if the proportion of patients with severe disease was more than 2-fold higher in the reference group (Appendix 4). Finally, we observed consistency in the direction of all reported pooled estimates, regardless of the data sources combined, which indicates a low potential that any data source had undue influence in our pooled estimates (Appendix 10, available at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.161485/-/DC1).
Interpretation
VTE has emerged as an important comorbid complication in patients with IBD.6–8 In this large observational study of more than 21 000 patients with IBD, we did not find a statistically significant association between risk of VTE and use of TNF-α inhibitors relative to nonbiologics in patients with IBD overall. However, the association between TNF-α inhibitors and reduced VTE risk was statistically significant for patients with Crohn disease and for younger patients.
Our findings are consistent with prior hypothesis-generating studies that reported a potential association between use of TNF-α inhibitors and reduced VTE risk.14,27,28 Our data support the notion that more aggressive control of inflammation in IBD by means of TNF-α inhibition may lead to reductions in coagulation and fibrinolysis, which ultimately could reduce the risk of VTE. The observed protective association between TNF-α inhibitor use and VTE was strongest for young patients, which suggests that when IBD is the predominant risk factor for VTE, aggressive inflammation control with TNF-α inhibitors may lead to clinically significant VTE risk reduction.
Our study has important clinical implications. Increased VTE incidence is a major cause of concern for patients with IBD, because it has been associated with an approximately 50% increase in length of hospital stay, an increase in hospitalization costs of nearly US$25 000 and a 2.1-fold increase in inhospital mortality.29 Although anticoagulant prophylaxis, as recommended by clinical guidelines, should be a major emphasis during hospital admissions for IBD,9,10 these data suggest that more aggressive inflammation control with TNF-α inhibitors in ambulatory patients has a potential role in reducing the IBD-attributable VTE risk. However, the risk–benefit ratio for TNF-α inhibitor treatment could vary depending upon the clinical scenario. For instance, in young patients with IBD who are at higher VTE risk because of very high disease activity, TNF-α inhibitors may clearly be preferable over nonbiologic agents, whereas in equivalent older patients, the lack of convincing benefit in terms of VTE risk, combined with a higher risk of serious adverse events such as serious infections,30 could shift the ratio toward risk over benefits. Moreover, drug selection in IBD is complex and depends on many factors in addition to VTE risk, most importantly, the expected effectiveness in controlling active disease and the risk of important adverse events, such as infections and malignancies, in certain patients. Although the potentially lower VTE risk that we observed in younger patients and patients with Crohn disease is an important clinical finding, eventual treatment decisions should be based on a multitude of patient-specific factors.
Strengths and limitations
Our study had several notable strengths. First, we accounted for various confounding factors through sound study design and rigorous statistical analysis. Second, owing to a large sample size and inclusion of patients from a wide age range, we were able to separately evaluate the effect of TNF-α inhibitor use on VTE risk in different age groups and to provide important evidence of heterogeneous treatment effect by age. Finally, we conducted a wide range of sensitivity analyses to evaluate the robustness of our findings.
The potential for residual confounding by indication is an important limitation of this study, as we were unable to account for IBD disease activity, body mass index or physical activity. However, we adjusted for a large number of IBD-related covariables as proxy variables for disease severity, as well as markers of health care utilization, and evaluated the impact of unmeasured confounding by disease activity on our estimates in a sensitivity analysis.
Conclusion
This large nationwide observational study of patients with IBD provides preliminary evidence of a potential association between treatment with TNF- αinhibitor and reduction in VTE risk among patients younger than 45 years and patients with Crohn disease. Although future research confirming this finding is recommended, this study adds to a growing body of evidence that optimizing medical management of IBD for better disease control could lead to reductions in the comorbidities associated with IBD and ultimately improve patients’ quality of life.
Footnotes
Competing interests: Rishi Desai is the principal investigator for a research grant from Merck to the Brigham and Women’s Hospital for unrelated work. Joshua Gagne is the principal investigator for a research grant from Novartis Pharmaceuticals Corporation to the Brigham and Women’s Hospital for unrelated work. He is also a consultant to Aetion, Inc., and Optum, Inc. Seoyoung Kim receives research grants from AstraZeneca, Eli Lilly, Pfizer, Bristol–Myers Squibb and Genentech to the Brigham and Women’s Hospital for unrelated studies. No other competing interests were declared.
This article has been peer reviewed.
Contributors: Rishi Desai, Joshua Gagne, Sonia Friedman and Seoyoung Kim contributed to study concept and design. Rishi Desai, Joyce Lii and Jun Liu contributed to analysis of the data. Rishi Desai had full access to all of the data in the study. All of the authors contributed to interpretation of the data, drafted the manuscript and revised it for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study was supported through internal sources of the Division of Pharmacoepidemiology and Pharmacoeconomics at Brigham and Women’s Hospital.
- Accepted July 13, 2017.