- © 2007 Canadian Medical Association
The case: A 41-year-old woman (gravida 4, para 3) presented in labour after 39 weeks' gestation. Her medical history included myasthenia gravis and thymectomy in 1995 and immunosuppression with azathioprine, which was stopped at 12 weeks' gestation. Her myasthenia was quiescent throughout the pregnancy.
The pregnancy was uncomplicated, as was the vacuum-assisted vaginal delivery. On postpartum day 1, profuse vaginal bleeding occurred, and curettage of the uterus revealed scant tissue. Large bilateral hematomas in the vaginal wall were incised and packed. On postpartum day 2, a second operation was required for continued bleeding. The hematomas were re-evacuated, and the packing was replaced. Postoperative coagulation studies revealed a normal international normalized ratio and fibrinogen levels, a prolonged activated partial thromboplastin time of 49 seconds and a platelet count of 140 × 109/L.
On postpartum day 3, the patient was transferred to our centre. After 3 days of observation, the patient was taken to the operating room for removal of the vaginal packing. Active vaginal hemorrhage was observed in the recovery room. The prolonged activated partial thromboplastin time failed to correct despite transfusion of fresh frozen plasma, packed red blood cells and platelets. Embolization of the left vaginal artery because of continued bleeding was carried out successfully twice that night. Initial 1:1 mixing of the patient's plasma with normal plasma resulted in only partial correction of the activated partial thromboplastin time. On incubation of the mixed plasma, the activated partial thromboplastin time remained prolonged. The patient's factor VIII level was less than 0.01, and factor VIII inhibitor was present, at a titre of 56 Bethesda units. The plasma was negative for lupus anticoagulant.
The patient was admitted to the intensive care unit because of her continuing hemorrhage. On postpartum day 7, pulmonary edema developed and necessitated intubation and ventilation. Recombinant activated factor VIIa concentrate (90 μg/kg [4.8 mg]) was given every 2 hours at first, and then every 3 hours as the bleeding slowed. On postpartum day 14, factor VIII inhibitor-bypassing activity, an activated prothrombin complex concentrate, was substituted for the recombinant activated factor VIIa concentrate, at a dose of 5000 units intravenously every 8 hours because of its lower dosing frequency and because it appeared to be equally efficacious in controlling the patient's bleeding.1 To facilitate eradication of the inhibitor, dexamethasone (4 mg orally every 6 hours) and immunoglobulin (25 g/d intravenously) were administered for 5 days. The dexamethasone was later replaced with prednisone. Cyclophosphamide (100 mg/d) was added in an attempt to achieve more rapid eradication of the inhibitor.2
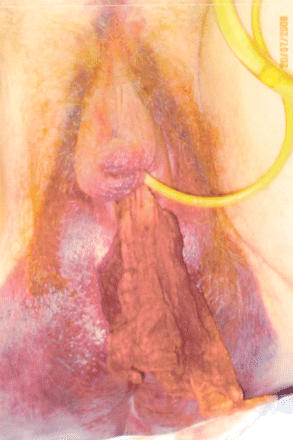
The patient's course was complicated by large perineal (Figure 1), presacral and iliopsoas hematomas. In addition, on postpartum day 8, extensive deep-vein thrombosis developed in the right femoral vein as a result of compression caused by a hematoma in her thigh. Activated coagulation factors (recombinant activated factor VIIa and factor VIII inhibitor-bypassing activity) are known to be thrombogenic. Although various management options were considered, because of the extremely high factor VIII inhibitor level and continued bleeding, the deep-vein thrombosis was managed conservatively and resolved spontaneously. The patient's bleeding gradually decreased, and she was discharged in stable condition on postpartum day 36 even though her activated partial thromboplastin time was still prolonged and factor VIII inhibitor level elevated. The patient continues to be followed in the Saskatchewan Bleeding Disorders Clinic and, at the time of writing, was taking immunosuppressive therapy.
Figure 1: Large perineal hematoma in woman during postpartum period.
Acquired coagulation disorders in which autoantibodies against factor VIII are produced are termed acquired hemophilia A. Although the overall incidence of acquired hemophilia is about 1 per million annually, only 7% of these cases occur in the postpartum period.3 Acquired hemophilia is associated with significant morbidity and potential death. Cases may be associated with underlying autoimmune disease or malignant disease, but at least 50% are idiopathic (see page 341).4 In young women, the most common association is with the puerperium. There is variation in the natural history of factor VIII inhibitors in pregnancy with respect to onset, site, severity of hemorrhage and inhibitor titre.
Our case illustrates the intensive multidisciplinary approach required to manage such complex cases, from surgical, obstetric and hematologic perspectives.
In contrast with our case, factor VIII inhibitors are most commonly found in primigravid patients.2 Acquired hemophilia is typically diagnosed in the postpartum period, but, rarely, it can be detected antenatally or during delivery.2 The median time to inhibitor onset is 2 months, but onset can occur from as soon as the antepartum period to 12 months after delivery.5 Common presentations are soft-tissue or vaginal bleeding, ecchymosis, postoperative bleeding and, rarely, hemarthrosis.2 This contrasts with congenital hemophilia, in which hemarthrosis is much more typical.6 The exact pathogenesis in pregnancy remains unclear.
Diagnosis is based on a high level of suspicion from the patient's history and physical findings. In our case, the discovery of hematomas in the vaginal wall after an intact perineum and vagina had been documented was suspicious, as was the isolated prolonged activated partial thromboplastin time. Laboratory diagnosis is based on the demonstration of an isolated prolonged activated partial thromboplastin time that fails to correct during mixing studies.6 The international normalized ratio and fibrinogen and platelet levels are typically normal. It is necessary to rule out a lupus anticoagulant, which can cause similar aberrations (Box 1). Other criteria include a reduced factor VIII level and evidence of factor VIII inhibitor (as determined by means of the Bethesda method). One Bethesda unit is the quantity of antibody that will inactivate 50% of normal factor VIII activity in a mixture of normal plasma and plasma from the patient after incubation at 37°C for 2 hours.2
Because of the rarity of acquired hemophilia, its management is based mostly on case series and limited prospective data. Management includes supportive care, treatment of acute hemorrhage and eradication of the factor VIII inhibitors.6 Hemorrhage can be treated with human factor VIII infusions; however, this may not be effective, particularly in patients with a high inhibitor titre (> 5 Bethesda units). For severe bleeding or high titres, it is best to reduce inhibitor levels with plasmapheresis or to use agents that circumvent factor VIII.6 These include activated prothrombin complex concentrates such as factor VIII inhibitor-bypassing activity (75 U/kg every 8–12 hours) or recombinant activated factor VIIa (90–120 μg/kg every 3 hours). Because no data are available on which of these agents may be more effective, the choice is at the discretion of the physician and the facility. Eradication of the inhibitors is more challenging and often requires immunosuppressive agents, such as prednisone, cyclophosphamide, azathioprine, immunoglobulins and cyclosporine. The use of rituximab after initial therapies have failed has shown promising results. Recently, it has been recommended as a first-line agent.6
In most cases the inhibitors disappear spontaneously after a median of 30 months and usually do not recur with subsequent pregnancies.3 In his literature review, Franchini2 showed that low inhibitor titres (< 5 Bethesda units) tend to disappear within months, whereas higher titres may persist for years despite treatment. Rarely, such persistence can cause life-threatening hemorrhage in a subsequent fetus because of transplacental transfer of IgG autoantibodies.2 Regardless, the prognosis for most women is favourable. The overall rate of death from all causes of acquired hemophilia is 22%.3 The prognosis for postpartum women tends to be better than this.
In conclusion, acquired postpartum hemophilia must always be considered in the differential diagnosis of postpartum hemorrhage, the general management of which is beyond the scope of this article. This is particularly important in the setting of an otherwise uncomplicated vaginal delivery with an intact perineum and placenta, as seen in our case. An isolated prolonged activated partial prothrombin time should also prompt systematic investigation for other unusual causes. Although rare, factor VIII inhibition, if not diagnosed, may result in life-threatening hemorrhage in otherwise healthy young women. Diagnosis is easily confirmed with the use of mixing studies, in which the activated partial thromboplastin time will fail to correct despite incubation with normal plasma. An array of treatment options to control hemorrhage and eradicate inhibitors is available; the most promising is rituximab for patients with severe bleeding or high inhibitor titres. Transplacental transfer of inhibitors may occur, and women must be counselled about this risk. Multidisciplinary care of these patients is essential.
@ See related article page 341
Footnotes
-
This article has been peer reviewed.
Competing interests: None declared for Kristine Mytopher, Robert Card or Barry Gilliland. Jill Dudebout received travel assistance from Novo Nordisk to attend the 2006 American Society of Hematology conference.