- © 2008 Canadian Medical Association
Abstract
Background: Recent lipid guidelines recommend aggressive low-density lipoprotein (LDL) cholesterol lowering in patients with coronary artery disease. To clarify the evidence for this recommendation, we conducted a meta-analysis of randomized controlled trials that compared different intensities of statin therapy.
Methods: We searched electronic databases (MEDLINE, EMBASE, Cochrane Central Registery of Controlled Trials, Web of Science) for randomized controlled trials published up to July 19, 2007, that compared statin regimens of different intensities in adults with coronary artery disease and that reported cardiovascular events or mortality. Data were pooled using random-effects models to calculate odds ratios (OR).
Results: A total of 7 trials (29 395 patients) were included. Compared with less intensive statin regimens, more intensive regimens further reduced LDL levels (0.72 mmol/L reduction, 95% confidence interval [CI] 0.60–0.84 mmol/L), and reduced the risk of myocardial infarction (OR 0.83, 95% CI 0.77–0.91) and stroke (OR 0.82, 95% CI 0.71–0.95). Although there was no effect on mortality among patients with chronic coronary artery disease (OR 0.96, 95% CI 0.80–1.14), all-cause mortality was reduced among patients with acute coronary syndromes treated with more intensive statin regimens (OR 0.75, 95% CI 0.61–0.93). Compared with lower intensity regimens, more intensive regimens were associated with small absolute increases in rates of drug discontinuation (2.5%), elevated levels of aminotransferases (1%) and myopathy (0.5%), and there was no difference in noncardiovascular mortality. All 7 trials reported events by randomization arm rather than by LDL level achieved. About half of the patients treated with more intensive statin therapy did not achieve an LDL level of less than 2.0 mmol/L, and none of the trials tested combination therapies.
Interpretation: Our analysis supports the use of more intensive statin regimens in patients with established coronary artery disease. There is insufficient evidence to advocate treating to particular LDL targets, using combination lipid-lowering therapy to achieve these targets or for using more intensive regimens in patients without established coronary artery disease.
Dyslipidemia is the most important modifiable risk factor for myocardial infarction worldwide,1 and serum cholesterol levels are directly related to mortality from coronary artery disease in all populations studied.2–4 Over the past decade, randomized controlled trials enrolling a wide variety of patients have confirmed that for every 1-mmol/L reduction in serum low-density lipoprotein (LDL) cholesterol achieved by statin therapy, the relative risks of cardiovascular events and mortality are reduced (by 21% and 12% respectively).5
Statins exert their beneficial effects primarily by reducing the level of LDL cholesterol,6 and the reductions in the relative risk of cardiovascular events achieved by statin therapy appears to be similar regardless of baseline cholesterol levels.5 As a result, attention has increasingly focused on defining optimal target LDL levels, particularly in patients at highest risk (i.e., those with coronary artery disease). Based on the observational studies mentioned above,2,3 the apparent lack of a lower threshold for statin benefit in the randomized controlled trials, and recent trials reporting greater benefits with more intensive statin regimens (compared with less intensive regimens), Canadian7 and American8 guidelines for secondary prevention now recommend target LDL levels below 2.0 mmol/L in patients with coronary artery disease. On the other hand, European guidelines specify a target LDL of 2.5 mmol/L in these patients.9 Questions have been raised about the safety and incremental benefits of more intensive statin regimens.10–12
We performed a systematic review and meta-analysis to critically examine the evidence for the safety, efficacy (LDL lowering) and clinical effectiveness from trials comparing more intensive statin therapy with less intensive statin therapy in patients with coronary artery disease.
Methods
Eligibility criteria
We included randomized clinical trials that compared different regimens of statin therapy intensity in adult patients with coronary artery disease and that reported cardiovascular events or mortality. We excluded studies that were only published in abstract form. We also excluded trials that compared different intensities of statin therapy but that used much lower statin doses in the control arm than currently used in clinical practice (such as the Post-CABG [Coronary Artery Bypass Graft] Trial conducted in the mid-1990s).13
Study identification and selection
A librarian searched MEDLINE (from 1966), EMBASE (from 1988) and the Cochrane Central Registery of Controlled Trials using relevant subject headings, chemical abstract registry names and text–word searching for statins or hydroxymethylglutaryl-CoA reductase inhibitors (for search terms, see online Appendix 1 available at www.cmaj.ca/cgi/content/full/178/5/576/DC2). The search was conducted on August 10, 2006, and it was updated July 19, 2007. We also conducted a “cited-reference search” in Web of Science for all eligible papers, reviewed the reference lists of all included studies and review articles, and contacted content experts.
Data extraction and quality assessment
K.J. and F.A.M. independently screened all citations, abstracted data and assessed methodologic quality. Disagreements were resolved by consensus. All of the included trials reported composite primary outcomes, but because these differed among studies, they were not appropriate for meta-analysis. Instead, we extracted the individual outcomes from each study that were objectively defined and common among studies (e.g., death, myocardial infarction, stroke).
Data analysis
We extracted intention-to-treat data about cardiovascular events or death, and we used RevMan 4.2 (Update Software Ltd., Oxford, United Kingdom) to perform the meta-analysis of studies reporting the same outcome. We used the DerSimonian and Laird random-effects model to calculate odds ratios (OR) and the I2 statistic to assess heterogeneity for each outcome of interest.14
Results
Study selection and evaluation
Our search identified 429 citations, of which 7 trials fulfilled our eligibility criteria (kappa = 0.70 for agreement on study eligibility) (Figure 1).15–21 We excluded 2 potentially eligible trials that compared different intensities of statin therapy: 1 trial enrolled only patients with familial hypercholesterolemia22 and 1 enrolled a wide variety of patients of whom the majority (54%) did not have coronary artery disease.23
Figure 1: Flow of trials through the selection process.
Study characteristics
Table 1 presents key characteristics from the 7 trials (reporting data from 29 395 patients with coronary artery disease) included in our review.15–21 Of these trials, 2 enrolled patients after acute coronary syndromes and 5 enrolled patients with chronic coronary artery disease. The demographic characteristics of study participants were similar across the included trials. Baseline LDL cholesterol ranged between 2.74 mmol/L in the PROVE IT–TIMI 22 (Pravastatin or Atorvastatin Evaluation and Infection Therapy–Thrombolysis in Myocardial Infarction 22) trial to 3.9 mmol/L in the REVERSAL (Reversal of Atherosclerosis with Aggressive Lipid-Lowering Therapy) trial, TNT (Treating to New Targets) trial (before the run-in period, 2.6 mmol/L before randomization) and the Vascular Basis trial. Three trials were designed with continuous outcomes as their primary outcome: the Vascular Basis and the SAGE (Study Assessing Goals in the Elderly) trials reported the number of ischemic episodes20,21 and the REVERSAL trial reported percent change in atheroma volume.19 Four trials used clinical primary composite outcomes, but the individual components differed (Table 1).15–18 Of note, the definitions for several of the composite cardiovascular outcomes were different in the primary publications from the PROVE-IT and the A-to-Z (Aggrastat to Zocor) trials than those used in the other trials; however, these outcomes were re-reported by the PROVE-IT and A-to-Z investigators (using the definitions from the other trials) in a secondary publication.24 We extracted data on “myocardial infarction or coronary death” and “stroke” for PROVE-IT and A-to-Z from this follow-up publication.24 All other outcomes were extracted from the primary study publication.
Table 1.
Quality of included trials
Although all 7 trials reported blinded outcome ascertainment, their Jadad scores varied from 3 to 5 (out of 5). Three trials adequately reported their method of allocation concealment (Table 2).16,18,19
Table 2.
Generalizability of included trials
Potential threats to study generalizability are reported in Table 2. For example, although the IDEAL (Incremental Decrease in End Points Through Aggressive Lipid Lowering) trial randomized over 90% of patients screened, TNT excluded almost half of those initially screened. Further, although both TNT and IDEAL included patients with stable coronary artery disease, they recruited patients from specialty clinics rather than from primary care settings. In addition, the percentage of patients using statins before randomization varied widely among the trials. The A-to-Z trial excluded all patients who had previously received statin therapy. The TNT trial included a run-in phase such that 100% of patients had received statin therapy for at least 8 weeks before randomization.
Quantitative data syntheses
LDL-cholesterol lowering efficacy
Compared with the lower intensity arm, the higher intensity arm in each trial achieved lower LDL cholesterol levels. The differences in LDL cholesterol levels were between 0.39 and 1.0 mmol/L (weighted mean difference 0.72, 95% CI 0.60–0.84 mmol/L). An LDL cholesterol of less than 2.0 mmol/L was achieved in about 50% of patients in the more intensive statin monotherapy groups (Table 1).
All-cause mortality
The pooled analysis revealed no difference in all-cause mortality between the more or less intensive statin treatment arms in all 7 trials (OR 0.87, 95% CI 0.74–1.03). However, there was substantial heterogeneity among trials (I2 = 42%), and pooling the data obscured the fact that more intensive statin therapy was associated with a 25% reduction in mortality in patients after acute coronary syndrome (based on 353 events in 8659 patients, OR 0.75, 95% CI 0.61–0.93), but had no impact on mortality in patients with chronic coronary artery disease (based on 1333 events in 20 734 patients, OR 0.96, 95% CI 0.80–1.14).
Myocardial infarction or coronary death
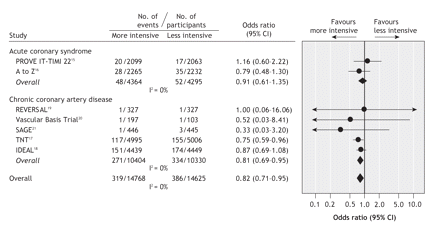
More intensive statin therapy led to a statistically significant reduction in myocardial infarction or coronary death (OR 0.83, 95% CI 0.77–0.91) with no heterogeneity among trials (I2 = 0%) (Figure 2). Examining each of these components separately confirmed that the benefits were seen in patients after acute coronary syndromes or with chronic coronary artery disease and for both nonfatal myocardial infarction (based on 1772 events in 28 439 patients, OR 0.85, 95% CI 0.77–0.93) and coronary deaths (based on 839 events in 28 439 patients, OR 0.84, 95% CI 0.71–0.98).
Figure 2: Risk of myocardial infarction or coronary death among patients with acute coronary syndromes or chronic coronary artery disease in 7 studies of statin therapy intensity.
Stroke
Although TNT was the only trial to demonstrate a significant benefit in stroke reduction, the pooled results demonstrate a statistically significant reduction in the number of strokes with more intensive statin therapy compared with less intensive therapy (OR 0.82, 95% CI 0.71–0.95) with no heterogeneity among trials (I2 = 0%) (Figure 3). The results were similar among patients with chronic coronary artery disease or with acute coronary syndromes (p = 0.21 for comparison between each OR).
Figure 3: Risk of stroke among patients with acute coronary syndromes or chronic coronary artery disease in 7 studies of statin therapy intensity.
Major cardiovascular events
Only the IDEAL, TNT, SAGE and Vascular Basis trials reported rates of major cardiovascular events (myocardial infarction, coronary death or stroke). The pooled analysis confirmed fewer major cardiovascular events in the more intensive statin treatment arm of these trials (OR 0.80, 95% CI 0.71–0.90, I2 = 27%).
Noncardiovascular mortality
Five trials15–18,21 reported noncardiovascular mortality. There was no difference between more intensive and less intensive statin regimens in these trials (based on 670 noncardiovascular deaths in 28 439 patients, OR 0.93, 95% CI 0.69–1.25).
Discontinuation rates
Discontinuation attributed specifically to drug-related adverse events was not significantly higher with more intensive statin therapy (pooled estimate 7.8% v. 5.3% in the less intensive statin arms) (Table 3, Table 4). To add context to the safety numbers, we have included the results from the pooled analyses of the placebo-controlled randomized statin trials (in which patients received doses similar to the “less intensive arm” of the trials reviewed in this meta-analysis) in Table 4.11
Table 3.
Table 4.
Aminotransferase levels
Of the 6 trials that reported this outcome,15–19,21 5 described a significant excess risk of elevated aminotrasferase levels (aspartate aminotransferase or alanine aminotransferase levels more than 3 times the normal upper limit) with more intensive statin therapy compared with less intensive therapy (Table 3, Table 4). The pooled rates were significantly different (1.5% v. 0.4%, OR 4.14, 95% CI 2.30–7.44).15–18,21
Myopathy and rhabdomyolysis
Myopathic adverse events were inconsistently reported and were not significantly more frequent among patients receiving more intensive statin therapy compared with less intensive therapy (Table 3, Table 4). Although the event rates were low, all of these trials used statin monotherapy, not the combination therapy that is frequently recommended to achieve target LDL levels less than 2.0 mmol/L.
Sensitivity analyses
None of the quality variables included in Table 2 influenced the study outcomes. For example, for the most frequent outcomes (myocardial infarction or coronary death), there was no difference in effect estimates for those trials with adequate allocation concealment (OR 0.85, 95% CI 0.77–0.97) compared to those without adequate allocation concealment (OR 0.80, 95% CI 0.71–0.91). There were also no differences in the trials with run-in periods (OR 0.78, 95% CI 0.67–0.91) and those without run-in periods(OR 0.86, 95% CI 0.78–0.95).
Interpretation
Our systematic review demonstrates that, among patients with coronary artery disease, the provision of more intensive statin monotherapy (compared with less intensive statin therapy) reduces LDL cholesterol levels by a further 0.72 mmol/L. This additional reduction in LDL cholesterol resulted in 17% fewer myocardial infarctions (absolute reduction 1.4%) and 18% fewer strokes (absolute reduction 0.5%) among patients randomized to more intensive statin regimens rather than less intensive regimens. These benefits of more intensive statin monotherapy were at the expense of small absolute increases in the frequency of drug discontinuation (about 2.5%), elevated aminotrasferase levels (about 1%) and myopathy (about 0.5%) when compared with less-intensive statin therapy (only the aminotransferase elevations were statistically significant). There was no difference in noncardiovascular mortality. All-cause mortality was not reduced among patients with chronic coronary artery disease, but it was reduced by one-quarter among patients treated after acute coronary syndromes (absolute reduction 1.1%, number needed to treat 91).
Our findings have several clinical implications. First, because all of the trials compared fixed-dose regimens of more intensive statin therapy with less intensive statin therapy and because none provided a breakdown of event rates by the level of LDL cholesterol reduction achieved, the available data cannot be used to define optimal target LDL cholesterol levels. In particular, it should be noted that less than half of all patients who received more intensive statin therapy achieved LDL levels of less than 2.0 mmol/L. Nonetheless, these trials support prescribing more intensive statin regimens for patients with established coronary artery disease, particularly among those with acute coronary syndromes. A recent modeling analysis confirmed that more intensive statin therapy was very cost-effective among acute coronary syndrome survivors (US$12 900 per quality-adjusted life-year). However, the incremental cost-effectiveness among patients with chronic coronary artery disease was uncertain because the small magnitude of benefits (0.10 quality-adjusted life-year) resulted in estimates that were highly sensitive to variations in drug costs and long-term adherence.25
Second, it is important to highlight that these trials provide evidence for more intensive statin monotherapy. None of these trials used, or even permitted, combination therapy. Achieving lower LDL cholesterol targets will often require the use of multidrug therapy, which may result in increased risk of adverse effects, drug–drug interactions and medication nonadherence as well as increased costs for both patients and health care payors.10,26 It is not enough that short-term trials with LDL cholesterol outcomes have demonstrated that other lipid-lowering agents can further lower LDL cholesterol when given along with statins.27–29 Large trials are needed to establish the clinical safety and effectiveness of combination therapy.
Third, the current literature provides limited insight into whether more intensive statin therapy should be used in patients without coronary artery disease but with multiple atherosclerotic risk factors. Although 2 small trials22,23 have demonstrated reductions in carotid intima medial thickness in patients with familial hypercholesterolemia or other atherosclerostic risk factors, there were too few clinical events in these trials to make definitive conclusions. In light of a recent secondary analysis of the TNT trial which suggested that patients with metabolic syndrome may derive even greater benefits from more intensive statin therapy compared with patients without metabolic syndrome, this area of research should be a priority.30
Fourth, although the INTERHEART study and cohort studies from various regions of the world have confirmed that dyslipidemia is the most important modifiable coronary risk factor worldwide,1–4 the trials showing the efficacy of statin therapy have largely enrolled white men. For example, in our meta-analysis 88%–97% of study participants in the 4 trials that reported participant ethnic background were white and 69%–81% were men.15,17,19,21 Although it is unsettling to discover evidence grey-zones for important demographic groups, and such findings should always stimulate research to address these evidence gaps, it is important not to deny treatment to patients in these under-researched groups. For example, although individual trials were unable to confirm the benefits of statin therapy in women, a meta-analysis that pooled individual patient data from 14 trials had sufficient outcomes to confirm that statins prevented coronary events in women (OR 0.82, 95% CI 0.73–0.93).5 Given that “most individuals with a particular condition benefit from therapies proven to be efficacious and relative treatment effects tend to be consistent across differing baseline risks,”31 it seems reasonable to assume that groups at higher coronary risk (such as South Asians) are likely to derive the greatest absolute benefits from more intensive rather than less intensive statin therapy.
Finally, although more intensive statin therapy was well tolerated and relatively safe in the trials we reviewed, it is likely that adverse events will be more common in clinical practice for several reasons. First, trial participants are generally younger and healthier and they are more closely followed than patients in usual clinical practice. In addition, these trials excluded over half of all screened patients because of comorbidities (e.g., advanced age, renal failure, alcohol abuse, hepatic failure) or use of concomitant medications (e.g., other lipid-lowering drugs, inhibitors of the cytochrome P450 system), which may increase the risk of adverse events.32 Second, 2 of these trials only enrolled patients after they had successfully completed a run-in period without complication.33 Third, the time frame of these randomized trials is relatively short and underscores the importance of postmarketing surveillance studies to track complication rates and drug-discontinuation rates in usual clinical practice. The ongoing SEARCH (Study of the Effectiveness of Additional Reductions in Cholesterol and Homocysteine) trial will provide much needed safety information on more intensive statin therapy (simvastatin, 80 mg/d v. 20 mg/d) over a longer timeframe (12 064 study participants, expected average follow-up of 7 years).
In summary, more intensive statin therapy is safe and well-tolerated. It provides incremental benefits over and above those of lower-intensity statin therapy in the secondary prevention of myocardial infarction and stroke in patients with known coronary artery disease, irrespective of their baseline LDL cholesterol levels. Further research is needed to define optimal LDL cholesterol targets, the role of more intensive statin therapy in patients without coronary artery disease and the role of combination therapy.
Footnotes
-
Une version française de ce résumé est disponible à l'adresse www.cmaj.ca/cgi/content/full/178/5/576/DC1
This article has been peer reviewed.
Contributors: All of the authors contributed to the study conception and design. Kiranbir Josan and Finlay McAlister were resposible for data acquisition and analysis. All of the authors contributed to the interpretation of the data, drafted and revised the manuscript critically, and approved the version to be published.
Acknowledgements: We thank Jeanette Buckingham MLIS for assistance with the literature search.
Finlay McAlister and Sumit Majumdar receive career salary support from the Alberta Heritage Foundation for Medical Research and the Canadian Institutes of Health Research. Finlay McAlister is also supported by the Merck Frosst/Aventis Chair in Patient Health Management at the University of Alberta. There was no project-specific funding for this study.
Competing interests: None declared for Kiranbir Josan and Sumit Majumdar. Finlay McAlister has received funding from Pfizer Canada for an investigator-initiated and investigator-designed trial that was peer reviewed and co-founded by the Heart and Stroke Foundation of Canada.