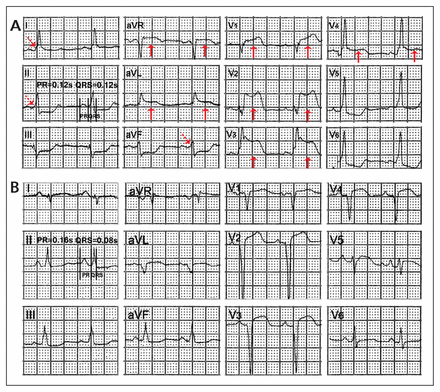
A 54-year-old man with a 20-year history of recurrent paroxysmal tachycardia was admitted to hospital because of chest pain. The electrocardiogram (ECG) on admission (Figure 1A) showed signs suggestive of an anteroseptal accessory pathway, as well as ST segment elevation in leads V1 through V4, aVL and aVR, with elevated levels of cardiac enzymes. This was consistent with a diagnosis of Wolff–Parkinson–White syndrome with acute myocardial infarction (MI).1 The patient’s chest pain persisted for two hours after coronary thrombolytic therapy. A subsequent ECG showed right bundle branch block, left anterior fascicular block and no evidence of preexcitation changes. Pathologic Q waves in leads V1 through V3 with persistent ST segment elevation were also noted. A coronary angiogram showed a proximal left anterior descending artery occlusion, which was successfully stented. Following this procedure, the patient became asymptomatic and a subsequent ECG (Figure 1B) showed complete resolution of the right bundle branch block and the preexcitation changes, with persistent anterior Q waves. An ECG taken 1 year after discharge showed no evidence of Wolff–Parkinson–White syndrome or tachycardia.
(A) Electrocardiogram of a 54-year-old man with chest pain. The QRS complex was in QS pattern in lead V1, the delta wave was positive in leads I, II and aVF (dashed arrows), and ST segment elevation was in leads V1 through V4, aVL and aVR (solid arrows), suggestive of Wolff–Parkinson–White syndrome (anteroseptal accessory pathway) with acute myocardial infarction of the anterior wall. (B) Electrocardiogram of the same patient after stent implantation showing no preexcitation wave.
The prevalence of Wolff–Parkinson–White syndrome is about 1–3 per 1000.2 In the syndrome, some ventricular depolarization occurs via a rapidly conducting accessory pathway. This results in a short PR interval and a delta wave (which prolongs the QRS). When a delta wave is present, repolarization also occurs more rapidly, altering the ST–T wave (in the opposite direction with the delta wave vector) and confounding the diagnosis of acute myocardial ischemia or infarction by routine ECG criteria. In our patient, the diagnosis of acute MI was well established by the marked ST–T wave elevation (concordant with the delta wave vector), the clinical picture and enzyme elevation.
There are at least two plausible explanations for the disappearance of the syndrom in our patient following acute MI: the preexcitation syndrome may have been intermittent, or he may have had an accessory bundle in the ischemic region. Because the blood supply of the accessory pathway was the infarct-related artery (proximal to the left anterior descending artery), we believe that the latter is most likely and our patient’s Wolff–Parkinson–White syndrome was effectively “cured” by his MI.
Footnotes
-
Competing interests: None declared.
-
This article has been peer reviewed.