Abstract
Background: Previous studies have suggested that the immunochemical fecal occult blood test has superior specificity for detecting bleeding in the lower gastrointestinal tract even if bleeding occurs in the upper tract. We conducted a large population-based study involving asymptomatic adults in Taiwan, a population with prevalent upper gastrointestinal lesions, to confirm this claim.
Methods: We conducted a prospective cohort study involving asymptomatic people aged 18 years or more in Taiwan recruited to undergo an immunochemical fecal occult blood test, colonoscopy and esophagogastroduodenoscopy between August 2007 and July 2009. We compared the prevalence of lesions in the lower and upper gastrointestinal tracts between patients with positive and negative fecal test results. We also identified risk factors associated with a false-positive fecal test result.
Results: Of the 2796 participants, 397 (14.2%) had a positive fecal test result. The sensitivity of the test for predicting lesions in the lower gastrointestinal tract was 24.3%, the specificity 89.0%, the positive predictive value 41.3%, the negative predictive value 78.7%, the positive likelihood ratio 2.22, the negative likelihood ratio 0.85 and the accuracy 73.4%. The prevalence of lesions in the lower gastrointestinal tract was higher among those with a positive fecal test result than among those with a negative result (41.3% v. 21.3%, p < 0.001). The prevalence of lesions in the upper gastrointestinal tract did not differ significantly between the two groups (20.7% v. 17.5%, p = 0.12). Almost all of the participants found to have colon cancer (27/28, 96.4%) had a positive fecal test result; in contrast, none of the three found to have esophageal or gastric cancer had a positive fecal test result (p < 0.001). Among those with a negative finding on colonoscopy, the risk factors associated with a false-positive fecal test result were use of antiplatelet drugs (adjusted odds ratio [OR] 2.46, 95% confidence interval [CI] 1.21–4.98) and a low hemoglobin concentration (adjusted OR 2.65, 95% CI 1.62–4.33).
Interpretation: The immunochemical fecal occult blood test was specific for predicting lesions in the lower gastrointestinal tract. However, the test did not adequately predict lesions in the upper gastrointestinal tract.
The fecal occult blood test is a convenient tool to screen for asymptomatic gastrointestinal bleeding.1 When the test result is positive, colonoscopy is the strategy of choice to investigate the source of bleeding.2,3 However, 13%–42% of patients can have a positive test result but a negative colonoscopy,4 and it has not yet been determined whether asymptomatic patients should then undergo evaluation of the upper gastrointestinal tract.
Previous studies showed that the frequency of lesions in the upper gastrointestinal tract was comparable or even higher than that of colonic lesions5–9 and that the use of esophagogastroduodenoscopy may change clinical management.10,11 Some studies showed that evaluation of the upper gastrointestinal tract helped to identify important lesions in symptomatic patients and those with iron deficiency anemia;12,13 however, others concluded that esophagogastroduodenoscopy was unjustified because important findings in the upper gastrointestinal tract were rare14–17 and sometimes irrelevant to the results of fecal occult blood testing.18–21 This controversy is related to the heterogeneity of study populations and to the limitations of the formerly used guaiac-based fecal occult blood test,5–20 which was not able to distinguish bleeding in the lower gastrointestinal tract from that originating in the upper tract.
The guaiac-based fecal occult blood test is increasingly being replaced by the immunochemical-based test. The latter is recommended for detecting bleeding in the lower gastrointestinal tract because it reacts with human globin, a protein that is digested by enzymes in the upper gastrointestinal tract.22 With this advantage, the occurrence of a positive fecal test result and a negative finding on colonoscopy is expected to decrease.
We conducted a population-based study in Taiwan to verify the performance of the immunochemical fecal occult blood test in predicting lesions in the lower gastrointestinal tract and to confirm that results are not confounded by the presence of lesions in the upper tract. In Taiwan, the incidence of colorectal cancer is rapidly increasing, and Helicobacter pylori-related lesions in the upper gastrointestinal tract remain highly prevalent.23 Same-day bidirectional endoscopies are therefore commonly used for cancer screening.24 This screening strategy provides an opportunity to evaluate the performance of the immunochemical fecal occult blood test.
Methods
Study design
For this prospective cohort study, we enrolled consecutive patients aged 18 years or more who voluntarily underwent bidirectional endoscopies as part of a self-paid medical check-up at the Far Eastern Memorial Hospital in Taiwan between August 2007 and July 2009. They were recruited through advertisements for health promotion purposes. Before the examination, a self-administered questionnaire was used to collect information on the participants’ demographic characteristics, social habits, clinical symptoms, and medical and medication histories. The examination protocol included an immunochemical fecal occult blood test, a face-to-face interview, blood chemistry tests, colonoscopy and esophagogastroduodenoscopy.
To ensure that the study population was asymptomatic and that bleeding was occult, we excluded people who had overt gastrointestinal symptoms (e.g., dysphagia or abdominal pain that normally would require an immediate medical evaluation) and overt gastrointestinal bleeding (e.g., hematemesis, tarry stool, melena and hematochezia). We also excluded people who reported a history of malignant disease, polyps in the colon, inflammatory bowel disease or bowel surgery.
The hospital ethics committee approved the study protocol (no. 97024), and people who met the inclusion criteria provided informed consent.
Fecal occult blood test
A one-step commercial immunochemical fecal occult blood test kit with a brush-type sampler (OC-Light, Eiken Chemical Co. Ltd., Tokyo, Japan) was given to all participants. The test has a positive cutoff level of 50 ng/mL. Participants were asked to collect stool samples within two days before the bowel preparation started for the screening endoscopies. They brought the stool samples to the hospital on the screening day; within 24 hours the samples were sent to the laboratory, where they were tested immediately.
Endoscopic examinations
Participants were given sodium phosphate (Fleet; C.B. Fleet Company Inc., Lynchburg, Virginia, USA) for bowel preparation, which they took at least four hours before the endoscopic examinations. The endoscopies were performed with the use of a standard colonoscope (CF-H260AZI; Olympus, Tokyo, Japan) and esophagogastroduodenoscope (GIF-H260Z; Olympus) by two of us (T.-H.C. and C.-H.T.), who were experienced endoscopists who had each performed at least 3000 colonoscopies. The two endoscopists were blinded to the results of the fecal tests.
Endoscopic findings were recorded on a standardized reporting form. They included information on the quality of bowel preparation; completeness of the colonoscopy; number, size and localization of lesions; and whether a biopsy was performed. Participants with an incomplete colonoscopy or poor bowel preparation were excluded from analyses.
Lesions that were identified as important were confirmed clinically. We defined important lesions in the lower gastrointestinal tract as colorectal cancer, colonic adenoma, carcinoid, colitis or ulcer, angiodysplasia and submucosal tumour. Hyperplastic polyps and hemorrhoids were not included in the definition. Important lesions in the upper gastrointestinal tract included cancer, esophageal varix, ulcer of at least 0.5 cm in diameter with a perceptible depth, angiodysplasia, submucosal tumour, and reflux esophagitis with a severity of at least Los Angeles class C or D.25
Statistical analysis
For descriptive findings, we present quantitative data as means and standard deviations, and categorical variables as percentages. Differences in demographic characteristics between participants with positive and negative fecal test results were determined using the Student t test or the χ2 test.
To determine the performance of the immunochemical fecal occult blood test, we used the test results and the colonoscopic findings to construct a 2 × 2 table and calculated the sensitivity, specificity, positive and negative predictive values, positive and negative likelihood ratios, accuracy and corresponding 95% confidence intervals (CIs). Because the immunochemical fecal test is specific for detecting bleeding in the lower gastrointestinal tract, we hypothesized that its specificity for predicting lesions in the colon would be high.
We set a minimally acceptable level of specificity at 85%, a level achieved by the guaiac-based test (Hemoccult SENSA, Beckman Coulter Inc., USA) in our institution. Assuming that the specificity of an immunochemical-based test should reach at least 90%, we determined that a sample size of 750 participants with a negative finding on colonoscopy would be required to detect this difference at α = 0.05, β = 0.1 and a single-tail hypothesis. Knowing that the prevalence of colon lesions in our population is about 20%,28 we determined that an overall sample size of 940 participants would suffice.
We then compared the prevalence rates of lesions in the lower and upper gastrointestinal tracts between participants with positive and negative fecal test results. To test our theory that the fecal test result is not confounded by the presence of a lesion in the upper gastrointestinal tract, we hypothesized that the prevalence of lesions in the colon would be higher among participants with a positive fecal test result than among those with a negative test result. In contrast, we hypothesized that the prevalence of lesions in the upper gastrointestinal tract would be similar among those with positive and negative fecal test results. We assessed statistical differences using the χ2 test.
We evaluated the association between a positive fecal test result and a positive finding on colonoscopy using multivariable logistic regression analysis and controlling for the presence of lesions in the upper gastrointestinal tract. Results were expressed as adjusted odds ratios (ORs) and 95% CIs. Because malignant disease commonly causes occult gastrointestinal bleeding, we compared rates of fecal test positivity between participants with colon cancer and those with esophageal or gastric cancer, expecting the former to be higher.
Finally, we identified risk factors associated with a positive fecal test result and a negative finding on colonoscopy using the backward elimination method in a multivariable logistic regression model. Odds ratios greater than 1.0 indicated an increased risk of a false-positive fecal test result. A two-tailed p value of less than 0.05 indicated statistical significance.
Results
Participant characteristics
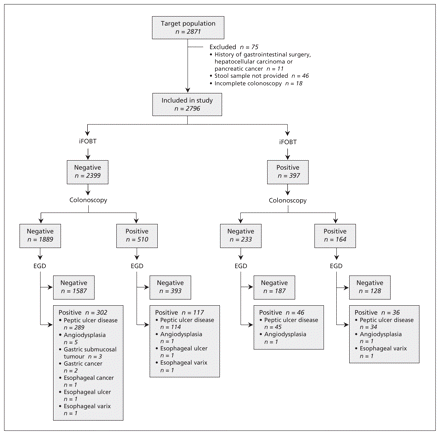
Of the 2871 consecutive people who volunteered to undergo the screening endoscopies during the study period, we excluded 75 because they had a history of gastrointestinal surgery (n = 8), hepatocellular carcinoma (n = 2) or pancreatic cancer (n = 1), they did not provide a stool sample (n = 46), or they had an incomplete colonoscopy (n = 18) (Figure 1). The remaining 2796 participants (1654 men and 1142 women) were included in the study. The mean age was 49.0 (standard deviation 11.2) years. Overall, 397 (14.2%) of the participants had a positive result of the immunochemical fecal occult blood test. The characteristics of the participants did not differ significantly between those with a positive fecal test result and those with a negative result except that a higher proportion of those with a positive result used antiplatelet drugs (Table 1).
Study flow of asymptomatic participants undergoing immunochemical fecal occult blood test (iFOBT), colonoscopy and esophagogastroduodenoscopy (EGD).
Demographic characteristics of 2796 study participants, by result of immunochemical fecal occult blood test
Specificity of fecal test
Lesions were more prevalent in the lower gastrointestinal tract (24.1%) than in the upper tract (17.9%); colon cancer was more prevalent than upper gastrointestinal cancers. Performance of the immunochemical fecal occult blood test in predicting lesions in the lower gastrointestinal tract is shown in Table 2. The test’s specificity was almost 90% for colorectal cancer, adenoma or any important lesion. The sensitivity, specificity, positive and negative predictive values, positive and negative likelihood ratios, and accuracy of the fecal test were all significant for lesions in the lower gastrointestinal tract.
Performance of immunochemical fecal occult blood test in predicting lesions in the lower gastrointestinal tract among the 2796 participants
Prevalence of gastrointestinal lesions by positivity of fecal test
Endoscopic findings are summarized in Table 3. Lesions in the lower gastrointestinal tract were more frequent among participants with a positive immunochemical fecal test result than among those with a negative result (41.3% v. 21.3%, p < 0.001). The frequency of lesions in the upper gastrointestinal tract was similar in both groups (20.7% and 17.5%, p = 0.12). Of the 397 participants with a positive fecal test result, 233 (58.7%) had a negative finding on colonoscopy; of these, 46 (19.7%) had an important lesion in the upper gastrointestinal tract, which was most often a peptic ulcer (97.8%).
Endoscopic findings among the 2796 participants, by result of immunochemical fecal occult blood test
The presence of a lesion in the lower gastrointestinal tract was significantly associated with positivity of the fecal test (adjusted OR 2.59, 95% CI 2.07–3.23). The presence of a lesion in the upper tract was not associated with the test’s positivity (adjusted OR 1.14, 95% CI 0.87–1.49).
Among participants found to have a malignant lesion, almost all who had colon cancer had a positive fecal test result (27/28, 96.4%). In contrast, none of the three participants found to have esophageal or gastric cancer had a positive fecal test result (p < 0.001).
Risk factors associated with a false-positive fecal test result
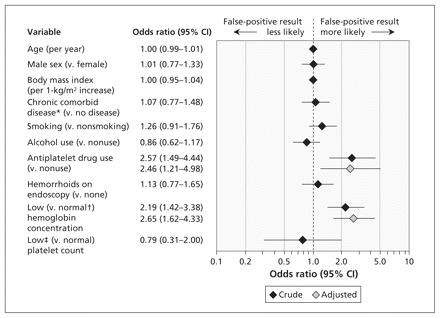
Use of antiplatelet drugs and having a low hemoglobin concentration were the only factors associated with a positive result of the immunochemical fecal occult blood test among participants with a negative finding on colonoscopy (Figure 2). These risk factors remained significant after adjustment for confounding variables (adjusted OR 2.46, 95% CI 1.21–4.98 for use of antiplatelet drugs; 2.65, 95% CI 1.62–4.33, for low hemoglobin concentration).
Risk factors associated with 233 false-positive results of the immunochemical fecal occult blood test among 2122 participants with a negative finding on colonoscopy. The multivariable model adjusted for all variables shown in the forest plot. An odds ratio greater than 1.0 indicates an increased risk of a false-positive result. CI = confidence interval. *Includes diabetes mellitus, hypertension and cardiovascular disease. †Normal hemoglobin concentration: < 120 g/L in women and < 130 g/L in men. ‡Low platelet count: <150 × 109/L.
Interpretation
We found that the specificity of the immunochemical fecal occult blood test was almost 90% for predicting colorectal cancer, adenoma or any important lesion in the lower gastrointestinal tract. These findings support those of previous studies showing that the immunochemical fecal test is a specific diagnostic tool.26–34 Also, the test’s sensitivity was 25% for neoplasms in the colon and 96% for colorectal cancer, findings that are consistent with most results of previous studies evaluating the immunochemical fecal test in either Eastern or Western populations (see Appendix 1 at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.101248/-/DC1).
Previous studies invited asymptomatic participants with a negative result of the immunochemical fecal occult blood test to undergo colonoscopy to validate the test results.26–30 We took the additional step of adding esophagogastroduodenoscopy to clarify the effect of lesions in the upper gastrointestinal tract on the fecal test results. In our study, the prevalence of lesions in the upper tract was consistently about 20% whether among participants with a positive immunochemical fecal test result, a negative fecal test result, or a positive fecal test result and a negative finding on colonoscopy. In addition, the rate of positivity of the fecal test was substantially higher among participants with colon cancer than among those with malignant lesions in the upper gastrointestinal tract. Furthermore, regression analyses showed a lack of association between positivity of the fecal test and the presence of a lesion in the upper gastrointestinal tract. These findings indicate that the immunochemical fecal occult blood test cannot predict the presence of lesions in the upper gastrointestinal tract.
Among participants with a negative finding on colonoscopy, risk factors associated with a false-positive result of the immunochemical fecal occult blood test has rarely been addressed. Bleeding from hemorrhoids is one of the commonly speculated causes of a false-positive result. However, in our study, the prevalence of hemorrhoids was similar among those with a positive fecal test result (61/397, 15.4%) and those with a negative test result (318/2399, 13.3%). In addition, the regression analyses showed a lack of association between positivity of the immunochemical fecal test and the presence of hemorrhoids among participants with a negative finding on colonoscopy. These findings indicate that hemorrhoids should not be used to explain a positive immunochemical fecal test result or hamper the indication for colonoscopy.
Recently, the effect of antiplatelet drugs on the performance of the immunochemical fecal occult blood test has attracted attention, because studies conducted in Western populations have shown that such use may increase the sensitivity of the test in detecting neoplasms in the colon with no or minimal change in specificity.29,30,35 However, in our study, although a low proportion of participants used antiplatelet drugs, their false-positive test rate did increase, such that the sensitivity was increased (32.3% v. 24.3% for the whole study population), but the specificity was reduced (72.9% v. 89.0%) (see Appendix 1).
Limitations
Our study has limitations. First, although we identified antiplatelet use and low hemoglobin concentration as risk factors associated with a false-positive immunochemical fecal test result, the performance of our multiple logistic regression model remains limited in accurately predicting the possibility of such a false-positive result. Also, low hemoglobin concentration appears to be a result rather than a cause. Therefore, it is worthwhile to investigate whether the participants, especially those who were using antiplatelet drugs, had other sources of bleeding, such as small-bowel lesions.
Second, the immunochemical fecal occult blood test does not adequately predict lesions in the upper gastrointestinal tract. Such lesions were present in 20% of the participants in our study who had a false-positive fecal test result and in 6% to 42% of participants in previous studies (see Appendix 2, at www.cmaj.ca/lookup/suppl/doi:10.1503/cmaj.101248/-/DC1). A pan-detecting assay based on stool samples is a recent development that offers an intriguing research opportunity for simultaneous multiple cancer screenings.36 A combination of immunochemical fecal occult blood test and a stool antigen test for H. pylori37 is an approach that may help to realize this goal in a population in which lesions in both the lower and upper gastrointestinal tracts are equally prevalent.38,39 This topic warrants further investigation.
Conclusion
Our findings confirm that the immunochemical fecal occult blood test is specific for predicting lesions in the lower gastrointestinal tract but it does not adequately predict lesions in the upper tract. Among participants who had a negative colonoscopy, a false-positive result of the immunochemical fecal test was associated with the use of antiplatelet drugs and a low hemoglobin concentration.
Footnotes
-
Competing interests: Yi-Chia Lee, Han-Mo Chiu and Ming-Shiang Wu have received grants from the National Science Council of Taiwan for research into the prevention of gastric cancer, the carcinogenesis of colon neoplasms and the pathogenesis of Helicobacter pylori. No competing interests declared by Tsung-Hsien Chiang or Chia-Hung Tu.
-
This article has been peer reviewed.
-
Contributors: Tsung-Hsien Chiang and Yi-Chia Lee contributed equally to this work. Tsung-Hsien Chiang contributed to the study design, data analysis and the drafting of the manuscript. Yi-Chia Lee contributed to the study design, data analysis, and the drafting and revision of the manuscript. Chia-Hung Tu was involved in the acquisition of data. Han-Mo Chiu was involved in the literature review and the interpretation of the data. Ming-Shiang Wu contributed to the study concept and design, the interpretation of data, and the revision of the manuscript. All of the authors gave final approval of the version submitted for publication.
-
Funding: This study was supported by research grants from the Taipei Institute of Pathology.