Abstract
Background: We sought to establish the long-term safety of drug-eluting stents compared with bare-metal stents in a usual care setting.
Methods: Using data from a prospective multicentre registry, we compared rates of death and of death or repeat revascularization during 3 years of follow-up of 6440 consecutive patients who underwent angioplasty with either drug-eluting or bare-metal stents between Apr. 1, 2003, and Mar. 31, 2006.
Results: Drug-eluting stents were inserted in 1120 patients and bare-metal stents in 5320. The drug-eluting stents were selected for patients who had a greater burden of comorbid illness, including diabetes mellitus (32.8% v. 20.8% in the bare-metal group, p < 0.001) and renal disease (7.4% v. 5.0%, p = 0.001). At 1-year follow-up, the drug-eluting stents were associated with a mortality of 3.0%, as compared with 3.7% with the bare-metal stents (adjusted odds ratio [OR] 0.62, 95% confidence interval [CI] 0.46–0.83). The rate of the composite outcome of death or repeat revascularization was 12.0% for the drug-eluting stents and 15.8% for the bare-metal stents (adjusted OR 0.40, 95% CI 0.33–0.49). In the subgroup of patients who had acute coronary syndromes, the adjusted OR for this composite outcome was 0.46 (95% CI 0.35–0.61). During the 3 years of observation, the relative risks for death and repeat revascularization varied over time. In year 1, there was an initial period of lower risk in the group with drug-eluting stents than in the group with bare-metal stents; this was followed by a shift toward outcome rates favouring bare-metal stents in years 2 and 3. The adjusted relative risk of the composite outcome of death or repeat revascularization associated with drug-eluting stents relative to bare-metal stents was 0.73 early in the first year of follow-up; it then rose gradually over time, to a peak of 2.24 at 3 years.
Interpretation: Drug-eluting stents are safe and effective in the first year following insertion. Thereafter, the possibility of longer term adverse events cannot be ruled out.
Drug-eluting stents now comprise at least 85% of stents used in the United States and up to 40% or more of stents elsewhere. The overwhelming worldwide use of drug-eluting stents has, however, been tempered by the cost differential to bare-metal stents, the lack of data on long-term outcomes in large patient populations and, more recently, emerging concerns about safety because of reports of late thrombosis.1–8
The use of stents has been shown to reduce the rates of repeat revascularization and restenosis after angioplasty compared with angioplasty alone.9 Despite this, the long-term efficacy of stent use has been limited by the need for repeat revascularization owing to restenosis.10 Drug-eluting stents were developed to address this problem. Both clinical trials11–20 and registry data21–25 have shown reduced rates of restenosis with drug-eluting stents up to 4 years after implantation. This advantage appears to extend to patients with acute coronary syndromes: a recent 2-year follow-up study involving 7217 patients with acute coronary syndromes suggested that rates of death were lower among patients with drug-eluting stents than among those with bare-metal stents.26
The possibility of late thrombosis associated with drug-eluting stents is, however, a concern. Rates of late thrombosis have been reported to be 3.6–5.9 events per 1000 patients receiving drug-eluting stents.27 This adverse event has been the subject of a review by the US Food and Drug Administration and has captured the attention of authoritative bodies around the world.
Because of concerns about the long-term safety of drug-eluting stents, we compared the rates of death and of death or repeat revascularization over 3 years among patients who received either bare-metal or drug-eluting stents during angioplasty.
Methods
Study design and patient population
We assembled a prospective cohort of all patients undergoing percutaneous coronary intervention with either bare-metal or drug-eluting stents between Apr. 1, 2003, and Mar. 31, 2006. Patients were followed up to Mar. 31, 2007. We chose the date of Apr. 1, 2003, to coincide with the date that drug-eluting stents were approved for use in Canada.
The Alberta Provincial Project for Outcome Assessment in Coronary Heart Disease (APPROACH) is an initiative for a prospective, geographically defined clinical registry that captures detailed clinical information on all patients undergoing catheterization in the province of Alberta (population about 3 million). Data collection for the registry began in 1995, with patients followed longitudinally for assessment of clinical, health-related quality-of-life and economic outcomes.28 Validation and enhancement of data are performed with the use of a validated methodology.29,30
For our study, we recorded the following clinical variables at the time of catheterization: patient age and sex, presence of congestive heart failure, peripheral vascular disease, cerebrovascular disease, chronic pulmonary disease, renal disease (as indicated by elevated serum creatinine level), diabetes mellitus, dialysis status, hyperlipidemia, hypertension, liver or gastrointestinal disease, malignant disease, smoking status (categorized as “never,” “former” or “current”), prior myocardial infarction (anytime), prior coronary artery bypass graft surgery, prior percutaneous coronary intervention and use of glycoprotein IIb/IIIa inhibitors. In addition, we recorded the primary indication for catheterization as acute coronary syndrome (myocardial infarction within 8 weeks before catheterization or unstable angina), non-acute coronary syndrome (stable angina) or “other.”
We recorded the extent of coronary disease and derived the weighted Duke Index and Duke Myocardial Jeopardy score, an estimate of the percentage of myocardium at risk given the extent of coronary disease.31 We graded left ventricular ejection fraction into 5 categories: less than 20%, 20%–34%, 35%–50%, more than 50% and “ventriculogram not done.” We recorded details of the percutaneous coronary intervention, including use of stent, type of stent (drug-eluting or bare metal), length of stent and number of stents.
Outcomes
We determined the occurrence of death through a semi-annual linkage to data from the Alberta Bureau of Vital Statistics. We obtained information on subsequent revascularization (percutaneous coronary intervention or coronary artery bypass graft surgery) from the APPROACH database. For the purposes of this paper, we chose all-cause mortality and a composite outcome of death or repeat revascularization as the outcomes of interest.
Statistical analysis
We used the χ2 test to compare the clinical and demographic characteristics of patients who received drug-eluting stents with those of patients who received bare-metal stents. We analyzed outcomes at 1 year because we had follow-up data for all patients to at least 1 year. We used logistic regression analysis to calculate risk-adjusted odds ratios (ORs) for death and the composite outcome of death or repeat revascularization at 30 days and at 1 year. ORs below 1.0 indicate a decreased risk of events among patients who received drug-eluting stents.
For survival beyond 1 year, we used Kaplan–Meier survival analyses to compare crude survival and rates of the composite outcome of death or repeat revascularization over the 3-year follow-up period. The survival analyses accounted for censoring of patients when follow-up ended without an event. Next, we extended our survival analysis to include a spline analysis — an analytical technique that dynamically determines the relative risk at moments in time — both in unadjusted and risk-adjusted forms for the 3 years of follow-up. Comprehensive risk adjustment was conducted to account for all of the baseline clinical risk variables presented in Table 1. Spline analysis is a graphical technique that plots the relative risk associated with drug-eluting stents relative to bare-metal stents against time using Schoenfeld residuals.32,33 The resulting curves show the change in relative risk over time, with 95% confidence intervals.
Table 1.
For a sensitivity analysis, we used propensity scores to compare the outcomes of patients in the 2 stent groups. The likelihood (or propensity) to receive drug-eluting stents was modelled, and we then compared outcomes of the 2 groups across tertiles of likelihood to receive drug-eluting stents. We extended the propensity analysis to conduct a 1-to-1 match of each patient with drug-eluting stents to a single patient with bare-metal stents, matched by their propensity to receive drug-eluting stents.
Results
A total of 6471 patients underwent percutaneous coronary intervention with stenting. Of these patients, 1120 (17.3%) received drug-eluting stents, and 5320 (82.2%) received bare-metal stents. Another 31 patients had undeployed stents and were excluded from further analysis. Patients with drug-eluting stents were more likely than those with bare-metal stents to be female and to have a higher rate of renal disease, diabetes mellitus, hyperlipidemia and hypertension (Table 1).
Complete follow-up data for all patients to at least the end of year 1 allowed us to estimate all event rates precisely at 30 days, 6 months and 1 year (Table 2). The 30-day mortality was significantly lower among patients who received drug-eluting stents than among those with bare-metal stents (0.7% v. 1.8%, risk-adjusted odds ratio [OR] 0.50, 95% confidence interval [CI] 0.33–0.76). This difference persisted to the end of year 1 (3.0% v. 3.7%, risk-adjusted OR 0.62, 95% CI 0.46–0.83). The rate of the composite outcome of death or repeat revascularization was significantly lower in the drug-eluting stent group than in the bare-metal stent group at 30 days and at the end of year 1 (4.1% v. 6.3%, adjusted OR 0.42, 95% CI 0.31–0.57 at 30 days; 12.0% v. 15.8%, adjusted OR 0.40, 95% CI 0.33–0.49 at 1 year). We noted the same general findings among patients with acute coronary syndromes. Among patients with non-acute coronary syndromes (i.e., stable angina), we did not detect statistically significant differences in either mortality or the composite outcome at any point between the 2 stent groups.
Table 2.
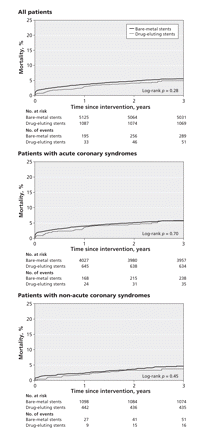
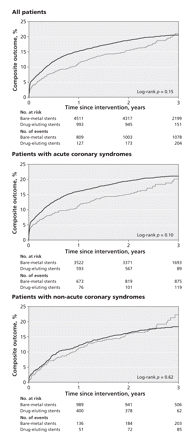
Figure 1 presents the Kaplan–Meier survival curves over the 3 years of follow-up. The survival curves for patients with acute coronary syndromes and for those with non-acute coronary syndromes were similar to the survival curves for the overall study population. When we examined overall event-free survival for the combined outcome (i.e., no death or repeat revascularization), we observed a similar pattern (Figure 2). There were only small differences in survival at year 3 (79.4% v. 78.5%, p = 0.15). The subgroup of patients with acute coronary syndromes had the same general pattern of survival (Figure 2, middle panel). In the subgroup with non-acute coronary syndromes, the differences in event-free survival between the 2 stent groups over time were not significant (Figure 2, bottom panel).
Figure 1: Unadjusted time to death among patients with bare-metal stents and those with drug-eluting stents. Top panel: all patients (n = 6440); middle panel: patients with acute coronary syndromes (n = 4864); bottom panel: patients with non-acute coronary syndromes (n = 1576).
Figure 2: Unadjusted time to composite outcome (death or repeat revascularization) among patients with bare-metal stents and those with drug-eluting stents. Top panel: all patients (n = 6440); middle panel: patients with acute coronary syndromes (n = 4864); bottom panel: patients with non-acute coronary syndromes (n = 1576).
Our stratified analysis of propensity scores revealed that the 1-year mortality was better among patients with drug-eluting stents than among those with bare-metal stents in the 2 tertiles of higher propensity to receive drug-eluting stents (Appendix 1, available at www.cmaj.ca/cgi/content/full/180/2/167/DC2). In the extended propensity analysis in which we conducted a 1-to-1 match of each patient with drug-eluting stents to a single patient with bare-metal stents, we found that survival was again better for patients with drug-eluting stents than for those with bare-metal stents (2.7% v. 4.3% for death, p = 0.043; 11.7% v. 12.7% for the combined outcome of death or repeat revascularization, p = 0.46) (Appendix 1).
Time-dependent spline analysis
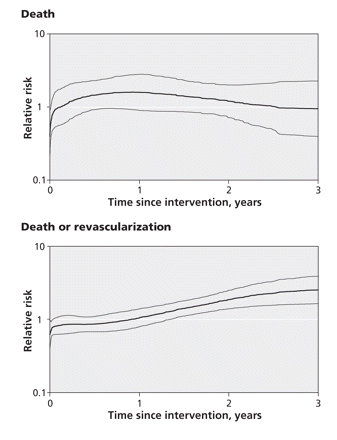
In our time-dependent spline analysis, we observed an initial survival benefit with drug-eluting stents (Figure 3, top panel). Over time the effect diminished, with the relative risk crossing 1.0 and indicating better survival with the bare-metal stents.
Figure 3: Unadjusted spline analysis of the relative risk of death (top panel) and the composite outcome of death or repeat revascularization (bottom panel) among patients with drug-eluting stents (v. those with bare-metal stents) over the 3-year follow-up period. A relative risk below 1.0 indicates a decreased risk of events among patients with drug-eluting stents. The thin lines above and below the thicker line represent 95% confidence intervals.
The corresponding analysis for the combined outcome of death or repeat revascularization showed a similar pattern (Figure 3, bottom panel). During the initial period, outcomes were better with the drug-eluting stents than with the bare-metal stents. Over time, the effect shifted, with the relative risk for the combined outcome favouring bare-metal stents from about 240 days onward. The descriptive statements presented here for Figure 3 emphasize the point estimates for the relative risks at individual moments in time, without attention to statistical significance at each moment. The figure does, however, present bands for the 95% confidence intervals to inform judgments of statistical significance over time.
After we adjusted the spline analysis for baseline risk factors, we found that the findings were similar to those of the unadjusted analysis (Figure 4). Again, there was an initial pattern of decreased risk of events with the drug-eluting stents, followed by a gradual transition over time toward better outcomes with the bare-metal stents. The adjusted relative risk of the composite outcome of death or repeat revascularization associated with drug-eluting stents relative to bare-metal stents was 0.73 early in the first year of follow-up; it then rose gradually over time, to a peak of 2.24 at 3 years.
Figure 4: Risk-adjusted spline analysis of the relative risk of death (top panel) and the composite outcome of death or repeat revascularization (bottom panel) among patients with drug-eluting stents (v. those with bare-metal stents) over the 3-year follow-up period. A relative risk below 1.0 indicates a decreased risk of events among patients with drug-eluting stents. The thin lines above and below the thicker line represent 95% confidence intervals.
Interpretation
Our study showed that patients who received drug-eluting stents during percutaneous coronary interventions had improved survival and lower rates of repeat revascularization in the first year of follow-up compared with patients who received bare-metal stents. However, our time-dependent spline analysis extending to 3 years showed an increased risk of death and repeat revascularization over time associated with the drug-eluting stents relative to the bare-metal stents.
The early studies of the efficacy of drug-eluting stents focused on outcomes in the shorter term, and in some cases restenosis of target vessels as the primary outcome.11,12,34 Our findings build on the results of more recent studies, some of which included patients in usual-care settings or had longer follow-up, or both.5,21,23–26,35
Despite recent concerns about the safety of drug-eluting stents, we did not detect an overwhelming increase in the risk of death among patients who received this type of stent. Also, we did not observe any notable survival drops or clusters of deaths, particularly when the survival of patients with drug-eluting stents was juxtaposed with that of patients with bare-metal stents.
Patients who received drug-eluting stents in our study had different baseline characteristics than those who received bare-metal stents. Drug-eluting stents appeared to be used in an appropriate manner, specifically when the risk of restenosis was known to be increased (i.e., in longer lesions, in smaller vessels, and in patients with diabetes). In the early years of our study, there appeared to be a relative aversion to the use of drug-eluting stents in patients with acute coronary syndromes. At the time, this clinical situation lacked the support of data from recently published randomized controlled trials supporting the use of drug-eluting stents in patients with ST-segment elevation myocardial infarction.36,37
The focus of our study — the long-term survival of patients with drug-eluting stents — enables inferences on the long-term safety profile of stents in light of the recent concerns about drug-eluting stents. Our comparison of the 2 types of stents was not a randomized controlled trial. Therefore, because of potential selection bias and unmeasured risks, we caution readers not to interpret our study as an efficacy analysis.
Despite these limitations, we did perform a risk-adjusted time-dependent outcome analysis to assess the relative risk of adverse events associated with drug-eluting stents relative to bare-metal stents. Interestingly, our findings suggest that the pace of occurrence of adverse events among patients who received drug-eluting stents was not uniform. In fact, there may have been a slight acceleration of events at about 1 year after the percutaneous coronary intervention, in both the unadjusted and risk-adjusted analyses. The potential pathophysiologic factors underlying this observation are not clear. The typical pattern of stopping thiopyridine therapy between 6 months and 1 year after stent placement may be a contributing factor.38 Despite the possible acceleration of adverse events at about 1 year, however, the overall survival of patients with drug-eluting stents remained favourable relative to that of patients with bare-metal stents, at least over 3 years of follow-up.
Strengths and limitations
Our study provides new insights into the long-term safety profile of drug-eluting stents. Indeed, data on the long-term follow-up of patients after stent placement are limited outside of clinical trials. Aside from the studies by Lagerqvist and colleagues23 and Marzocchi and colleagues,35 the principal reports in this area have described populations with exclusive11 or near exclusive22 use of drug-eluting stents with comparison to historical controls. We provide a description of the long-term safety of drug-eluting stents compared with bare-metal stents. Furthermore, our data showed interesting differences in the outcomes between patients with stable angina and those with acute coronary syndromes.
Our study has limitations. Because of the lack of data on long-term medication use, we can only speculate on the potential role the discontinuation of antiplatelet therapy had on the later onset of adverse outcomes in patients with drug-eluting stents. Furthermore, despite a reasonably large sample, our statistical power was not sufficient to comment on small differences in adverse events between the stent groups in the longer term. Also, we confined our analysis of outcomes to death and clinically driven repeat revascularization. However, many patients who experience myocardial infarction following stent insertion would undergo subsequent repeat catheterization, and some may also die. An additional caveat is that, although we performed a careful risk-adjustment analysis, our findings may relate to unmeasured confounding clinical factors, rather than to the type of stent used.39 Finally, because our composite outcome included repeat revascularization, we probably captured procedures done because of disease progression rather than restenosis. However, this limitation probably did not significantly affect our analysis of the composite outcome, because disease progression was likely similar in the 2 stent groups.
Conclusions
Our study findings suggest that the long-term survival (to 3 years) of patients with drug-eluting stents remains favourable overall. It is not measurably worse than that of patients with bare-metal stents. Further work, however, is needed to characterize better the possibly dynamic risk profile of drug-eluting stents over time. Also, data are needed on the potential value of long-term dual antiplatelet therapy to protect against late adverse events in patients with drug-eluting stents.
@@ See related commentary by Généreux and Mehran, page 154
Footnotes
-
Funding: APPROACH was initially funded with a grant from the W. Garfield Weston Foundation. The ongoing operation of this project has been made possible through funding from the Canadian Cardiovascular Outcomes Research Team (CCORT [www.ccort.ca]), a Canadian Institutes of Health (CIHR)-funded team grant initiative. APPROACH is also supported by the Province Wide Services Committee of Alberta Health and Wellness and the following industry sponsors: Merck Frosst Canada Inc., Roche Canada, Eli Lilly Canada Inc., Bristol-Myers Squibb, Philips Medical Systems Canada, Searle Pharmaceuticals, Boston Scientific Ltd. and Cordis — a Johnson & Johnson Company.
Une version française de ce résumé est disponible à l'adresse www.cmaj.ca/cgi/content/full/180/2/167/DC1
This article has been peer reviewed.
Contributors: Andrew Philpott developed the study concept and design, compiled background literature and drafted the manuscript. Danielle Southern and Fiona Clement conducted the data analyses. Diane Galbraith oversaw the collection of data from the APPROACH database. Mouhieddin Traboulsi contributed to the study design and literature search. Merril Knudtson is the principal investigator of APPROACH; he contributed to the study design and literature search. William Ghali oversaw and contributed to all aspects of the study, from conception and design, to analysis, to completion of the final manuscript for publication. All of the authors critically revised the manuscript for important intellectual content and approved the final version submitted for publication.
Acknowledgements: We appreciate the support from the Calgary Health Region, Capital Health Authority, the Libin Cardiovascular Institute and the Mazankowski Heart Institute. We gratefully acknowledge personnel at the cardiac catheterization laboratories of the Foothills Medical Centre, the Royal Alexandra Hospital and the University of Alberta Hospital for their diligence in collecting and entering data.
Merril Knudtson receives partial support from the Libin Trust Fund. William Ghali is supported by a Government of Canada Research Chair in Health Services Research and by a Senior Health Scholar Award from the Alberta Heritage Foundation for Medical Research.
Competing interests: Merril Knudtson received fees from Medtronics for chairing an interventional workshop during the 2006 and 2007 Canadian Cardiovascular Society meetings. None declared for Andrew Philpott, Danielle Southern, Fiona Clement, Diane Galbraith, Mouhieddin Traboulsi or William Ghali.