Abstract
Background: Shortfalls in deceased organ donation lead to shortages of solid organs available for transplantation. We assessed rates of deceased organ donation and compared hospitals that had clinical services for transplant recipients (transplant hospitals) to those that did not (general hospitals).
Methods: We conducted a population-based cohort analysis involving patients who died from traumatic brain injury, subarachnoid hemorrhage, intracerebral hemorrhage or other catastrophic neurologic conditions in Ontario, Canada, between Apr. 1, 1994, and Mar. 31, 2011. We distinguished between acute care hospitals with and without transplant services. The primary outcome was actual organ donation determined through the physician database for organ procurement procedures.
Results: Overall, 87 129 patients died from catastrophic neurologic conditions during the study period, of whom 1930 became actual donors. Our primary analysis excluded patients from small hospitals, reducing the total to 79 746 patients, of whom 1898 became actual donors. Patients who died in transplant hospitals had a distribution of demographic characteristics similar to that of patients who died in other large general hospitals. Transplant hospitals had an actual donor rate per 100 deaths that was about 4 times the donor rate at large general hospitals (5.0 v. 1.4, p < 0.001). The relative reduction in donations at general hospitals was accentuated among older patients, persisted among patients who were the most eligible candidates and amounted to about 121 fewer actual donors per year (adjusted odds ratio 0.58, 95% confidence interval 0.36–0.92). Hospital volumes were only weakly correlated with actual organ donation rates.
Interpretation: Optimizing organ donation requires greater attention to large general hospitals. These hospitals account for most of the potential donors and missed opportunities for deceased organ donation.
Many eligible patients never receive an organ transplant because of biological or psychological human factors that contribute to shortfalls in organ availability.1,2 Immune suppression and surgical complications, for example, require that transplant recipients receive centralized care at hospitals staffed by highly specialized clinicians.3,4 However, the subtleties of patient preferences, community support and maximizing available organs require that donors be identified from a wide range of general hospitals.2 Ultimately, these practices can mean that hospitals responsible for identifying potential donors do not always encounter the patients who received the successful transplantations.
A request for organ donation after death requires initiative, appropriate referral, consent, formal declaration of brain death and maintenance of donor viability at a time when other patients may also require attention.5,6 The time pressure is substantial, because incidents often arise on weekends or at night when clinicians are short staffed, sleep deprived and uncertain about diagnosing brain death.7,8 The diversity of cultural preferences and distraught emotions in relatives of potential donors can also cause well-intentioned requests to be misunderstood and followed by negative conflict.9,10 Hence, the ongoing rates of deceased organ donation represent substantial efforts both in the community and in hospitals.11
Decision science research suggests that clinical behaviour does not always follow the standard model of rational thought.12 Self-identity, for example, can sometimes influence decisions by shaping people’s preferences and defining institutional norms.13–15 We questioned whether the difference between hospitals that observe gratifying recipient outcomes and hospitals that initiate deceased organ donation might potentially contribute to lower donation rates owing to the lost transmission of information and the attitudes of hospitals that do not have transplant programs. To test this question, we assessed deceased organ donation rates and compared hospitals that have clinical services for transplant recipients to hospitals that do not have such services.
Methods
Patients
We identified all consecutive patients (children and adults) who died in any Ontario hospital between Apr. 1, 1994, and Mar. 31, 2011, from conditions associated with catastrophic brain injury. We chose this time frame because data were available for all hospitals, the coding system remained mostly unchanged (with the exception of one revision in 2002), and none of the hospitals changed their transplantation designation during this period. We excluded patients who did not have a valid health card identifier. The study was approved by the Sunnybrook Research Ethics Board, including a waiver of individual consent.
Selected causes of death
The patient’s cause of death was classified using the International Classification of Disease (ICD) codes (versions 9 and 10, as appropriate). We included 3 specific conditions that were common causes of brain death, consistently documented in available records and established in previous research.16–18 Traumatic brain injury was defined as cerebral laceration, contusion or other intracranial hemorrhage after trauma (ICD codes 800, 801, 804, 851–853 and S06–S09). Subarachnoid hemorrhage included events related to aneurysms or vascular malformations (ICD codes 430 and I60). Intracerebral hemorrhage included rupture of any cerebral blood vessel or unspecified nontraumatic hemorrhage (ICD codes 431, 432, I61 and I62).
Additional causes of death
We included 5 additional neurologic causes of death to provide a comprehensive assessment of potential donors: anoxic brain damage (ICD codes 348 and G931), cerebral edema (ICD codes 348 and G936), cerebral infarction (ICD codes 434 and I639), cerebral thrombosis (ICD codes 434, 436, I136, I138, I630–I635, I639 and I640) and asphyxiation (ICD codes 994 and T71).
Patient characteristics
We obtained patient age at death, sex and residence (urban or rural) through the official vital statistics registry.19 We derived patient socioeconomic status using the Statistics Canada algorithm.20 We determined previous admissions to hospital and outpatient visits in the year before death using linked databases validated in previous research (i.e., the Canadian Institute for Health Information hospital inpatient database and the Ontario Health Insurance Plan medical outpatient database).21,22 We determined comorbid conditions by any physician diagnosis in the year before death.23 We addressed referral patterns by classifying mode of hospital arrival into 3 groups: direct from the community, by ambulance or transferred from another hospital. The databases did not contain information on patient preferences, religion, race, ethnicity, vital signs, mental status, medications or formal declaration of brain death.
Hospital characteristics
We focused our attention on where the patient died and distinguished between hospitals that averaged more than 20 deaths per year (“larger hospitals”) and those that averaged fewer than 20 deaths per year (“smaller hospitals”). Larger hospitals were further divided into those that had clinical services for transplant recipients (“transplant hospitals”) and those that did not (“general hospitals”). Without exception, transplant hospitals announced transplantation services prominently as part of their identity, whereas general hospitals made no mention of such services on public websites or in community services. Otherwise, the hospitals operated under the same regulations, offered similar emergency services and obtained funding through identical fee structures.
Actual organ donation
We determined solid organ donation through the population-based physician billing database using a comprehensive set of billing codes for any combination of heart, lung, kidney, liver, pancreas or bowel procurement (codes E753, G347, G348, G411, M157, R872, S196, S201, S265, S274, S302 and S436). Some surgeons received funding from alternate payment programs and did not always use such codes; however, anesthesiologists and other clinicians without alternate funding used such codes in all relevant cases. These billing codes are specific (100%, 95% confidence interval [CI] 99%–100%), but not fully sensitive (75%, 95% CI 73%–78%) when validated through the Trillium Gift of Life Network registry (A.H. Li, Western University, unpublished data).
Subgroup of eligible candidates
We identified a subgroup of patients who would be the most eligible candidates for organ procurement based on a combination of demographic and clinical data. We used the data from this subgroup to check the robustness of our primary analysis after confining the sample based on objective criteria; specifically, patients with a diagnosis of any of the 3 specific conditions that were common causes of brain death, patients aged 59 years or younger, patients who received mechanical ventilation on their first day in hospital, patients who did not have a disqualifying condition (HIV, tuberculosis or cancer) and patients who survived for no longer than 1 week in hospital.
Control procedure
We examined cornea donation as another type of donation that follows a different procurement procedure. In contrast to organ donation, cornea donation (code E108) is subject to much less time pressure and can be scheduled hours after death. The person initiating the request for cornea donation tends to be affiliated with a central agency in Ontario and is not directly attached to the particular hospital involved. Finally, the request for cornea donation usually occurs when a donor is no longer receiving life-sustaining therapies and all physiology has ended.
Statistical analysis
Our primary analysis evaluated rates of actual organ donation among patients who were potential donors and compared transplant hospitals to general hospitals. We used hierarchical logistic regression to account for patient clustering in hospitals of different sizes. Our secondary multivariable analyses accounted for demographic factors including patient age, sex, residence, socioeconomic status and time of death (year, season, day). We performed additional stratified analyses to repeat the primary comparison using only data from the subgroup of patients who would be most eligible for donation. We conducted all analyses using the privacy safeguards of the Institute for Clinical Evaluative Sciences.
Results
We identified a total of 87 129 patients who died during the study period, of whom 1930 were identified as actual organ donors, (i.e., about 9 donors per million population annually). The number of actual donors increased with time so that the number of donors in the final year of the study was about twice that of the first year (Figure 1). The median and upper quartile of age of actual organ donors also increased with time, with an increase in median age of about 11 years (40 v. 51 yr) and an increase in upper quartile age of about 10 years (51 v. 61 yr). The 87 129 deaths were distributed across 140 hospitals, of which a total of 84 hospitals averaged fewer than 20 deaths annually. After excluding these small general hospitals, we obtained a sample of 56 hospitals (40%), 79 746 deaths (92%) and 1898 (98%) actual organ donors.
(A) Number of deceased donors and (B) number deaths from catastrophic neurologic conditions during the study period. An anomaly in the numbers of catastrophic neurologic deaths between 2001 and 2002 denotes a change from International Classification of Diseases (ICD), 9th revision, to the ICD 10th revision. An increase in donors over time with a persistent gap between the number of patients who died from catastrophic neurologic conditions and those who became donors can be seen.
A total of 22 515 patients died at transplant hospitals and the remaining 57 231 died at large general hospitals. Those who died in transplant hospitals had a distribution of demographic characteristics similar to that of patients who died at large general hospitals (Table 1). Patients who died generally lived in an urban area, had diverse characteristics and had a diagnosis of intracerebral hemorrhage or some other catastrophic neurologic condition. The main differences between the 2 settings were that patients who died in large general hospitals tended to be older and more likely to have a diagnosis of a miscellaneous catastrophic neurologic condition than those who died at a transplant hospital. Socioeconomic status was well balanced, albeit with a lower representation of people with high socioeconomic status in both groups.
Characteristics of patients included in the study
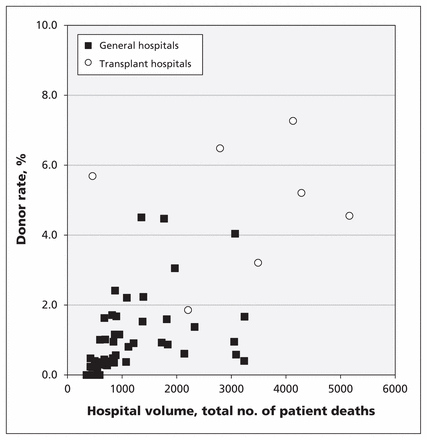
Overall, 1898 of the 79 746 patients included in our primary analysis became donors, for a procurement rate of about 1 in 40. Transplant hospitals accounted for 1118 donors and 22 515 deaths, for a procurement rate of 5.0 per 100 deaths. Large general hospitals accounted for 780 donors and 57 231 deaths, for a procurement rate of 1.4 per 100 deaths (Figures 2 and 3). This difference between hospitals was equal to a relative reduction in donation rates of about 74% (odds ratio [OR] 0.26, 95% confidence interval [CI] 0.24–0.29), was evident in both the first and second halves of the dataset (before and after Jan. 1, 2003) and amounted to an absolute reduction of about 121 actual organ donors each year in large general hospitals. Comparisons based on data from all hospitals (including small general hospitals) showed a relative reduction of 76% (OR 0.24, 95% CI 0.22–0.27). Restricting analyses to the 13 largest hospitals (> 2000 total deaths) showed a relative reduction of 73% (OR 0.27, 95% CI 0.24–0.31).
Rates of organ donation, by hospital, among patients who died from catastrophic neurologic conditions and the overall rate of deceased organ donation, showing a modest correlation of total deaths with overall donation rates (r = 0.61, p < 0.001).
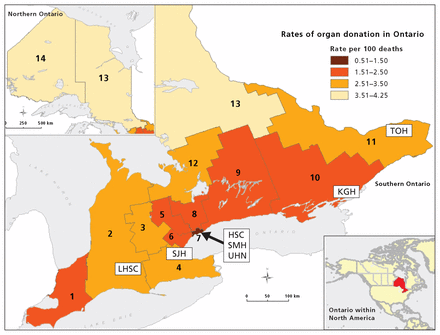
Organ donation by geographic area in Ontario. The 14 areas represent official Local Health Integration Networks. Note: HSC = Hospital for Sick Children, Toronto; KGH = Kingston General Hospital, Kingston; LHSC = London Health Sciences Centre, London; TOH = The Ottawa Hospital, Ottawa; SJH = St. Joseph’s Healthcare, Hamilton; SMH = St. Michael’s Hospital, Toronto; UHN = University Health Network, Toronto.
We identified patients who would be the most eligible donors by determining those with a diagnosis of traumatic brain injury, subarachnoid hemorrhage or intracerebral hemorrhage and who were younger than 59 years, were free of disqualifying clinical conditions, received mechanical ventilation starting on their first day in hospital and survived in hospital no longer than 1 week. A total of 3961 patients met these criteria, of whom 1035 were actual donors, for an overall procurement rate of about 1 in 4. Transplant hospitals accounted for 652 donors and 2205 deaths (procurement rate 29.6/100 deaths), large general hospitals accounted for 383 donors and 1756 deaths (procurement rate of 21.8/100 deaths), for a relative reduction in donation rates of about 34% (OR 0.66, 95% CI 0.57–0.77).
Basic patient characteristics were additional predictors of actual organ donation. Patients aged 59 years or younger were about 10 times more likely to be actual donors than patients aged 60 years or older (Table 2). Patients with lower socioeconomic status were about 30% less likely to be actual donors than patients with higher socioeconomic status. Cause of death was a good predictor of organ donation, with the highest rates of donation among patients who died after subarachnoid hemorrhage. Accounting for all predictors yielded a relative reduction in donation rates of about 42% (OR 0.58, 95% CI 0.36–0.92) at large general hospitals compared with transplant hospitals.
Predictors of organ procurement in 56 hospitals in Ontario
Our control analysis showed no large differences in cornea donation when comparing different hospitals. Overall, 2104 of the 79 746 patients who died became cornea donors. Transplant hospitals accounted for 688 cornea donors (procurement rate 3.1/100 deaths); large general hospitals accounted for 1416 cornea donors (procurement rate 2.5/100 deaths). This difference was equal to a relative reduction in donation rates of about 20% (95% CI 12%–27%). The subgroup analyses restricted to the 3961 patients who were the most eligible donors showed a procurement rate of about 1 in 8 and a 14% relative (but nonsignificant) increase in cornea donation (OR 1.14, 95% CI 0.94–1.37) at large general hospitals compared with transplant hospitals.
Interpretation
In this population-based cohort study, rates of organ donation in Ontario were low, showed increases over time and were highest among patients less than 45 years of age with higher socioeconomic status. An additional predictor of organ donation was not a characteristic of the patient, but of the hospital; namely, whether the hospital itself had clinical services for transplant recipients. The apparent decrease in donations in otherwise large general hospitals was difficult to attribute to chance, persistent over time and not fully explained by patient characteristics. Together, these data underscore the effect of circumstances, rather than personal preferences, on profound medical decisions.24
Actual organ donation is linked to several personal factors including the patient’s ethnicity and religious beliefs.25,26 Interactions between clinicians and substitute decision-makers can also influence consent rates for organ donation.9–11 Hospital factors associated with actual donation include large size, the availability of neurosurgical services, the presence of an emergency department and various unmeasured attributes.2,27 The main limitations of previous studies were small sample, the use of self-report surveys or limited time spans. One previous study suggested that hospital identity might influence organ donation rates based on chart review at a subset of volunteer hospitals and was restricted to patients who were formally documented as having brain death.2
Limitations
Hospital identity is a reflection of multiple attributes, including the availability of specialty services such as neurosurgery, neurology and trauma. Large general hospitals, therefore, might differ so that some could have higher than average procurement rates and, in theory, rival transplant hospitals. A more detailed analysis of all potential hospital attributes exceeds the limits of available data and would not nullify our main finding — the many missed opportunities for organ donation at large general hospitals. More generally, opportunities for improvement may vary depending on exact hospital identity, thus suggesting that procurement policies need to be tailored to local circumstances.
Our study is not a randomized trial that minimizes confounding. The incidence of brain death, prehospital transportation of patients who are dying and staffing of hospitals are important determinants of donation rates that cannot be assigned in a controlled experiment. One explanation for our results could be that some hospitals that are inherently enthusiastic about organ donation are early adopters of clinical services for transplant recipients. Alternatively, communities with positive attitudes toward donation might be the most successful at obtaining clinical transplant services at their own hospitals. Regardless of the explanation, our research suggests that actual organ donation may not be an accurate reflection of individual patient wishes.
We used codes for organ donation that were specific rather than sensitive, and they may have missed unidentified cases of organ donation; however, fallible coding is unlikely to explain the size of the discrepancy we saw between transplant hospitals and large general hospitals. In addition, we evaluated a region distinguished by universal health care and public awareness campaigns about transplantation; hence, shortfalls in donation for other regions may be larger. Finally, the ideal rate of organ donation is not known given the importance of patient preferences and community circumstances.
Conclusion
Our research suggests a lower frequency of deceased organ donation at large general hospitals than at transplant hospitals. In particular, we found a substantial age gradient underlying the shortfall in organ donation. This gradient may be partly explained by appropriate medical judgment, given that older organs are sometimes unsuitable for transplantation to young recipients.28–31 However, unfair age discrimination may be an additional explanation.32,33 Indeed, there is no age limit for organ donation, and successful organ procurement can occur from patients older than 80 years of age.34,35
The practice patterns in large hospitals are a reflection of identity, education, incentives and myriad other factors. Thus, addressing the shortfall in organ donation rates could include training, encouragement, regulations, policies, consent procedures and academic detailing campaigns targeting large general hospitals. Any of these approaches would require tact, is prone to misinterpretation and is rarely included in national campaigns for organ donation. The current data suggest, however, that prevailing practices lead to missed potential opportunities for solid organ donation in large general hospitals.
Acknowledgements
The authors thank Sonny Dhanani, Barry McLellan, Sam Shemie, Amit Garg and Robbie Redelmeier for helpful comments. The views expressed in this paper do not necessarily reflect those of the Ontario Ministry of Health and Long-term Care.
Footnotes
Competing interests: Donald Redelmeier, Frank Markel and Damon Scales have received grant funding from the Trillium Gift of Life Network. Frank Markel has served as president of the Trillium Gift of Life Network. No other competing interests were declared.
This article has been peer reviewed.
Contributors: All of the authors contributed to the conception and design of the study, the analysis and interpretation of the data, and drafting the manuscript. All of the authors approved the final version of the manuscript. Donald Redelmeier is the guarantor of the data and results.
Funding: This project was supported by the Canada Research Chair in Medical Decision Sciences, a New Investigator Award from the Canadian Institutes of Health Research, a Fellowship in Translational Health Research from the Physicians’ Services Incorporated Foundation, and an unrestricted grant from the Trillium Gift of Life Network. The funding organizations had no role in the design and conduct of the study; collection, management, analysis and interpretation of the data; and preparation, review or approval of the manuscript.