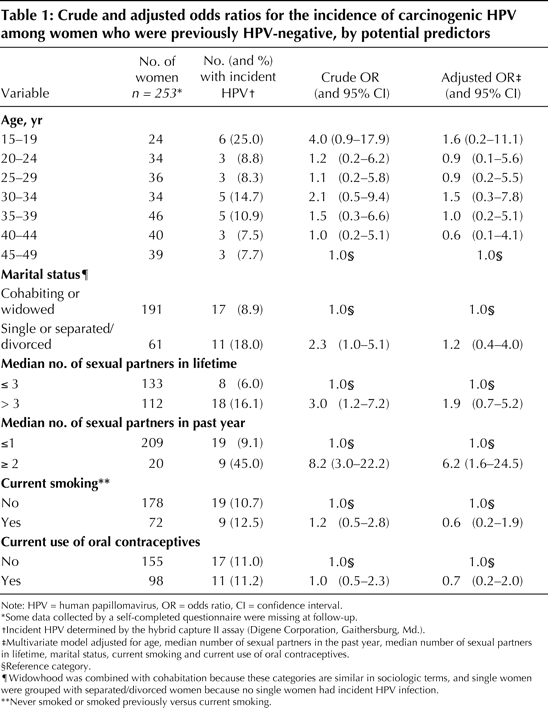
Article Figures & Tables
Figures
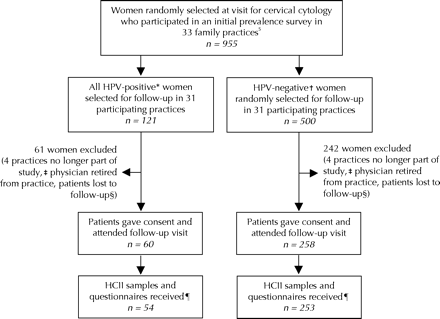
Fig. 1: Flow diagram showing how 253 HPV-negative and 54 HPV-positive women were recruited for 1-year follow-up from among those who had been enrolled in our previous population-based survey of HPV prevalence in Ontario.5 *HPV-positivity for follow-up purposes was determined by the hybrid capture II (HCII) assay (Digene Corporation, Gaithersburg, Md.). †HPV-negative women were found to be negative by both HCII and polymerase chain reaction (PCR) testing; PCR testing indicates the presence of any type of HPV, and genotyping may be used to identify carcinogenic types. ‡Practices that withdrew from the study or discontinued recruitment. §Women were considered lost to follow-up if they refused to participate in the follow-up survey, did not have the physicians' office procedures (no study samples taken at visit for cervical cytology and/or or no appointment within 3 months of Pap test), changed physicians, did not attend their appointment, could not have Pap test (third trimester of pregnancy or recent childbirth), could not be contacted or had no reason/contact recorded (practices did not complete follow-up for lists provided). ¶Several samples were lost or improperly stored and/or questionnaires were not completed by patients or forwarded by the practices.
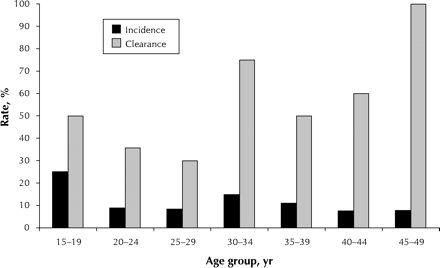
Figure 2. Fig. 2: Age-specific incidence and clearance rates for carcinogenic types of HPV determined by the hybrid capture II (HCII) assay performed at a follow-up visit after an average interval of 14 months on 253 women who had been negative for HPV by both the HCII assay and PCR testing (incidence) and 54 who had been positive by the HCII assay (clearance).