Abstract
OBJECTIVES:
To evaluate performance of the Minolta JM-103 Jaundice Meter (JM) as a predictor of total serum bilirubin (TSB) in outpatient neonates during the first week postnatal, and to estimate the number of TSB determinations that might be avoided in clinical use.
STUDY DESIGN:
In neonates evaluated posthospital discharge, JM and TSB results were compared using linear regression and a Bland–Altman plot, and predictive indices were calculated for various JM cutoff values. Utilizing the 2004 American Academy of Pediatrics (AAP) guidelines, the ability of JM to predict risk zone status was determined.
RESULTS:
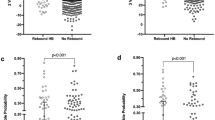
Overall correlation between JM and TSB was 0.77 (p<0.001; n=121). When TSB was >17 mg/dl, a cutoff value for JM of 13 mg/dl had a sensitivity of 1.0, and 50% of TSB determinations would be avoided.
CONCLUSIONS:
JM may facilitate outpatient management of hyperbilirubinemia by reducing the number of TSB determinations required; however, it does not provide a reliable substitute for laboratory measurement of TSB.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
PICK website. Available from: http://www.bilirubin.net.
JCAHO. Sentinel Event Alert: Kernicterus threatens healthy newborns. www.jcaho.org.Issue 18, April 2001.
Centers for Disease Control and Prevention. Kernicterus in full-term infants, United States, 1994–1998. MMWR Morbid Mortal Wkly Rep 2001;50:491–494.
National Quality Forum. A national framework for healthcare quality measurement and reporting: a consensus report. Available from: http://www.qualityforum.org.
American Academy of Pediatrics Subcommittee on Hyperbilirubinemia. Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation. Pediatrics 2004;114:297–316.
Tayaba R, Gribetz D, Gribetz I, Holzman IR . Noninvasive estimation of serum bilirubin. Pediatrics 1998;102 (3). Available from: http://www.pediatrics.org/cgi/content/full/102/3/e28.
Maisels MJ, Newman TB . Jaundice in full-term and near-term babies who leave the hospital within 36 hours. Clin Perinatol 1998;25:295–302.
Robertson A, Kazmierczak S, Vos P . Improved transcutaneous bilirubinometry: comparison of SpectRx BiliCheck and Minolta Jaundice Meter JM-102 for estimating total serum bilirubin in a normal newborn population. J Perinatol 2002;22:12–14.
Schumacher RE . Noninvasive measurements of bilirubin in the newborn. Clin Perinatol 1990;17:417–435.
Rubaltelli FF, Gourley GR, Loskamp N, et al. Transcutaneous bilirubin measurement: a multicenter evaluation of a new device. Pediatrics 2001;107:1264–1271.
Kazmierczak SC, Robertson AF, Briley KP, Kreamer B, Gourley GR . Transcutaneous measurement of bilirubin in newborns: comparison with an automated Jendrassik-Grof procedure and HPLC. Clin Chem 2004;50:433–435.
Schumacher RE . Transcutaneous bilirubinometry and diagnostic tests: “The right job for the tool”. Pediatrics 2002;110:407–408.
Engle WD, Jackson GL, Sendelbach D, Manning D, Frawley WH . Assessment of a transcutaneous device in the evaluation of neonatal hyperbilirubinemia in a primarily Hispanic population. Pediatrics 2002;110:61–67.
Wong CM, van Dijk PJE, Laing IA . A comparison of transcutaneous bilirubinometers: SpectRx BiliCheck versus Minolta AirShields. Arch Dis Child Fetal Neonatal Ed 2002;87:F137–F140.
Briscoe L, Clark S, Yoxall CW . Can transcutaneous bilirubinometery reduce the need for blood tests in jaundiced full term babies? Arch Dis Child Fetal Neonatal Ed 2002;86:F190–F192.
Nanjundaswamy S, Petrova A, Mehta R, Bernstein W, Hegyi T . The accuracy of transcutaneous bilirubin measurements in neonates: a correlation study. Biol Neonate 2004;85:21–25.
Maisels MJ, Ostreia EM, Cepeda E, et al. Evaluation of a new transcutaneous bilirubinometer. Pediatrics 2004;113:1628–1635.
Yasuda S, Itoh S, Isobe K, et al. New transcutaneous jaundice device with two optical paths. J Perinat Med 2003;31:81–88.
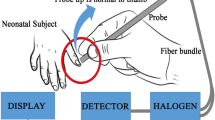
Jaundice Meter JM-103 Instruction Manual. Minolta Co., Ltd. 2002.
Poland RL, Hartenberger C, McHenry H, Hsi A . Comparison of skin sites for estimating serum total bilirubin in in-patients and out-patients: chest is superior to brow. J Perinatol 2004;24:541–543.
Bland JM, Altman DG . Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986;1:307–310.
Bhutani VK, Johnson L, Sivieri EM . Predictive ability of a pre-discharge hour-specific serum bilirubin for subsequent significant hyperbilirubinemia in healthy term and near-term newborns. Pediatrics 1999;103:6–14.
Stevenson DK, Fanaroff AA, Maisels MJ, et al. Prediction of hyperbilirubinemia in near-term and term infants. Pediatrics 2001;108:31–39.
Johnson LH, Bhutani VK . System-based approach to management of neonatal jaundice and prevention of kernicterus. J Pediatr 2002;140:396–403.
Bhutani VK, Gourley GR, Adler S, Kreamer B, Dalin C, Johnson LH . Noninvasive measurement of total serum bilirubin in a multiracial predischarge newborn population to assess the risk of severe hyperbilirubinemia. Pediatrics 2000;106:2/e17.
Acknowledgements
The JM-103™ used in this study was provided without charge by Hill Rom Air-Shields.
Author information
Authors and Affiliations
Additional information
Presented in part at Pediatric Academic Societies meeting, May 2, 2004, San Francisco, CA, USA.
Rights and permissions
About this article
Cite this article
Engle, W., Jackson, G., Stehel, E. et al. Evaluation of a Transcutaneous Jaundice Meter Following Hospital Discharge in Term and Near-Term Neonates. J Perinatol 25, 486–490 (2005). https://doi.org/10.1038/sj.jp.7211333
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7211333
This article is cited by
-
Validity of BiliDx as a point-of-care bilirubin measurement device to diagnose and monitor neonatal jaundice at Muhimbili National Hospital, an observational study
BMC Pediatrics (2024)
-
Evaluation of a new transcutaneous bilirubinometer in newborn infants
Scientific Reports (2022)
-
Reliability of transcutaneous bilirubin determination based on skin color determined by a neonatal skin color scale of our own
European Journal of Pediatrics (2021)
-
Large scale validation of a new non-invasive and non-contact bilirubinometer in neonates with risk factors
Scientific Reports (2020)
-
Evaluation of transcutaneous bilirubinometer (DRAEGER JM 103) use in Zimbabwean newborn babies
Maternal Health, Neonatology and Perinatology (2018)