Article Figures & Tables
Figures
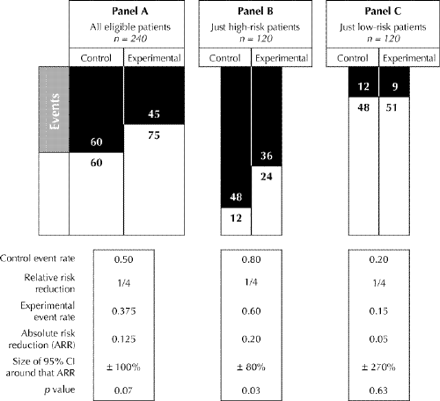
Fig. 1: Effect of enrolling only patients with higher control event rates (“high-risk” patients). In panel A we have randomly assigned 240 patients into equal-sized control and experimental groups (and have lost none to follow-up). Although their overall risk of an event if given conventional therapy is 50% (control event rate 0.50), they are a heterogeneous lot: half of them (panel B) are at high risk if left untreated (control event rate 0.80) and half (panel C) are at low risk (control event rate 0.20). The relative risk reduction (1/4) is the same in all groups. Confidence intervals (CIs) shown here are calculated as the CI for a difference in absolute risk reductions. (6)
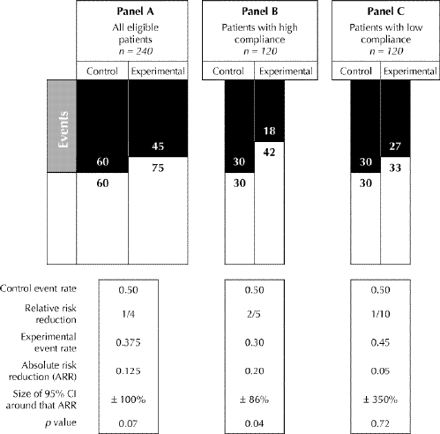
Fig. 2: Effect of enrolling only patients with higher relative risk reductions (highly responsive patients) in an RCT. In panel A we have randomly assigned 240 patients into equal-sized control and experimental groups (and have lost none to follow-up). Although their overall compliance rate is great enough to achieve a relative risk reduction of 1/4, they are a heterogeneous lot: half of them (panel B) are highly compliant and achieve a relative risk reduction of 2/5, and half (panel C) display low compliance and achieve a relative risk reduction of only 1/10. The control event rate (0.50) is the same in all groups.
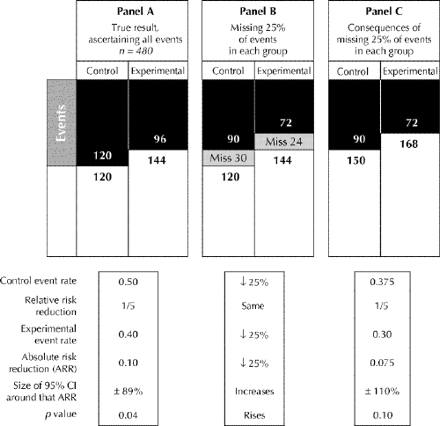
Fig. 3: Effect of equally incomplete ascertainment of events in both control and experimental patients. Panel A displays the true effect of the experimental treatment: a relative risk reduction of 1/5, generating an absolute risk reduction signal of 0.10 whose confidence interval excludes zero. If the experimental and control patients are equally affected by this incomplete ascertainment (missing, say, 25% of events in each group) the misclassification of events depicted in panel B would occur. As a consequence (panel C), although the relative risk reduction is preserved, the absolute risk reduction signal declines from 0.10 to 0.075, its confidence interval now crosses zero, and the trial result becomes indeterminate.
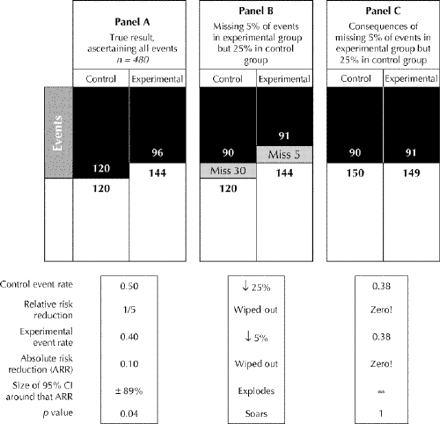
Fig. 4: Effect of better ascertainment of events in the experimental group than in the control group. Panel A displays the true effect of the experimental treatment: as in Fig. 3, there is a relative risk reduction of 1/5, generating an absolute risk reduction signal of 0.10 whose confidence interval excludes zero. If the experimental and control patients are unequally affected by this incomplete ascertainment (missing 25% of events in the control group but only 5% of events in the experimental group) the misclassification of events depicted in panel B would occur. As a consequence (panel C), both the relative and absolute risk reductions are falsely reduced, and the trial draws a false-negative conclusion.