Over the past two decades, the use of noninvasive positive-pressure ventilation and noninvasive continuous positive airway pressure by mask has increased substantially for acutely ill patients. Initial case series and uncontrolled cohort studies that suggested benefit in selected patients1–13 led to many randomized controlled trials (RCTs).14–151 Both methods of ventilation have been used in the setting of acute respiratory failure to avoid endotracheal intubation in different patient populations and settings, with variable success.14–69,117–123,134–141 In addition, noninvasive positive-pressure ventilation has been used to facilitate early liberation from conventional mechanical ventilation72–82 and to prevent reintubation.70,71,83–87
These guidelines were developed to answer the following questions: What evidence is available in the literature to support recommendations for the use of noninvasive positive-pressure ventilation and continuous positive airway pressure for patients who are at risk of or who have acute respiratory distress or failure, patients who have undergone surgery and patients who are being weaned from mechanical ventilation or have recently undergone extubation? In addition, how can these two modes of noninvasive ventilation be optimally applied in these settings?
Consensus conference statements152,153 and guidelines154–157 exist for the use of noninvasive ventiliation (the term used throughout this paper to refer to both noninvasive positive-pressure ventilation and continuous positive airway pressure) in the acute care setting. However, since publication of the two English guidelines addressing use in the acute care setting,154,156 many new RCTs have been published. In addition, the first154 of the two previous guidelines predated the development of the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group methodology,158–160 and a less rigorous methodology was used in the second.156 Accordingly, we conducted a comprehensive search and appraisal of the current literature, using the GRADE methodology to assess the quality of the research and to generate clinical recommendations.
Methods
Leadership and scope
An 18-member guidelines panel of university-affiliated clinicians, led by two cochairs, was formed in June 2007 as an initiative of the Canadian Critical Care Trials Group/Canadian Critical Care Society Noninvasive Ventilation Guidelines Group. The panel consisted of 15 physicians and 3 respiratory therapists who had a variety of backgrounds in clinical and/or nonclinical expertise: 15 were experts in critical care medicine, 12 in pulmonary medicine, 5 in noninvasive positive-pressure ventilation, 15 in clinical research methodology and 5 in guideline development.
The target audience for these guidelines is clinicians who use either form of noninvasive ventilation for acutely ill patients. The referent setting was the Canadian health care system.
Data sources
We searched the following electronic databases from their inception to June 2009: MEDLINE, EMBASE, CINAHL, CENTRAL (the Cochrane Central Register of Controlled Trials), DARE (the Database of Abstracts of Reviews of Effectiveness), the Cochrane Database of Systematic Reviews, ACP Journal Club database, the metaRegister of Controlled Trials, ClinicalTrials.gov and the Journals@Ovid database. For the search strategies, see Appendix 1 (available at www.cmaj.ca/cgis/content/full/cmaj.100071/DC1). We also searched personal files and the bibliographies of relevant articles.
Selection criteria
We identified RCTs that included adults admitted to acute care hospitals who were at risk of or who had acute respiratory failure. We included studies involving patients with acute or acute-on-chronic respiratory failure. In clinical terms, there is a continuum from respiratory distress, marked by increased work of breathing (where an increase in the respiratory rate reflects the increase in work of breathing), to respiratory failure, for which immediate endotracheal intubation is required. Various definitions of respiratory failure have been used conventionally for years, but they are somewhat arbitrary and not always useful for the clinician. Some patients who are in clear respiratory extremis may not meet the blood gas criteria for respiratory failure. Conversely, other patients have respiratory failure as defined by clinical criteria but do not need urgent ventilation. Clearly, there is a spectrum of severity of respiratory compromise that, in general, is worse with increasing respiratory rate and signs of increased work of breathing. Some studies, but not others, have specified strict blood gas criteria for respiratory distress and failure. Rather than defining respiratory failure a priori, we included trials that used either clinical or arterial blood gas criteria, as well as studies in which patients were in respiratory distress according to clinical criteria and were believed to be at risk of requiring ventilatory support in the future.
We excluded studies of noninvasive ventilation for patients with chronic respiratory failure in an outpatient setting. Each included trial directly compared the two types of noninvasive ventilation with standard therapy or with each other and reported physiologic outcomes (e.g., arterial blood gases, vital signs) and/or clinical outcomes (e.g., endotracheal intubation, hospital mortality). We included only parallel-design RCTs. We excluded studies with crossover design because inferences about clinical outcomes are limited in these trials. We also excluded pseudorandomized trials (such as those in which patients were assigned to study group by alternate assignment or hospital registry number) and abstract publications.
No language restrictions were applied; non-English publications were professionally translated into English.
The panel cochairs independently reviewed all study abstracts and selected potentially relevant trials for further review. Disagreements on inclusion of articles were resolved through consensus between the two cochairs.
Quality assessment and data abstraction
Of the 18 members of the guideline panel, 16 participated in the review of RCTs, working in eight pairs. Working independently, each member of each pair of reviewers critically appraised assigned RCTs using a tool developed by the panel specifically for this project. For each RCT, each member of the pair of reviewers independently abstracted data regarding trial validity, and the two reviewers in each pair achieved consensus on the validity and results of assigned RCTs.
The following data were abstracted to determine validity: concealment of allocation, selection of patients, similarity of groups at baseline, treatment protocol, co-interventions, definitions of outcomes, extent of follow-up, adherence to the intention-to-treat principle, patient population, hospital location, number of sites and strategies for the initial application and titration of noninvasive ventilation. All reported outcomes were recorded.
We conducted meta-analyses for topics for which at least two RCTs were identified. For each population and intervention (noninvasive positive-pressure ventilation, continuous positive airway pressure by mask), we used relative risk (RR) as the summary estimate of effect for dichotomous outcomes and weighted mean difference for continuous outcomes. We used a random-effects model.
Development of recommendations
All members were given literature describing the GRADE methodology to help generate a summary statement for each guideline topic.158–160 In addition, a GRADE expert reviewed this methodology with the panel members on two occasions before the start of the deliberation process. The panel met in person three times and communicated frequently by email. Before each meeting, summaries of the pooled results (based on meta-analyses) and evidence tables were sent to the panel members for review.
At the first meeting, the guideline panel agreed to use endotracheal intubation and hospital mortality as the primary outcomes of interest. We used length of the hospital stay if endotracheal intubation and mortality were either unavailable in the original publications or not significantly different between groups. For trials that assessed different modes of ventilation or interfaces (i.e., types of masks), we also used measures of patient tolerance as an outcome of interest.
The outcome of “treatment failure,” defined by trial-specific physiologic and gas exchange criteria, was reported in a number of recent trials. Many of these trials allowed crossover of patients from the standard therapy arm to either noninvasive positive-pressure ventilation or continuous positive airway pressure, which potentially confounds the outcomes of endotracheal intubation and hospital mortality. For clinical topics where this approach was used in either a large proportion of trials or in a few trials accounting for a large proportion of patients, we also considered treatment failure as an outcome of interest. In pooling results for treatment failure, we used rates of endotracheal intubation when specific criteria for treatment failure were not included.
The GRADE methodology initially entailed assigning to each guideline statement both a number (to summarize the strength of the statement) and a letter (to summarize the quality of the evidence). More recently, some guideline experts have preferred to include or substitute symbols and/or statements for the traditional number and letter grades. Table 1 summarizes the GRADE Working Group’s definitions of these categories.158–160 Standard terms in these statements were “recommend,” “suggest” or “no recommendation.” For topics we considered to have sufficient quality and quantity of supporting evidence from RCTs, strong recommendations were assigned a grade of 1 and were described by the phrase “we recommend”; weaker recommendations were assigned a grade of 2 and were described by the phrase “we suggest.” For topics that lacked sufficient evidence, we made no recommendation. In these situations, we stated that noninvasive positive-pressure ventilation or continuous positive airway pressure could be considered but that there was a lack of literature to support either intervention because published RCTs were inconclusive (denoted by the term “lack of sufficient evidence”) or because no RCTs had been published (denoted by “lack of RCTs”).
We used GRADEprofiler (GRADEpro) software, version 3.2 (available through Cochrane IMS) to generate evidence tables from which panel members developed the final guideline statements. The GRADEpro software generated letter grades, which incorporated limitations of study design, inconsistency, indirectness, imprecision and publication bias, to assess the quality of evidence as high, moderate, low or very low. The panel then systematically evaluated these quality criteria. The panel also incorporated semiquantitative assessments of safety (estimated probability of avoiding significant harm that may be associated with the intervention), feasibility (ease of implementation of the intervention) and cost (estimated cost of implementing the intervention) into the GRADEpro document for each topic. We did not include direct assessments of patient values and preferences. Our underlying values and preferences were to ensure that any recommended use of these technologies was safe, effective, affordable and feasible within the Canadian health care system.
After each panel meeting, the cochairs compiled the summaries and recommendations, and the panel members reviewed this material for clarity and accuracy. To assess the degree of agreement of each panel member with the final guideline statements, all members independently rated their agreement with each statement using a Likert-type scale from 1 to 9, anchored by “disagree completely” at the low end and “agree completely” at the high end.
The panel members prepared background documents for the topics they reviewed, and the cochairs compiled these documents into the results section of this guideline document. We formatted the full guideline document to include a summary of the evidence and a statement for each topic. We also developed a summary of the guideline statements as a quick reference guide.
A draft of the guideline document was submitted to the executive of the Canadian Critical Care Society and for internal review by three reviewers of the Canadian Critical Care Trials Group. Both groups reviewed the guidelines and endorsed the final version. In addition, an international expert on noninvasive ventilation reviewed the final guidelines. All reviewers were asked to assess the logic, clarity and practicality of the guideline. The cochairs revised the document on the basis of feedback received. As an additional assessment of the comprehensiveness of these guidelines, and to be as transparent as possible about its potential deficiencies, we evaluated the final guideline using the AGREE (Appraisal of Guidelines Research and Evaluation) instrument.161
Funding
Funding for this work was provided by a grant from the Canadian Institutes of Health Research and an unrestricted grant from Philips/Respironics Inc., Murrysville, Pennsylvania. The funding sources played no role in selection of studies, interpretation of the appraisals, or development, review or approval of the guidelines.
Clinical recommendations
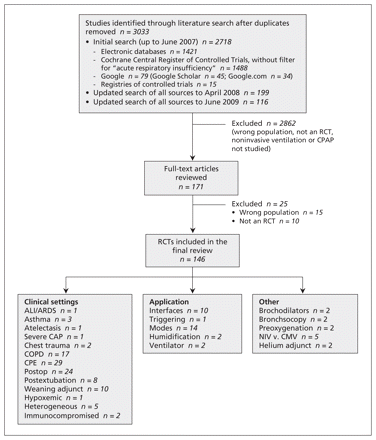
The literature search, beginning with a general overview of the literature and then focusing on the RCTs, is summarized in Figure 1. In total, we identified 3033 studies and included 146 RCTs in our review. We report here the final summary statements and their grades for use of noninvasive positive-pressure ventilation or continuous positive airway pressure by mask in acutely ill patients in the following three clinical settings: acute respiratory failure, management after intubation, and the postoperative setting. We also report recommendations on technical strategies to optimize the use of noninvasive ventilation (e.g., interfaces, mode). See Table 2 for a summary of the statements. The abridged version of this article (available at www.cmaj.ca) includes a table organized by the strength and quality of the recommendations. The degree of agreement among members of the consensus panel was high (see Appendix 2, available at www.cmaj.ca/cgis/content/full/cmaj.100071/DC1), which added some validity to the final statements.
Summary of search results. ALI = acute lung injury, ARDS = acute respiratory distress syndrome, CAP = community-acquired pneumonia, CMV = conventional mechanical ventilation, COPD = chronic obstructive pulmonary disease, CPAP = continuous positive airway pressure, CPE = cardiogenic pulmonary edema, NIV = noninvasive ventilation, RCT = randomized controlled trial.
Summary of clinical practice guideline statements for the use of noninvasive positive-pressure ventilation and noninvasive continuous positive airway pressure in the acute care setting
Acute respiratory failure
Exacerbation of COPD
Noninvasive positive-pressure ventilation versus standard therapy
We identified 16 published RCTs14–24,26,28–31 comparing noninvasive positive-pressure ventilation plus standard therapy with standard therapy alone for patients with exacerbation of COPD, 14 of which reported endotracheal intubation and hospital mortality. We excluded two trials in which patients who experienced respiratory failure were not offered endotracheal intubation.14,17 The included trials had lower incidence of endotracheal intubation (RR 0.39, 95% confidence interval [CI] 0.28–0.54) and hospital mortality (RR 0.52, 95% CI 0.36–0.76) among patients who received noninvasive positive-pressure ventilation. Inclusion and exclusion criteria were variable, but the majority of trials included patients with severe exacerbations, defined as initial arterial pH less than 7.35 (and relative hypercarbia), and a correspondingly high intubation rate in their control arms. The three RCTs involving patients with milder exacerbations did not find an important reduction in risk of endotracheal intubation (RR 0.71, 95% CI 0.16–3.08) or hospital mortality (RR 1.05, 95% CI 0.07–6.36).21,29,31 A single RCT included a subgroup of patients with acute exacerbation of COPD in the setting of severe community-acquired pneumonia,38 whereas almost all other trials involving patients with exacerbation of COPD excluded patients with pneumonia. In that trial,38 use of noninvasive positive-pressure ventilation was associated with a reduction in endotracheal intubation (0/12 in intervention arm v. 5/12 in control arm, p = 0.005).
Helium–oxygen with noninvasive positive-pressure ventilation
One RCT evaluated the use of helium–oxygen (heliox; 80:20 mixture) in addition to noninvasive positive-pressure ventilation for patients with exacerbation of COPD.33 There was no difference in rate of endotracheal intubation (8/59 with heliox v. 13/64 for control; p = 0.33) or hospital mortality (9/59 v. 6/64; p = 0.48).
Noninvasive positive-pressure ventilation versus conventional mechanical ventilation
Two published RCTs compared noninvasive positive-pressure ventilation with intubation and conventional mechanical ventilation for patients with exacerbation of COPD severe enough to warrant immediate assisted ventilation.120,123 These patients constitute a subgroup that is often excluded from other trials designed to prevent intubation in patients with COPD, because they meet the criteria for intubation at the time of presentation. Use of noninvasive positive-pressure ventilation resulted in avoidance of intubation for the majority of patients in these two trials (34 of 61), but there was no significant difference in mortality in the intensive care unit (ICU) (RR 1.24, 95% CI 0.45–3.41)121,124 or in hospital mortality.121 The generalizability of these trial results beyond specialized centres is uncertain because for all of the patients, there was a high risk that noninvasive positive-pressure ventilation would fail and they would need intubation.
Continuous positive airway pressure for patients with exacerbation of COPD
We did not identify any RCTs investigating continuous positive airway pressure by mask for patients with COPD.
Statements
-
We recommend the use of noninvasive positive-pressure ventilation in addition to usual care in patients who have a severe exacerbation of COPD (pH < 7.35 and relative hypercarbia) (grade 1A recommendation).
-
We suggest that heliox not be routinely used in patients with severe exacerbation of COPD who are receiving noninvasive positive-pressure ventilation (grade 2C recommendation).
-
We make no recommendation about the use of noninvasive positive-pressure ventilation versus intubation and conventional mechanical ventilation in patients who have a severe exacerbation of COPD that requires ventilator support, because of insufficient evidence.
-
We make no recommendation about the use of continuous positive airway pressure by mask in patients who have a severe exacerbation of COPD, because of a lack of RCTs.
Exacerbation of asthma
Three small RCTs on the use of noninvasive positive-pressure ventilation in patients with exacerbation of asthma have been published.34–36 One trial, which cited selection bias in patient enrolment leading to a small sample size (n = 35), found no differences in outcomes.34 The second trial was a single-centre trial of 33 patients that used sham noninvasive ventilation in the control group.35 None of the patients were intubated, and all survived the hospital stay. However, there was a more rapid improvement in the forced expiratory volume in the first second (FEV1), and fewer patients required admission to hospital (3/17 v. 10/16; p = 0.013). The sham therapy may have disadvantaged the control group by increasing the work of breathing. Finally, in a recent three-arm trial (n = 40) comparing two pressure levels of noninvasive positive-pressure ventilation with oxygen therapy alone, a greater reduction in dyspnea and a greater increase in FEV1 were reported for the group that received noninvasive ventilation.36 Importantly, the patients in the latter two RCTs did not have severe exacerbation of asthma (none required endotracheal intubation). Overall, the quality and quantity of the evidence were low, and there were very few outcome events in any of the studies. We did not identify any RCTs on the use of continuous positive airway pressure in this population.
Statements
-
We make no recommendation about the use of noninvasive positive-pressure ventilation in patients who have an exacerbation of asthma, because of insufficient evidence.
-
We make no recommendation about the use of continuous positive airway pressure by mask in patients who have an exacerbation of asthma, because of a lack of RCTs.
Cardiogenic pulmonary edema
The use of noninvasive ventilation for patients with cardiogenic pulmonary edema and acute respiratory failure has been studied extensively. We identified 20 relevant RCTs.42–46,48–51,53–57,59,61,62,64–66 These trials examined the use of noninvasive positive-pressure ventilation plus usual therapy versus usual therapy alone,48,49,53 continuous positive airway pressure by mask plus usual therapy versus usual therapy alone,42–44,46,51,55,66 continuous positive airway pressure by mask plus usual therapy versus noninvasive positive-pressure ventilation plus usual therapy45,54,57,59,61,62 or all three of these treatments.50,56,64,65 Before publication of a recent large RCT,65 which accounted for 40% of all patients who have been studied in RCTs of continuous positive airway pressure for cardiogenic pulmonary edema and 70% of patients receiving noninvasive positive-pressure ventilation for this indication, five separate systematic reviews162–166 had consistently demonstrated a significant reduction in endotracheal intubation with both types of noninvasive ventilation. When this large trial was included in the meta-analysis, there was a trend toward reduction in endotracheal intubation with noninvasive positive-pressure ventilation (RR 0.55, 95% CI 0.29–1.03) and a significant reduction in endotracheal intubation with continuous positive airway pressure by mask (RR 0.42, 95% CI 0.28–0.63) relative to oxygen alone.
Notably, this recent large trial65 differed from most others, in that patients who met the criteria for treatment failure were allowed to cross over to one of the two forms of noninvasive ventilation rather than undergoing insertion of an endotracheal tube. Although the rate of endotracheal intubation did not differ between arms, a much higher proportion of patients in the oxygen arm crossed over to noninvasive ventilation (56/367 patients v. 5/346 from continuous positive airway pressure to noninvasive positive-pressure ventilation and 12/356 from noninvasive positive-pressure ventilation to continuous positive airway pressure). In addition, the proportion of patients who did not remain in their assigned treatment arm because of respiratory distress was significantly higher in the oxygen-only arm (8.4% v. 3.4% for noninvasive positive-pressure ventilation and 1.4% for continuous positive airway pressure; p < 0.001). Because of the potential for crossover in this trial to confound the outcomes of endotracheal intubation and hospital mortality, we considered the outcome of “treatment failure” in developing our final guideline statement. Pooled treatment failure for all trials was significantly lower for both noninvasive positive-pressure ventilation (RR 0.36, 95% CI 0.25–0.51) and continuous positive airway pressure (RR 0.23, 95% CI 0.17–0.32). Including all trials, there was a trend toward lower hospital mortality (for noninvasive positive-pressure ventilation, RR 0.84, 95% CI 0.63–1.13; for continuous positive airway pressure, RR 0.73, 95% CI 0.51–1.05).
The literature on continuous positive airway pressure by mask has evolved over the past two decades, during which time co-interventions for cardiogenic pulmonary edema that may affect mortality, including use of acute revascularization techniques, has changed. Inclusion and exclusion criteria varied among the trials. All 15 trials from 2000 to 2009 excluded patients with cardiogenic shock. In addition, 9 of these 15 trials, and 8 of the 10 trials in the period 2005 through 2009, also excluded patients who required acute coronary revascularization48,53,56,61,62,64,65 or who had acute coronary syndrome.54,59 Hence, it is difficult to extend recommendations to these specific populations. An early RCT suggested that noninvasive positive-pressure ventilation was associated with a greater risk of myocardial infarction than was continuous positive airway pressure,45 but many subsequent RCTs have not confirmed this finding.50,54,56,57,59,61,62,64,65
Statement
-
We recommend the use of either noninvasive positive-pressure ventilation or continuous positive airway pressure by mask in patients who have cardiogenic pulmonary edema and respiratory failure in the absence of shock or acute coronary syndrome requiring urgent coronary revascularization (grade 1A recommendation).
Acute lung injury
Three RCTs have assessed noninvasive ventilation in patients with acute lung injury, including acute respiratory distress syndrome. Two trials on noninvasive positive-pressure ventilation included subgroups of patients with acute lung injury, but the numbers of events and patients were too small to allow interpretation (total of 30 patients in the two trials39,40). One RCT was published on the use of continuous positive airway pressure plus usual therapy versus usual therapy alone in patients with acute hypoxemic respiratory failure.37 One-third of these patients had associated acute or chronic cardiac disease, although the relative proportion of each and the precise definition of cardiac disease were not reported. In the subgroup of patients who did not have acute or chronic cardiac disease, the addition of continuous positive airway pressure did not affect endotracheal intubation (RR 0.86, 95% CI 0.54–1.37) or hospital mortality (RR 1.04, 95% CI 0.6–1.7) but was associated with more adverse events (including four cardiac arrests), which suggested a potential for harm.
Statements
-
We make no recommendation about the use of noninvasive positive-pressure ventilation in patients who have acute lung injury, because of a lack of RCTs.
-
We recommend that continuous positive airway pressure not be used in patients who have acute lung injury (grade 1C recommendation).
Severe community-acquired pneumonia in patients without COPD
Two RCTs studied noninvasive positive-pressure ventilation in patients with community-acquired pneumonia and acute hypoxemic respiratory failure but no prior history of COPD. In one trial, patients with COPD were included, and results for patients who did and did not have this condition were reported separately.38 In the subgroup of patients who did not have COPD (n = 33), the addition of noninvasive positive-pressure ventilation did not reduce endotracheal intubation (6/16 v. 8/17 in control group) or hospital mortality (6/16 v. 4/17). In the other trial, which involved patients with acute hypoxemic respiratory failure but no underlying COPD, there was benefit (reduced endotracheal intubation and ICU mortality) for the subgroup of patients (n = 34) with severe community-acquired pneumonia.39 For these two small subgroups (67 patients in total) from two RCTs, the results were conflicting regarding the addition of noninvasive positive-pressure ventilation to usual therapy for patients with severe community-acquired pneumonia but no prior history of COPD (pooled results: RR 0.54, 95% CI 0.24–1.17 for endotracheal intubation and RR 0.70, 95% CI 0.13–3.63 for hospital mortality). We did not identify any RCTs on the use of continuous positive airway pressure for patients with severe community-acquired pneumonia and without COPD.
Statements
-
We make no recommendation about the use of noninvasive positive-pressure ventilation in patients who have severe community-acquired pneumonia and no prior history of COPD, because of insufficient evidence.
-
We make no recommendation about the use of continuous positive airway pressure by mask in patients who have severe community-acquired pneumonia and no prior history of COPD, because of a lack of RCTs.
Chest trauma
There were two RCTs on the use of continuous positive airway pressure in patients with severe chest trauma: one for prevention and one for treatment of respiratory failure in patients without an endotracheal tube at the time of presentation.137,138 No RCTs have evaluated noninvasive positive-pressure ventilation for these indications. In the prevention trial, which included all patients with severe chest trauma regardless of whether they had respiratory distress,137 co-interventions differed between treatment groups, the noninvasive ventilation group receiving epidural analgesia and the control group receiving systemic analgesia. In addition, the comparator to continuous positive airway pressure in this trial was intubation and mechanical ventilation. Hence, although the group receiving noninvasive ventilation had a shorter length of stay in the ICU and in hospital, the design of the study did not reflect current clinical practice, as endotracheal tubes are not usually inserted prophylactically for patients similar to those in the control group. The RCT involving patients with respiratory failure reported a lower mortality rate (2/22 v. 7/21; p < 0.01) for the group receiving continuous positive airway pressure by mask,138 but the small total number of patients (n = 43) and the single-centre design raise concerns regarding generalizability.
Statements
-
We make no recommendation about the use of noninvasive positive-pressure ventilation or continuous positive airway pressure in patients who have chest trauma without respiratory distress, because of a lack of RCTs.
-
We make no recommendation about the use of noninvasive positive-pressure ventilation in patients who have chest trauma and respiratory distress, because of a lack of RCTs.
-
We make no recommendation about the use of continuous positive airway pressure by mask in patients who have chest trauma and respiratory distress, because of insufficient evidence.
Immunosuppression
We identified two RCTs that evaluated the effect of noninvasive positive-pressure ventilation added to usual therapy in patients with immunosuppression (defined as receiving immunosuppressive therapy for solid organ or bone marrow transplant or immunosuppressive chemotherapy) who also had acute respiratory distress or failure.40,41 Both studies (total 92 patients) showed a reduction in endotracheal intubation (RR 0.46, 95% CI 0.22–0.95) and a reduction in hospital mortality (RR 0.62, 95% CI 0.43–0.90). However, members of the panel questioned the generalizability of the results from centres with highly experienced staff to other centres. No RCTs were found on the use of continuous positive airway pressure in this population.
Statements
-
We suggest that noninvasive positive-pressure ventilation be used for immunosuppressed patients who have acute respiratory failure (grade 2B recommendation).
-
We make no recommendation about the use of continuous positive airway pressure by mask in immunosuppressed patients who have acute respiratory failure, because of a lack of RCTs.
Bronchoscopy in patients with hypoxemia
Patients with hypoxemia who undergo bronchoscopy are at risk of respiratory deterioration and need for endotracheal intubation. One RCT using noninvasive positive-pressure ventilation118 and a second using continuous positive airway pressure by mask119 suggested benefit in terms of oxygenation when these interventions were used during bronchoscopy. However, few patients were included (total of 56 for the two RCTs), and the impact on endotracheal intubation (0.28, 95% CI 0.05–1.64) was inconsistent.
Statement
-
We make no recommendation about the use of either noninvasive positive-pressure ventilation or continuous positive airway pressure in patients who have hypoxemia and who undergo bronchoscopy, because of insufficient evidence.
After intubation
Adjunct to early liberation
Nine RCTs found that noninvasive positive-pressure ventilation can be used as an alternative to continued invasive ventilation in patients who become clinically stable but still require ongoing ventilation (e.g., failure of spontaneous breathing trials but meeting criteria for control of pulmonary infection72,73,75,76,78–82) (RR 0.49, 95% CI 0.29–0.83 for hospital mortality). However, six of the nine trials included only patients who had exacerbations of COPD.72,75,78–81 Two of the remaining three RCTs included both patients without COPD and a large proportion of patients with COPD (58%76 and 76%73). The pooled effect on hospital mortality for these three studies was not significant (RR 0.52, 95% CI 0.15–1.85). The wide confidence intervals arose because of benefit in the two studies that had larger proportions of patients with COPD and no benefit in the single study that had fewer patients with COPD.
At this time, the literature supports use of noninvasive positive-pressure ventilation only in patients who have COPD because the minimal data on patients without this condition suggest no benefit. Because of methodologic limitations in these studies, as well as important safety, feasibility and cost limitations, the use of noninvasive positive-pressure ventilation for this indication requires both considerable expertise and the ability to closely monitor the patients, because urgent reintubation may be required.
No RCTs have been published on the use of continuous positive airway pressure by mask in this population.
Statements
-
We suggest that noninvasive positive-pressure ventilation be used to facilitate early liberation from mechanical ventilation in patients who have COPD, but only in centres that have expertise in this type of therapy (grade 2B recommendation).
-
We make no recommendation about the use of noninvasive positive-pressure ventilation to facilitate early liberation from mechanical ventilation in patients who do not have COPD, because of insufficient evidence.
-
We make no recommendation about the use of continuous positive airway pressure by mask to facilitate early liberation from mechanical ventilation, because of a lack of RCTs.
After planned extubation
Noninvasive ventilation may prevent the need for reintubation if applied immediately after planned extubation. One small, low-quality trial assessed noninvasive positive-pressure ventilation after extubation and found no benefit.83 The study had limited generalizability because it included a high proportion of patients who had performed the extubation themselves. Members of the panel expressed concern regarding the feasibility and cost of noninvasive positive-pressure ventilation in this setting.
Four recent RCTs suggested benefit from noninvasive positive-pressure ventilation after extubation in patients who were at high risk of deterioration.84–87 High-risk patients were defined differently among the RCTs: 1) age greater than 65 years, cardiac failure as the cause of intubation or Acute Physiology and Chronic Health Evaluation (APACHE) II score greater than 12 at the time of extubation;84 2) more than one of the following: failure of consecutive weaning trials, chronic cardiac failure, arterial pressure of carbon dioxide greater than 45 mm Hg after extubation, more than one noncardiac comorbidity, weak cough or stridor after extubation not requiring immediate intubation;85 3) acute exacerbation of COPD86 or 4) history of chronic respiratory disease with ventilation for more than 48 hours and hypercapnia during the spontaneous breathing trial.87 Although the four trials defined higher risk differently, they reported consistent decreases in rates of reintubation (RR 0.42, 95% CI 0.25–0.70) and ICU mortality (RR 0.35, 95% CI 0.16–0.78) but less benefit in terms of hospital mortality (RR 0.66, 95% CI 0.42–1.04).
Although these results are promising, we suggest that noninvasive positive-pressure ventilation be used in this setting only where there is expertise in both this type of therapy and invasive airway management. We did not identify any RCTs investigating the role of continuous positive airway pressure by mask in this population.
Statements
-
We suggest that noninvasive positive-pressure ventilation be used after planned extubation in patients who are considered to be at high risk of recurrent respiratory failure, but only in centres that have expertise in this type of therapy (grade 2B recommendation).
-
We suggest that noninvasive positive-pressure ventilation not be used after planned extubation in patients who are considered to be at low risk of respiratory failure (grade 2C recommendation).
-
We make no recommendation about the use of continuous positive airway pressure by mask after planned extubation, because of a lack of RCTs.
Respiratory failure after extubation
Noninvasive ventilation has been used in patients who experience respiratory failure during the postextubation period (generally considered to be 48–72 hours after extubation). Two RCTs have evaluated the addition of noninvasive positive-pressure ventilation to usual care compared with usual care alone for postextubation respiratory failure, and both found no reduction in rates of reintubation (RR 1.03, 95% CI 0.84–1.25).70,71 The larger, multicentre trial suggested potential harm, finding a higher rate of ICU mortality among patients receiving noninvasive positive-pressure ventilation (28/114 v. 15/107, p = 0.048).71 Notably, the earlier study excluded patients with COPD after the first of 4 years of recruitment,70 and in the second study too few patients with COPD were available to be analyzed separately.71 Others have raised concerns that noninvasive positive-pressure ventilation was not optimally applied in either trial.167,168 At present, safety concerns (consequences of delayed reintubation and increased mortality) and issues of feasibility (centre expertise) preclude recommendation of routine use of noninvasive positive-pressure ventilation in this patient population.
We did not identify any RCTs evaluating use of continuous positive airway pressure by mask in this population.
Statements
-
We suggest that noninvasive positive-pressure ventilation not be routinely used in patients who do not have COPD and who have postextubation respiratory failure (grade 2C recommendation).
-
We make no recommendation about the use of noninvasive positive-pressure ventilation in patients who have COPD and postextubation respiratory failure, because of insufficient evidence.
-
We make no recommendation about the use of continuous positive airway pressure by mask in patients who have postextubation respiratory failure, because of a lack of RCTs.
Postoperative setting
Noninvasive ventilation may be used in the postoperative setting to either prevent or treat respiratory failure. For prevention, noninvasive ventilation is added to usual care for all patients who undergo specified surgeries. For treatment, noninvasive ventilation is used only in patients who experience postoperative hypoxemia or early signs of respiratory failure. The potential benefit of these interventions when applied as part of routine postoperative care (i.e., for prevention of respiratory failure) may depend on the baseline risk of respiratory failure. This risk can arise from variables related to either the patient or the surgical procedure itself.169,170 For example, one trial of patients undergoing abdominal surgery110 included only high-risk patients as defined by a previously validated risk-scoring system, which included body mass index, the presence of cardiac comorbidities, COPD and an oncologic diagnosis.170 Unfortunately, other than this trial110 the literature comprises only studies that do not specify the risk level of the included patients.
Prevention of respiratory failure
For the purposes of this guideline we considered high-risk surgical procedures to include major vascular procedures, such as elective abdominal vascular surgery92 and elective thoracoabdominal vascular surgery.94 The average age of patients enrolled in these trials was about 65 years, which represents an additional risk factor for postoperative respiratory failure, and the risk due to patients’ comorbidities was not specifically defined.169 Although cardiac and thoracic surgery may be considered high-risk procedures, the actual incidence of respiratory failure with these types of surgery is relatively low.89,91,95,97–99
For the purposes of these guidelines, low risk was necessarily defined by the surgical procedure alone, as no trials specified that their respective patient populations were uniformly at low risk. As such, trials of low-risk surgical procedures might have included patients with comorbidities that would place them in either high- or low-risk categories. Trials of the use of noninvasive ventilation in patients who underwent low-risk surgical procedures were generally small and reported only physiologic outcomes or found no difference in endotracheal intubation, hospital mortality or length of stay.89,91,95,97–99,102–107,109,111 The only exception was a recent large trial of continuous positive airway pressure by mask in 468 patients after cardiac surgery.101 There was a reduction in a composite end point of pulmonary complications (arterial pressure of oxygen/fraction of inspired oxygen < 100, pneumonia or reintubation) and readmission to the ICU or an intermediate care unit. The authors found no differences in reintubation, mortality or length of stay. Although none of the trials involving patients who underwent low-risk surgery specified the patients’ risk level in terms of associated comorbidities, the lack of evidence of effectiveness of either noninvasive positive-pressure ventilation or continuous positive airway pressure in these heterogeneous populations implies a lack of evidence for low-risk patients.
One trial evaluated noninvasive positive-pressure ventilation in patients considered at high risk of respiratory failure (because of associated comorbidities) who underwent abdominal surgery, considered a lower-risk procedure.110 The authors reported a nonsignficant reduction in reintubation during the postoperative period (0/21 v. 2/20). No trials evaluated continuous positive airway pressure for a specifically defined high-risk group of patients undergoing low-risk surgery.
Two RCTs evaluated continuous positive airway pressure by mask in addition to usual therapy or intermittent breathing exercises in patients who underwent high-risk surgical procedures, including elective open repair of abdominal aortic aneurysm or thoracoabdominal vascular surgery.92,94 Neither trial specified the risk level of the patient population. Pooled results from these two trials showed a trend toward reduced rate of endotracheal intubation (RR 0.23, 95% CI 0.05–1.03) but no difference in hospital mortality.
For prevention of respiratory failure after low-risk surgical procedures:
-
We make no recommendation about the use of either noninvasive positive-pressure ventilation or continuous positive airway pressure by mask in low-risk patients after low-risk surgery, because of insufficient evidence.
-
We make no recommendation about the use of noninvasive positive-pressure ventilation to prevent respiratory failure after cardiac surgery, because of a lack of RCTs, and no recommendation about the use of continuous positive airway pressure by mask in this setting, because of insufficient evidence.
-
We make no recommendation about the use of either noninvasive positive-pressure ventilation or continuous positive airway pressure by mask in patients at high risk (because of associated comorbidity) to prevent respiratory failure after low-risk surgical procedures, because of insufficient evidence.
For prevention of respiratory failure after high-risk surgical procedures:
-
We make no recommendation about the use of noninvasive positive-pressure ventilation to prevent respiratory failure after high-risk surgical procedures, because of a lack of RCTs.
-
We make no recommendation about the use of continuous positive airway pressure by mask to prevent respiratory failure after high-risk surgical procedures, because of insufficient evidence.
Treatment of respiratory failure
Two RCTs90,112 assessed the use of noninvasive ventilation to treat respiratory failure in the postoperative setting. One trial examined continuous positive airway pressure by mask in patients who experienced hypoxemia after abdominal surgery involving visceral exposure times exceeding 90 minutes,112 and the other evaluated noninvasive positive-pressure ventilation in patients who experienced hypoxemia and increased work of breathing after lung resection.90 Both RCTs found a reduction in rate of endotracheal intubation (RR 0.14, 95% CI 0.03–0.62112 and RR 0.42, 95% CI 0.17–1.00,90 respectively). Patients who had underlying congestive heart failure and valvular heart disease were excluded from the abdominal surgery study.112 The current evidence is therefore limited, as few patients were enrolled in these studies and few experienced outcome events.
Statements
-
We make no recommendation about the use of noninvasive positive-pressure ventilation in patients who have respiratory failure after abdominal surgery, because of a lack of RCTs.
-
We suggest that continuous positive airway pressure be used in patients who have respiratory failure after abdominal surgery (grade 2C recommendation).
-
We suggest that noninvasive positive-pressure ventilation be used in patients who have respiratory failure after lung-resection surgery (grade 2C recommendation).
-
We make no recommendation about the use of continuous positive airway pressure by mask in patients who have respiratory failure after lung-resection surgery, because of a lack of RCTs.
Optimal use of noninvasive ventilation
All of the preceding statements have referred to the use of either noninvasive positive-pressure ventilation or continuous positive airway pressure by mask in specific populations. Several additional questions remain unanswered: Which interface is optimal: nasal, oronasal mask (mask covering nose and mouth), full face mask (mask covering entire face) or helmet? Which mode of noninvasive positive-pressure ventilation is optimal: pressure support ventilation, volume control ventilation or proportional assist ventilation? Is there a superior model of ventilator or optimal triggering? What is the role of humidification?
We searched the literature to address these questions and found that most studies were uncontrolled or used a randomized crossover rather than parallel design. We did not identify any trials on optimal triggering, ventilator type or use of humidification. Therefore, our statements in this area are limited.
Choice of interface
Two RCTs, with parallel design, compared use of an oronasal mask with use of a nasal mask in patients who had acute respiratory failure.125,126 Although there was no difference in endotracheal intubation or mortality, the oronasal mask was better tolerated. One small RCT (34 patients) compared the use of a full face mask with use of an oronasal mask.127 The study investigators reported better tolerance of the oronasal mask, but the small number of patients precludes any strong inferences.
Statements
-
We suggest the use of an oronasal mask rather than a nasal mask in patients who have acute respiratory failure and who are receiving noninvasive positive-pressure ventilation (grade 2C recommendation).
-
We make no recommendation about the use of an oronasal mask versus full face mask for noninvasive positive-pressure ventilation in patients who have acute respiratory failure, because of insufficient evidence.
-
We make no recommendation about the use of an oronasal mask versus nasal or full face mask in patients who have acute respiratory failure and who are receiving continuous positive airway pressure by mask, because of a lack of RCTs.
Preferred mode for noninvasive positive-pressure ventilation
Two parallel-design RCTs compared the use of pressure support ventilation and proportional assist ventilation.148,149 Neither found differences in rates of intubation or mortality. In one RCT, different ventilator models were used in each of the two arms,149 a difference that may confound the better tolerance found in the group receiving proportional assist ventilation. The second RCT, which used the same model of ventilator to compare the two modes of ventilation, also reported better comfort and tolerance with proportional assist ventilation.148 Important methodologic issues limit inferences from these trials. Moreover, feasibility and cost considerations associated with proportional assist ventilation preclude the widespread use of this mode of noninvasive positive-pressure ventilation.
Statement
-
We make no recommendation about the use of proportional assist ventilation versus pressure support ventilation in patients who are receiving noninvasive positive-pressure ventilation for acute respiratory failure, because of insufficient evidence.
Implementation
Implementation of these guidelines may require clinician education, additional health care personnel, organizational change or additional resources (equipment or beds with cardiopulmonary monitoring) to ensure safe and appropriate application of noninvasive positive-pressure ventilation and continuous positive airway pressure. Timely endotracheal intubation may be required if noninvasive ventilation fails. Strategies for the implementation of these guidelines should be developed for each relevant clinician group (physicians in different clinical areas and with different levels of training and expertise, respiratory therapists and bedside nurses).171,172
Updating
We plan to update this guideline every four years (sooner if sufficient new data from RCTs become available).
Discussion
The guideline statements that we have developed for the use of noninvasive ventilation for patients in the acute care setting build on previous consensus conference statements152,153 and guidelines154,156,157 published by other societies. Our guideline is based on the most current, systematic and comprehensive review of RCTs in this area. We used GRADE methodology in our critical appraisal of the quality of the RCT evidence with respect to study validity. Moreover, we incorporated assessments of safety, feasibility and cost in developing our recommendations. Where there was insufficient RCT evidence to make a recommendation, we have indicated “no recommendation,” which means that the evidence neither favours nor opposes the use of noninvasive ventilation in those clinical situations.
During the development of this guideline, we enlisted the help of a multidisciplinary panel of critical care physicians, pulmonary physicians and respiratory therapists, including methodologists with expertise in evidence-based medicine and guideline development. To prepare for using the GRADE methodology, we educated the panel members in advance about use of this systematic and transparent method of grading the evidence and developing statements. After using a consensus process to develop our final statements, we obtained an anonymous agreement score for each statement from each panel member. In addition, this guideline was critiqued by external reviewers from two professional associations and by two international experts in noninvasive positive-pressure ventilation research and application.
This guideline does not address the use of noninvasive ventilation in some clinical settings where it is currently used because of a lack of RCTs for either intervention. Some examples include use of noninvasive ventilation for acute respiratory failure in patients with cystic fibrosis or HIV/AIDS or in patients who have declined intubation and resuscitation. Although there is no RCT-level evidence to support effectiveness in these settings, the absence of such evidence does not preclude benefit. For these and other areas for which noninvasive ventilation is currently used, these technologies may be considered as possible treatment options. In some settings, experimental studies may be ethically challenging, and RCTs may never be conducted.
Other guidelines
The literature review and consensus processes used to arrive at the recommendations presented here are similar to those used for the British Thoracic Society guideline published in 2002,154 and the scope and recommendations of our guidelines closely resemble those of the British guideline. However, for our guideline, we considered more recent literature (an additional 58 RCTs), used RCT evidence alone and used the GRADE methodology. The British Thoracic Society guideline includes recommendations for patients who have chest wall deformity or neuromuscular disease, decompensated obstructive sleep apnea, and cystic fibrosis or bronchiectasis, but recommendations for these patient groups were based on non-RCT evidence. Our guideline addresses several additional clinical settings: postoperative care, weaning from conventional mechanical ventilation, transition to spontaneous breathing and respiratory failure after extubation; it also includes more thorough review and discussion of the topic of immunocompromised patients. The British Thoracic Society guideline describes optimal application of and weaning from noninvasive positive-pressure ventilation, including monitoring requirements and guidance on setting up and running a noninvasive positive-pressure ventilation service. Our recommendations are limited to areas for which there is RCT evidence to make recommendations regarding the interface and modes of noninvasive positive-pressure ventilation.
The Indian Society of Critical Care Medicine published guidelines in 2006,156 using a more limited search strategy. In addition, individual panel members summarized each area of interest before meeting. The 10-member panel that prepared those guidelines used a consensus approach to reach their recommendations but did not use the GRADE methodology. They incorporated non-RCT evidence into weak recommendations for settings where we did not make any recommendation, such as severe community-acquired pneumonia. They also had a broader focus, including application of noninvasive positive-pressure ventilation, monitoring, location of use and initiating a service of this type. Although they made similarly strong recommendations for noninvasive positive-pressure ventilation in patients with cardiogenic pulmonary edema and exacerbation of COPD, their other recommendations tended to be stronger than those in our guideline. We also considered 24 new RCTs that were published since the release of these guidelines.
The most recent guidelines previous to ours were published in 2008 by a group of 28 experts from 12 German medical societies.157 Their recommendations are similar to ours for patients who have COPD or cardiogenic pulmonary edema. However, we differed from the German guidelines in our recommendations about use of noninvasive ventilation for immunocompromised patients, patients who are being weaned from mechanical ventilation and those who experience respiratory failure after extubation. The German guidelines made a strong grade A recommendation (where grade of recommendation was based on the study design) for use of noninvasive positive-pressure ventilation for these groups of patients, but we were more conservative in our level of recommendation because of lesser quantity and/or quality of evidence to support the use of this type of therapy in these settings. Importantly, because there were no RCTs, we did not include an assessment of noninvasive ventilation for patients who have declined invasive mechanical ventilation. In contrast, the German guidelines recommend (level B recommendation) that noninvasive positive-pressure ventilation is an acceptable alternative. Furthermore, we limited our guidelines to adults, whereas the German guidelines included recommendations for children as well (level C).
Limitations
In critiquing this guideline using the AGREE Critical Appraisal Instrument, we found that it meets 20 of the 23 items (organized within 6 domains).161 Specifically, we fulfilled only one of the three criteria within the “stakeholder involvement domain” of the instrument, because we did not include emergency department physicians or nurses in the development process, nor did we take patients’ perspectives into account. We did not conduct a formal economic evaluation or incorporate existing economic evaluations of noninvasive ventilation into the development of this guideline. We pretested the guidelines informally among the target users. However, the development of formal strategies for implementing the guidelines and the testing of those strategies in acute and critical care settings are the subject of ongoing research. The aim of this research is to identify the most effective and most cost-effective strategies adapted to the local level.
Knowledge gaps
There are several topics for which we could not make recommendations because of insufficient RCT evidence. Areas we suggest for future research include the use of noninvasive ventilation for severe community-acquired pneumonia, asthma, acute lung injury and chest trauma; for early liberation from mechanical ventilation; after planned extubation; and in the postoperative setting. Important unresolved technical issues that require further research include the optimal ventilator, mode of ventilation, trigger, interface and level of humidification.
Conclusion
Noninvasive ventilation is an important option in the management of patients who are at risk of or who have respiratory failure in the acute care setting. However, there is considerable variability in its use.173,174 These guidelines provide a means to reduce variations in practice while applying evidence-based recommendations for care of these acutely ill patients. The guidelines were developed within the context of the Canadian health care system, but they are founded on RCTs from around the world. On the basis of these characteristics, we believe that these recommendations are generalizable and potentially useful to all clinicians who care for patients who are at risk of or who have acute respiratory failure in the acute care setting.
-
Noninvasive positive-pressure ventilation should be the first option for ventilatory support for patients with either a severe exacerbation of chronic obstructive pulmonary disease (COPD) or cardiogenic pulmonary edema.
-
Continuous positive airway pressure delivered by mask appears to be just as effective as noninvasive positive-pressure ventilation for patients with cardiogenic pulmonary edema.
-
Patients with acute respiratory distress or hypoxemia, either in the postoperative setting or in the presence of immunosuppression, can be considered for a trial of noninvasive positive-pressure ventilation.
-
Patients with COPD can be considered for a trial of early extubation to noninvasive positive-pressure ventilation in centres with extensive experience in the use of noninvasive positive-pressure ventilation.
Key points
Acknowledgements
The authors are grateful to Roman Jaeschke for his advice regarding GRADE methodology and to Dr. Stefano Nava for his input. They thank the Canadian Critical Care Trials Group and the Canadian Critical Care Society for their support in developing this guideline.
Footnotes
-
See related commentary by Bersten, page 293
-
An abridged version of this article appeared in the February 22, 2011 issue of CMAJ
-
Competing interests: Sean Keenan received an honorarium from Philips/Respironics Inc. to give a talk based on these guidelines. No competing interests declared by the other authors.
-
This article has been peer reviewed.
-
Members of the Canadian Critical Care Trials Group/Canadian Critical Care Society Noninvasive Ventilation Guidelines Group: Dr. Sean P. Keenan, Department of Critical Care Medicine, Royal Columbian Hospital, University of British Columbia, Vancouver, BC; Dr. Tasnim Sinuff, Department of Critical Care Medicine, Sunnybrook Health Sciences Centre, and Interdepartmental Division of Critical Care Medicine, University of Toronto, Toronto, Ont.; Dr. Karen E.A. Burns, Interdepartmental Division of Critical Care Medicine, the Keenan Research Centre and the Li Ka Shing Knowledge Institute, St. Michael’s Hospital, Toronto, Ont.; Dr. John Muscedere, Department of Medicine, Queen’s University and Kingston General Hospital, Kingston, Ont.; Dr. Jim Kutsogiannis, Division of Critical Care Medicine, University of Alberta, Edmonton, Alta.; Dr. Sangeeta Mehta, Department of Medicine and the Interdepartmental Division of Critical Care Medicine, Mount Sinai Hospital, University of Toronto, Toronto, Ont.; Dr. Deborah J. Cook, Department of Medicine, McMaster University, Hamilton, Ont.; Dr. Najib Ayas, Department of Medicine, University of British Columbia, Vancouver, BC; Dr. Neill K.J. Adhikari, Interdepartmental Division of Critical Care Medicine, University of Toronto, and the Sunnybrook Research Institute, Toronto, Ont.; Lori Hand, Department of Clinical Epidemiology and Biostatistics, McMaster University, Hamilton, Ont.; Dr. Damon C. Scales, Interdepartmental Division of Critical Care Medicine, University of Toronto, and the Sunnybrook Research Institute, Toronto, Ont.; Rose Pagnotta, Department of Respiratory Therapy, Royal Inland Hospital, Kamloops, BC; Lynda Lazosky, Division of Critical Care Medicine, St. Paul’s Hospital, Vancouver, BC; Dr. Graeme Rocker, Division of Respirology, Queen Elizabeth II Health Sciences Centre and Dalhousie University, Halifax, NS; Dr. Sandra Dial, Critical Care Medicine, Sir Mortimer B. Davis Jewish General Hospital, and the Respiratory Epidemiology and Clinical Research Unit, Montreal Chest Institute, McGill University, Montréal, Que.; Dr. Kevin Laupland, Department of Critical Care Medicine, University of Calgary, Calgary, Alta; Dr. Kevin Sanders, Department of Medicine, North York Hospital, Toronto, Ont.; and Dr. Peter Dodek, Division of Critical Care Medicine, Department of Medicine, St. Paul’s Hospital and University of British Columbia, Vancouver, BC
-
Contributors: Sean Keenan and Tasnim Sinuff are joint first authors on this paper. They led the process of guideline development, performing initial and subsequent selection of trials for review, coordinating distribution of material for review and face-to-face meetings and leading manuscript writing and submission. All of the authors were involved in the perception and design of the guideline development project and took part in a review of the GRADE process of guideline development before meetings. All of the authors except for Deborah Cook and Peter Dodek were involved in reviewing and evaluating selected trials and presenting summaries of these trials at the meetings. All authors were involved in discussions of the literature presented and in drafting initial statements following the GRADE process during the meetings. All of the authors contributed to the writing and revising of the manuscript and approved the final version submitted for publication.
-
Funding: This work was funded by a grant from the Canadian Institutes of Health Research (CIHR) and an unrestricted grant from Philips/Respironics Inc., Murrysville, Pa. Neither funding body was involved in any stage of the development of the guidelines or the preparation of the manuscript.
Tasnim Sinuff and Karen Burns are supported by a CIHR Research Clinician Scientist Award. Deborah Cook is a Canada Research Chair of the CIHR. Damon Scales has received a CIHR New Investigator Award