Intracranial bleeding associated with direct oral anticoagulants
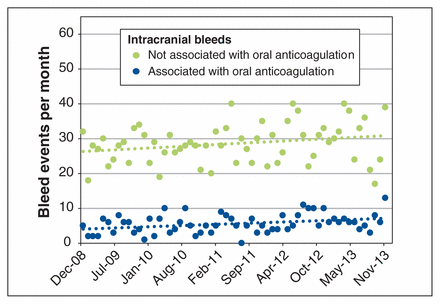
Several direct oral anticoagulants have been approved in Canada for the prevention of stroke in nonvalvular atrial fibrillation. Although phase III studies have shown a lower risk of intracranial bleeding with these agents than with warfarin, there is a lack of real data to validate this finding. In this cohort study, the authors looked at time trends in atraumatic intracranial bleeding at a Canadian neurological centre during the 5-year period (2009–2013) when direct oral anticoagulants were introduced. A total of 2050 patients presented with spontaneous intracranial bleeds during the study period. Of the 371 (18%) prescribed an anticoagulant in the week before presentation, 335 were prescribed an oral anticoagulant. There was an increasing time trend in intracranial bleeding, associated both with (6 additional events per year; p = 0.009) and without oral anticoagulants from 2009–2013, although the latter finding was not statistically significant (p = 0.06) (Figure 1). The authors did not have access to individual patient data, but Ontario prescribing data showed a decrease in warfarin prescriptions during this time (64% of all oral anticoagulant prescriptions by 2013) and increases in prescriptions for direct oral anticoagulants (dabigatran 16%, rivaroxaban 17%). The authors conclude that the increase in spontaneous intracranial bleeding associated with and without oral anticoagulant use likely represents an aging population, rather than bleeding associated with a change in prescribing patterns. CMAJ Open 2015;3:E432–7.
Time trends in atraumatic intracranial bleeding among patients prescribed oral anticoagulants and patients not prescribed oral anticoagulants.
Treatment for mild cognitive impairment
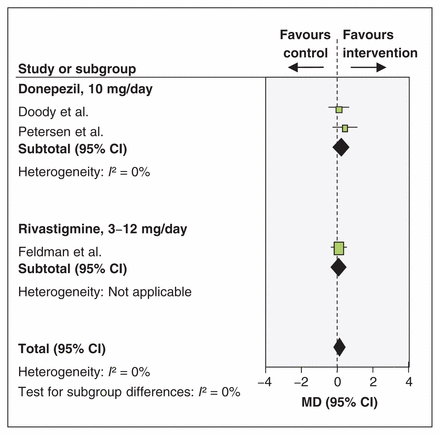
About 10% of Canadians 65 years and older have some form of mild cognitive impairment that is often undetected. Will they benefit from pharmacologic or nonpharmacologic interventions? Seventeen randomized controlled trials were included in this systematic review that looked at the effectiveness of treatment for mild cognitive impairment. Of the studies evaluating effects of treatment on cognition (using the Alzheimer’s Disease Assessment Scale, cognition subscale), there was no difference between intervention and control groups for cholinesterase inhibitors, behavioural therapy and vitamin E. In studies looking at effects of treatment on the Mini-Mental State Examination, cholinesterase inhibitors and dietary supplements or vitamins showed no effect (Figure 2); however, there was a significant difference favouring intervention for behavioural therapies (mean difference 1.01, 95% confidence interval 0.25–1.77). There was no increase in the risk of serious adverse events for both pharmacologic and nonpharmacologic interventions. This review found no evidence for the benefit of pharmacologic interventions, including cholinesterase inhibitors, in treating mild cognitive impairment. Although a small cognitive benefit was observed with behavioural therapies, the clinical significance of this remains uncertain. CMAJ Open 2015;3:E419–27.
Effects of cholinesterase inhibitors on patient cognition as assessed by the Mini-Mental State Examination. CI = confidence interval, MD = mean difference.