The evidence is mostly anecdoctal but researchers say an increasing number of Canadians are venturing to other countries for stem cell treatments that are expensive and unproven, probably ineffective and likely risky.
So concerned are they about the rising incidence of stem cell tourism that they’re now developing guidance and talking points that Canadian physicians can use in discussions with patients who are considering a trip abroad for stem cell treatment.
Clinics in China, India, Mexico, Panama, Thailand and Ukraine are actively marketing stem cell therapies to foreigners, says Dominique McMahon, a postdoctoral fellow at the Dalla Lana School of Public Health of the University of Toronto in Ontario.
But it’s difficult to estimate exactly how many Canadians are venturing abroad, she adds. One company, Shenzhen Beike Biotechnology Co., Ltd., offers stem cell therapy in 26 hospitals in China and Thailand and estimates it has treated 900 foreign patients, while the Xishan Institute for Neuroregeneration and Functional Recovery in Beijing estimates it has treated 1000 foreign patients, according to a recent study (Regen Med 2010;5:35–44).
And those are just a few of the more than 200 hospitals offering stem cell therapies in China alone, McMahon says.
Among the Canadians who have travelled abroad for stem cell treatment is 49-year-old Barbara Pineau of Prince Edward Island, who sought treatment in Shenyang, China, in 2007 for multiple system atrophy (Shy–Drager syndrome). For $30 000, along with travel and living expenses for her and husband Philip that amounted to about $3000, she received four weeks of treatment consisting of six stem cell injections (five spinal and one intravenous), as well as acupuncture and physiotherapy five to six times per week.
The package was arranged through Beike Biotechnology. On its website, the firm offers to arrange treatment for a wide variety of conditions, including ataxia, brain injury, cerebral palsy, diabetic foot disease, lower limb ischemia, multiple sclerosis, muscular dystrophy, spinal cord injury and optic nerve damage.
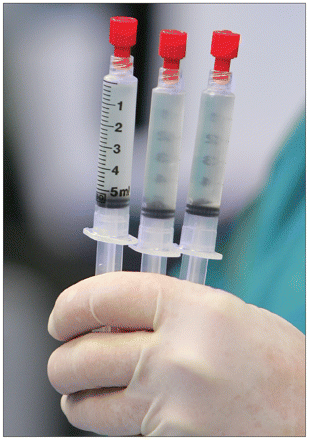
A doctor in Thailand holds vials containing stem cells cultivated from a patient’s blood at a hospital in Bangkok.
Image courtesy of Reuters/Chaiwat Subprasom
Philip Pineau says another Canadian, along with patients from Ireland, Spain, Mexico and Trinidad and Tobago were in the hospital simultaneously receiving treatment for conditions ranging from spinal cord injuries to Alzheimer disease and amyotrophic lateral sclerosis. He adds that neither the firm nor his wife’s physician in Shenyang made any specific promise regarding the outcome of the treatment. The only possible risk mentioned was the possibility of a headache following lumbar puncture.
Nebulous claims about the conditions for which stem cell treatment can be used aren’t uncommon, McMahon says. One company publishes a list of conditions treated that is “a page long, from migraines to impotency to major neurodegenerative diseases,” she asserts.
According to the Stem Cell Network, a National Centre of Excellence, stem cells are used effectively in Canada only for bone marrow transplant, skin grafting, and treating blood diseases such as lymphoid and myeloid leukemias, Fanconi anemia, aplastic anemia, β-thalassemia, sickle cell disease and Hurler syndrome (www.stemcellnetwork.ca/index.php?page=for-patients).
McMahon says companies are typically careful not to guarantee results, although their marketing materials are often more positive “than warranted by the evidence.”
Serious complications of stem cell therapies have been documented, including cases of meningitis (Neurorehabil Neural Repair 2006;20[1]:5–13), a case in which a boy in Russia who was injected with fetal neural stem cells subsequently developed brain and spinal tumours (PLoS Med 2009; 6:e1000029), and a case in which a stem cell transplant led to serious lesions (J Am Soc Nephrl 2010;21: 1218–22).
“In some cases, it is not clear what is being injected,” McMahon says, adding that some facilities use a patient’s own stem cells, while others use embryonic or fetal cells, which can create a risk of rejection. “There’s no proof of safety and efficacy. The quality of facilities varies. The protocols are poorly documented and not available to the patients. Even in the best-case scenarios, the doctor doesn’t know whether it’s safe or efficacious because of a lack of data.” Moreover, there’s rarely any form of follow-up, she says.
Although China revised its regulations in 2010 to require more proof of safety and efficacy, many of its clinics continue to offer treatment for a wide range of illnesses, McMahon says. “Patients are not at fault for going or seeking these treatments, and they have the right to decide the care options available for them. The problem is the regulatory systems that allow these treatments to be available without proof of safety or efficacy.”
For Canadian physicians, dealing with patients who express a desire to venture abroad for treatment, providing advice can be problematic.
The Pineaus say their family physician was neutral, saying “this is a useful area of research and is quite likely to be a source of treatment or cure down the line, but now there’s no science to support what they’re doing.” Their neurologist told them it was a wild goose chase.
Concern over the type of advice provided to patients has prompted a team of health care professionals and researchers, led by Timothy Caulfield, research director at the Health Law Institute at the University of Alberta in Edmonton, to develop some manner of guidance for physicians. After conducting a workshop on the issue in Toronto, Ontario, in 2011, the team plans to soon publish talking points for stem cell tourism, says Amy Zarzeczny, an assistant professor in the Johnson-Shoyama Graduate School of Public Policy at the University of Regina in Saskatchewan.
“We’re trying to distill a complex issue in a user-friendly way,” she says. “Physicians and patients would likely need to go further, depending on their circumstances.”
The International Society for Stem Cell Research has already published a handbook that counsels patients to watch for such things as “claims based on patient testimonials”; claims that there are no risks; the hidden costs of treatment; the need to determine the source of the stem cells; and the need to ensure that the type of treatment offered correlates to the disease (www.isscr.org/clinical_trans/pdfs/ISSCRPatientHandbook.pdf).
Barbara Pineau experienced some short-term improvement in her condition following treatment in China, which she and her husband believe may be due to the acupuncture and physiotherapy. But the atrophy continues to progress.











