- © 2008 Canadian Medical Association
Many countries around the world — Canada is no exception — are alarmed at the rising incidence of type 2 diabetes and are especially concerned about its earlier age of onset.1 The underlying reason for this dire predicament appears to be the increasing prevalence of obesity in the population and, more precisely, the dysfunctional state of the adipose tissue associated with obesity.2 It is critical to implement strategies for safe and effective prevention or treatment of obesity in order to reduce metabolic and vascular disease.3,4
For a better understanding of the epidemiologic relation between obesity and type 2 diabetes, many clinical investigators have turned to the detailed databases generated by prospective cohort studies. With such large-scale study populations, “gold-standard” approaches to assess the amount and location of body fat, such as underwater weighing and magnetic resonance imaging, are not practical. Instead, cohort members are characterized by other indices of adipose tissue mass that are convenient to perform, but not entirely accurate. For example, body mass index is proportional to body fat, but it is also influenced by lean body mass components, such as muscle, and does not account for anatomical distribution of adipose tissue. Waist circumference focuses on abdominal fat, but it cannot distinguish the subcutaneous depot (metabolically benign) from the visceral adipose tissue depot (metabolically dangerous). Nevertheless, body mass index and waist circumference are highly correlated with one another and perform relatively well as markers and predictors of type 2 diabetes and cardiovascular disease in the general population.5
In this issue, Ray and colleagues6 suggest that we consider a new anatomic predictor of type 2 diabetes: breast size.6 In conjunction with the Nurses' Health Study II, they studied data for 92 106 women (mean age 38 years) who were followed for 10 years. During that time, type 2 diabetes was diagnosed in 1844 of them. The investigators discovered that breast size at age 20 (assessed by recall of bra cup size) was a significant predictor for the development of type 2 diabetes mellitus over the 10-year study period. The age-adjusted hazard ratio was 4.99 for the largest breast size in relation to the smallest breast size. After adjustment for other relevant factors, such as body mass index, waist circumference and family history of diabetes, the hazard ratio dropped to 1.58 but remained significant. The authors carefully note that their Nurses' Health Study II population data emanates from women who were mainly of white ancestry, and that their analysis is based on recall and self-report. Furthermore, the authors call for more research in this area.
While we await future studies, the results reported by Ray and colleagues remind us that white adipose tissue is a complex multi-depot system that should not be oversimplified.7 In addition to managing energy homeostasis via regulated storage of free fatty acids as triglyceride and their release via lipolysis, adipocytes also release a wide array of bioactive molecules (adipokines). Some of these adipocyte-derived proteins are pro-inflammatory and are also produced by adipose tissue macrophages (newly recognized cellular neighbours within adipose tissue). Other adipokines control insulin sensitivity, appetite, energy expenditure and coagulation.
We should consider fat as functional adipose tissue (FAT). It is the dysfunction, rather than the amount, of adipose tissue that appears to be responsible for generating a state of low-grade inflammation and insulin resistance that predisposes to type 2 diabetes. Adipose tissue dysfunction may frequently, but not inevitably, develop as a consequence of excessive adipose tissue growth, or abnormally stunted adipose tissue development seen in congenital or acquired forms of lipodystrophy.8 The question is why does adipose tissue malfunction in obese people?
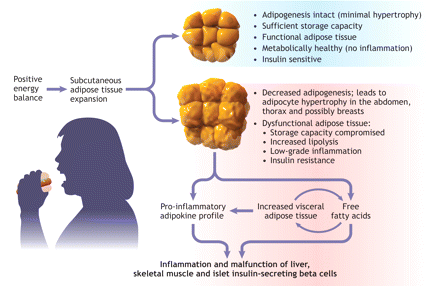
Adipose tissue normally expands by storing energy as triglyceride in existing adipocytes, which causes them to enlarge, and in new adipocytes that differentiate from stromal fibroblast-like preadipocytes (adipogenesis). In our increasingly common state of positive energy balance and under the chronic pressure to store calories, the adipogenic capacity within subcutaneous adipose tissue may falter, which leads to overly hypertrophied adipocytes9 (Figure 1). The swollen adipocytes overproduce pro-inflammatory adipokines and inappropriately release free fatty acids to skeletal muscle, liver and islet insulin-secreting beta cells, which leads to inflammation and dysfunction in those tissues.10 Despite this attempt to compensate, subcutaneous storage capacity is compromised and free fatty acids are relayed to the visceral adipose tissue depot. An increase in visceral fat may be a marker for the compromised ability of subcutaneous adipose tissue to store excess energy.2 Subcutaneous adipose tissue is now established as the main contributor to free fatty acids in the systemic and portal circulation.11 It remains to be seen whether visceral adipose tissue will be directly implicated as a cause of insulin resistance via distinctive adipokine profiles. If adipocyte hypertrophy could be averted via effective ongoing adipogenesis, insulin sensitivity might be maintained despite obesity (metabolically healthy but obese). Indeed, there is a large clinical heterogeneity among obese individuals with respect to insulin sensitivity that remains unexplained.12,13
Figure 1: Figure 1: Model of obesity-induced adipose tissue dysfunction. Positive caloric balance leads to deposition of excess energy as triglyceride in subcutaneous adipose tissue. Defective adipogenesis may limit storage capacity, resulting in hypertrophied, inflamed and insulin-resistant adipocytes. Elevated levels of pro-inflammatory adipokines and free fatty acids are released inappropriately to visceral adipose tissue and to liver, muscle and pancreatic beta cells, which contributes to metabolic malfunction. Image by: Lianne Friesen and Nicholas Woolridge
Even more refined anatomically and functionally distinct depots of adipose tissue, (e.g., femoral v. abdominal subcutaneous depot14 and superficial v. deep subcutaneous adipose tissue layer15) have been detected. Breast adipose tissue, an easily accessible source for sampling human adipose tissue (e.g., after reduction mammoplasty), has been used routinely by researchers for cell-culture-based experimentation for many years. However, it has not been thought of as a functional depot that could influence body-wide metabolism and the development of type 2 diabetes. A nonlactating breast generally comprises less than 20% glandular content, with stromal adipose and connective tissues making up the rest.16 Paracrine interaction between breast adipose cells and ductal cells is an area of active investigation in the context of breast cancer, and breast size has been linked to breast cancer incidence and plasma levels of insulin-like growth-factor–binding protein 1.17–19 Perhaps it is time to ask whether breast adipose tissue could represent a distinct and significant source of circulating adipokines that might have distal effects on metabolic function. Of course, it is also possible that the degree of breast enlargement at age 20 is a result of pre-existing childhood and pubertal physiology. Therefore, it may be an indirect marker of childhood or adolescent obesity and insulin resistance, which in and of itself may set the stage for an elevated risk of type 2 diabetes in adulthood.
Ray and colleagues have left us with an intriguing but preliminary epidemiologic association between breast size and incidence of type 2 diabetes. If confirmed in other studies using more accurate assessments of breast size and thoracic body adipose tissue, it will provide an incentive to search for an underlying mechanistic explanation. It is too early to say whether breast size will be a meaningful indicator to identify women at increased risk for type 2 diabetes. A better understanding of who is predisposed to diabetes will help in the design of more effective and targeted interventions for diabetes prevention.
@ See related article page 289
Key points of the article
• Adipose tissue actively manages complex energy exchanges and produces a variety of influential bioactive factors (adipokines), thereby contributing to metabolic health.
• Adipose tissue dysfunction, induced by obesity, predisposes to type 2 diabetes.
• Inappropriate release of free fatty acids and overproduction of pro-inflammatory adipokines by dysfunctional adipose tissue result in low-grade inflammation and insulin resistance locally and in key target areas such as the liver and skeletal muscle.
• Ray and colleagues6 studied data from 92 106 women in conjunction with the Nurses' Health Study II and found that breast size at age 20, assessed by recall of bra cup size, correlated positively with the incidence of type 2 diabetes.
• It is too early to tell whether breast size is a meaningful predictor of the development of type 2 diabetes.
• A better understanding of who is predisposed to diabetes will help in the design of more effective and targeted interventions for diabetes prevention.
Footnotes
-
Acknowledgements: This work was supported by operating grants from the Canadian Institutes of Health Research and the Heart and Stroke Foundation of Ontario.
Competing interests: None declared.