There are currently about 85 000 Canadians living with spinal cord injuries, more than half of which are secondary to trauma.1 As the population ages, the incidence and prevalence of traumatic spinal cord injury are expected to increase, primarily as a result of fall-related injuries among older adults2. Therefore, treating spinal cord injuries is relevant not only to spine surgeons and physiatrists, but also to the general clinician who will increasingly encounter such patients in the emergency department or family practice.
Here, we review relevant pathophysiology and recent evidence pertaining to the medical, surgical and cellular-based treatment of acute traumatic spinal cord injury. Most of the identified pharmacologic studies were randomized trials or early phase nonrandomized prospective studies. Research relating to the remaining topics was predominately observational in design (Box 1).
Evidence used in this review
We performed a comprehensive literature search of MEDLINE for the key words “spinal cord injury” and the medical subheading “treatment.” We limited the search to clinical articles published between 1980 and 2012 in English journals. We excluded studies involving animals, review articles and case reports. We supplemented this strategy by searching the Cochrane Database of Systematic Reviews for the term “spinal cord injury.” We identified and reviewed 401 abstracts for relevance to the topic. We selected 45 abstracts for which we obtained the full-text version to use as the basis for this review. In addition, we reviewed the 2002 American Association of Neurological Surgeons/Congress of Neurological Surgeons cervical spinal cord injury consensus guidelines, as well as the Consortium for Spinal Cord Medicine 2008 spinal cord injury early acute management clinical practice guidelines.
What mechanisms underlie neural injury and repair?
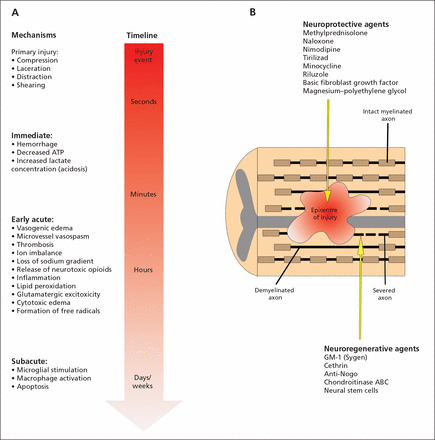
The initial trauma, or primary injury to the spinal cord, starts a sequence of pathological events collectively referred to as secondary injury. These secondary mechanisms begin within seconds of the primary injury and continue for several weeks thereafter, leading to an expanded region of tissue destruction (Figure 1). The initial disruption of the spinal cord vasculature leads to the development of microhemorrhages in the grey and white matter, interstitial edema and the release of coagulation factors and vasoactive amines.3 These events promote thrombosis and vasospasm of the microvasculature of the spinal cord causing tissue hypoxia and impaired neuronal homeostasis. At the cellular level, impairments include ionic imbalance, peroxidation of membrane lipids, formation of free radicals and release of toxic levels of the excitatory neurotransmitter glutamate.4 Neuroprotective agents act to mitigate secondary injury mechanisms to reduce the extent of neural damage.
(A) Primary and secondary mechanisms of injury determining the final extent of spinal cord damage. The primary injury event starts a pathobiological cascade of secondary injury mechanisms that unfold in different phases within seconds of the primary trauma and continuing for several weeks thereafter. (B) Longitudinal section of the spinal cord after injury. The epicentre of the injury progressively expands after the primary trauma as a consequence of secondary injury events. This expansion causes an increased region of tissue cavitation and, ultimately, worsened long-term outcomes. Within and adjacent to the injury epicentre are severed and demyelinated axons. The neuroprotective agents listed act to subvert specific secondary injuries and prevent neural damage, while the neuroregenerative agents act to promote axonal regrowth once damage has occurred. ATP = adenosine triphosphate.
The regenerative capacity of the neurons of the central nervous system (CNS) is severely limited compared with neurons in the peripheral nervous system, largely because of the production of inhibitory molecules that thwart axonal growth, preventing regeneration of injured nerve tracts. Nogo is a family of inhibitory proteins that bind to the Nogo receptor found on regenerating axons.5 This binding leads to the activation of the Rho pathway, causing inhibition of both axonal growth and neuronal cytoskeletal development.6 In contrast to neuroprotective therapies, which limit the extent of acute neural injury, neuroregenerative therapies facilitate neuronal regrowth by several mechanisms, including the blockade of these inhibitory pathways.
What supportive and surgical management is effective?
Historically, it was common for patients with spinal injuries to be placed in unmonitored beds on hospital wards for prolonged periods while elements of the bony injury healed. This approach has been supplanted by aggressive medical and surgical methods focused on maintaining cord perfusion, avoiding complications, decompressing the spinal cord and restoring spinal stability.7
Medical support
The negative consequences of hypotension on the injured CNS are well established.8 There is consistent evidence that avoiding hypotension and maintaining aggressive blood pressure targets during the acute phases after injury improves neurologic recovery and reduces mortality.9 Based on existing, largely retrospective data, the American Association of Neurological Surgeons recommends that patients’ mean arterial pressure be maintained at 85–90 mmHg for the first 7 days after injury.10 When volume replacement is inadequate to achieve this goal, intravenous vasopressor medications may be introduced.
Patients, particularly those with severe cervical injuries, should receive treatment in an intensive care unit (ICU) with continuous cardiac, hemodynamic and respiratory monitoring for the first 7–14 days after injury. In observational studies, the standardized admission of patients with spinal injuries to an ICU has been associated with reduced mortality and morbidity, in addition to improved neurologic recovery.11
Surgical decompression
The preclinical literature provides a strong biological imperative to decompress the spinal cord early after injury.12 In spite of compelling laboratory findings, for many years, surgeons were reluctant to operate acutely owing to concerns that perioperative hemodynamic changes would compromise cord perfusion.13 In addition, until the proliferation of instrumented spinal fixation techniques in the 1990s, decompression meant further destabilizing an already unstable spine.
Retrospective studies provide conflicting results as to the effect of early surgery on neurologic recovery, but all have shown early decompression to be safe.14 The Surgical Timing in Acute Spinal Cord Injury Study (STASCIS),15 a prospective, multicentre, nonrandomized cohort study, compared 313 patients who underwent either early (< 24 h after injury) or late (≥ 24 h after injury) surgical decompression. Early surgery was associated with better neurologic recovery at 6 months as defined by a 2-grade improvement in the American Spinal Injury Association impairment scale (odds ratio [OR] 2.57, 95% confidence interval [CI] 1.11 to 5.97), with no increase in acute complications compared with late surgery (24.2% v. 30.5%, p = 0.21). These results validate those of recent consensus surveys suggesting surgeon preference toward early decompression.16
In the specific case of dislocation of the cervical spine, rapid closed reduction of the spine using skeletal traction remains a valid treatment option. In this case, surgery is performed after closed reduction to re-establish spinal stability.
Therapeutic hypothermia
Preclinical studies have suggested that cooling mitigates secondary injury mechanisms.17 Initial clinical studies involving direct cooling of the cord during surgery failed to show any benefit. However, a recent phase I trial investigated the acute use of modest (33ºC) systemic intravascular hypothermia.18 Complications did not differ between the 14 patients receiving the treatment and the 14 patients to whom they were matched in the control group. At 1-year follow-up, 6 of 14 patients in the treatment group (42.9%) converted from complete (no motor or sensory function below the level of injury) to incomplete (motor or sensory function present below the level of injury) status (3/14 [21.4%] of patients in the control group showed the same degree of recovery), which compares favourably with a neurologic recovery rate of 20% reported in the literature. Although there is currently insufficient evidence to support the use of systemic hypothermia, a multicentre efficacy trial exploring this therapy is being planned.
What drugs have been evaluated for treating spinal cord injury?
To date, 5 pharmacologic therapies have been evaluated in phase III trials (Table 1).19–23 None of them have become standard of care.
Pharmacologic agents evaluated in phase III trials for acute treatment of traumatic spinal cord injury
Neuroprotective agents
Methylprednisolone sodium succinate
Methylprednisolone, which attenuates the peroxidation of membrane lipids and post-traumatic inflammation, has consistently been associated with improved neurobehavioural outcomes in preclinical studies.24 However, the use of methylprednisolone in the clinical setting remains controversial. The most recent Cochrane review pooled the results of 3 studies (meta-analyses) and found no effect for a high-dose 24-hour infusion of methylprednisolone in terms of motor recovery at 6 months (weighted mean difference 0.85, 95% CI −1.79 to 3.49).25 However, when started within 8 hours after injury, methylprednisolone was associated with an additional 4-point improvement in NASCIS motor score (weighted mean difference 4.06, 95% CI 0.58 to 7.55).25 Trends toward increased rates of gastrointesinal hemorrhage (relative risk [RR] 2.18, 95% CI 0.80 to 5.93) and wound infections (RR 2.11, 95% CI 0.81 to 5.49) were seen among patients receiving methylprednisolone.25 Critics of the drug cite these complications, as well as the use of subgroup analyses to prove effect, as arguments against its use.26 Balancing the available evidence, consensus guidelines recommend that 24-hour infusion of methylprednisolone, started within 8 hours after injury, is a treatment option that should only be undertaken with knowledge of the potential complications.27
Other medications
Other treatments studied for neuroprotection include naloxone, an opioid antagonist that blocks the neurotoxic effects of the endogenous opioid dynorphin A;19,28 tirilazad, a nonglucocorticoid 21-aminosteroid developed to inhibit the peroxidation of neuronal membranes;22 and nimodipine, a calcium-channel blocker that prevents calcium-dependent activation of destructive cellular enzymes and presynaptic glutamate release.21,29 Although each of these agents showed efficacy in animals studies, clinical trials of naloxone versus placebo,19 tirilazad versus methylprednisolone22 and nimodipine versus placebo21 have all failed to show differences in motor recovery.
Neuroregenerative agents
GM-1 (Sygen)
Gangliosides are complex glycolipid molecules comprising an important structural component of neuronal membranes. Laboratory studies have shown that gangliosides can enhance axonal regeneration after injury.30 In addition, a variety of neuroprotective effects have been attributed to these compounds. However, a randomized placebo-controlled trial of the ganglioside compound GM-1 (Sygen) involving 760 patients23 reported no difference in the proportion of patients achieving marked neurologic recovery at 6 months, although quantitative results were not presented.
What drugs are in development for treating spinal cord injury?
Several neuroprotective and neuroregenerative agents targeting specific pathological mechanisms are currently in the midst of clinical translation. Although promising, these agents have yet to show efficacy in phase III trials.
Neuroprotective agents
Riluzole
Riluzole is a sodium-channel blocker approved by the US Food and Drug Administration and Health Canada for the treatment of amyotrophic lateral sclerosis (ALS), in which it reduces motor neuron degeneration, thereby prolonging survival.31 In preclinical models of spinal cord injury, riluzole mitigates secondary injury by blocking pathological activation of sodium channels and reducing the release of neuronal glutamate.4 A phase I/II trial evaluating the safety and pharmacokinetics of riluzole for injuries in humans began in 2010 and was completed in January 2012, with full results awaiting publication.32,33
Minocycline
Minocycline, a chemically modified form of tetracycline, has shown to be neuroprotective in animal injury models, although its exact mechanisms of action remain incompletely understood.34 Its use in other clinical conditions, such as acne, shows it has a favourable safety profile in humans. In a randomized placebo-controlled phase II trial, minocycline was associated with a trend toward improved motor recovery at 1 year (difference in American Spinal Injury Association motor score 6 points, 95% CI −3 to 14, p = 0.20).35 A single case of transiently elevated serum transaminases was the only drug-related complication reported.
Basic fibroblast growth factor
Injection of basic fibroblast growth factor has been shown to improve functional and respiratory parameters in animal injury models, presumably by reducing glutamate-mediated excitotoxicity.36 A recombinant version of this molecule is the subject of a phase I/II trial currently recruiting patients.
Neuroregenerative agents
Cethrin
BA-210 is a bacterial-derived toxin that inhibits the Rho pathway of inhibitory proteins and promotes axonal growth in vitro. When combined with a biohemostatic adhesive, BA-210 forms a permeable paste called Cethrin that is applied to the dura of the spinal cord postinjury. Based on documentation of preclinical efficacy, a phase I/IIa trial was undertaken during which 1 of 2 Cethrin dosages was applied to the dura during surgery in 48 patients with complete injuries.6 No serious complications were attributed to Cethrin at 1-year follow-up. Furthermore, among patients receiving doses of 1 and 3 mg, those with cervical injuries showed improvement on the American Spinal Injury Association motor score (mean of 27 points for patients in the 1 mg group and 21 points for patients in the 3 mg group). Such improvements compare favourably to the 10 points of motor recovery reported for similar patients in historical case series.
What is the current status of cellular transplantation as a treatment option?
The transplantation of stem cells and autologous non–stem cells has been intensively studied in preclinical injury models. Various cellular subtypes have been used for this purpose, seeking optimal balance of the one or more key mechanisms through which each is theorized to act (release of growth-promoting trophic factors, environmental modification [i.e., reduction of scar or inflammation] and cellular replacement). In preclinical studies, cellular transplantation, either alone or in combination with other therapies, has been associated with enhanced neurobehavioural recovery, with no single cellular subtype showing superiority.39 Although prematurely used to treat patients with spinal injuries in several countries, no study has established efficacy for the transplantation of any cellular line. However, in the existing early phase trials, major adverse events related to transplantation have been rare. When interpreting the results of clincal studies involving transplantation of cellular subtypes (Table 2),40–48 it is important to consider that, independent of treatment, most patients will undergo some natural neurologic recovery that will plateau 4–6 months after injury. For noncontrolled studies in which patients receive transplants before this plateau, it is impossible to discern whether improvements are related to treatment or simply to the patient’s natural recovery potential. For this reason, results must be interpreted with caution. Overall, cellular transplantation is purely an investigational therapy, which should currently only be undertaken in the context of clinical trials.
Summary of types of cells and the results of associated published or ongoing clinical research into their use for the treatment of spinal cord injury
Future directions
Box 2 provides a fictional case in which the results of this review are applied in clinical practice. Recent laboratory work has identified other promising therapies yet to appear on the clinical landscape. Chondroitinase ABC is a bacterial-derived enzyme that has shown beneficial effects in rodent injury models by degrading elements of the glial scar preventing post-traumatic axonal growth.49 When combined with magnesium, the hydrophilic polymer polyethylene glycol has shown neuroprotective properties in animal models by preserving neuronal membrane integrity.50 Both of these treatments appear poised for eventual translation to the clinic.
Applying the results of this review in clinical practice (fictional case)
A 25-year-old unrestrained male driver presents to the emergency department 30 minutes after a high-speed motor vehicle accident. His mean arterial pressure is 65 mmHg. Neurological examination shows a lack of motor or sensory function below C5. Subsequent computed tomography and magnetic resonance imaging show a fracture and dislocation of the cervical spine causing compression of the spinal cord. No other major injuries are identified.
Immediate attention should be placed on optimizing oxygenation and hemodynamic status, as well as on ensuring immobilization of the craniospinal axis. A mean arterial pressure of 85–90 mmHg should be achieved and maintained for the first week after injury using volume resuscitation, augmented with vasopressors as needed. Consultation with the spine surgery service should occur as early as possible to evaluate the patient’s suitability for immediate decompression and stabilization surgery. Although the patient presents within 8 hours after his injury, methylprednisolone is not standard therapy; rather, it could lead to additional complications. The patient should be admitted to an intensive care unit with continuous cardiac, hemodynamic and respiratory monitoring for the first 1–2 weeks after his injury. Although several additional treatments such as pharmacologic agents, systemic hypothermia and cellular transplantation appear promising in early phase trials, none are currently recommended for routine clinical use.
More recently, several groups have begun to investigate the role of nanomedicine in promoting neuroprotection and neuroregeneration after injury.51 Cerium oxide and gold nanoparticles have shown positive results both in vitro and in vivo. Finally, apart from pharmacologic therapies, researchers are in the early phases of investigating neuromodulatory approaches such as epidural spinal cord stimulation to aid rehabilitation efforts during the chronic phases after injury.52 Although only described in the form of case reports, such approaches appear promising and may someday augment the benefits contributed by acute therapeutics to maximize patients’ long-term potential for recovery.
Key pointsHemodynamic support to maintain mean arterial pressure at 85–90 mmHg and monitoring in an intensive care unit for the first week after spinal cord injury are recommended.
Decompressive surgery within 24 hours after injury has been shown to be safe and feasible; in prospective nonrandomized trials, it has been associated with improved rates of neurologic recovery.
Intravenous methylprednisolone has been used as a neuroprotective strategy based on limited evidence, but this is not a standard of care and may be associated with an increased risk of complications.
The safety of cellular transplantation in human spinal cord injury is under study in early phase clinical trials, but is currently purely an investigational therapy
Early phase clinical trials are investigating potential regenerative (e.g., Rho and Nogo inhibitors) and neuroprotective (e.g., riluzole, minocycline, hypothermia) therapies.
Aknowledgement
Jefferson Wilson received postdoctoral fellowship support from the Christopher and Dana Reeve Foundation.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: All of the authors contributed substantially to the conception and design of the study, to the drafting and critical revision of the article for important intellectual content and to the final approval of the published version.