In the midst of pandemic (H1N1) 2009 in our region, a five-year-old girl with mild asthma presented to the emergency department of a children’s hospital in acute respiratory distress. She had an 11-day history of cough, rhinorrhea and progressive chest discomfort. She was otherwise healthy, with no history of severe respiratory illness, prior hospital admissions or immunocompromise. Outside of infrequent use of salbutamol, she was not taking any medications, and her routine childhood immunizations, including conjugate pneumococcal vaccine, were up to date. She had not received the pandemic influenza vaccine because it was not yet available for her age group.
The patient had been seen previously at a community health centre a week into her symptoms, and a chest radiograph had shown perihilar and peribronchial thickening but no focal consolidation, atelectasis or pleural effusion. She had then been reassessed 24 hours later at an influenza assessment centre and empirically started on oseltamivir. Two days later, with the onset of vomiting, diarrhea, fever and progressive shortness of breath, she was brought to the emergency department of the children’s hospital. On examination, she was in considerable distress; her heart rate was 170 beats/min, her respiratory rate was 60 breaths/min and her blood pressure was 117/57 mm Hg. Her oxygen saturations on room air were consistently 70%. On auscultation, she had decreased air entry to the right side with bronchial breath sounds. Repeat chest radiography showed almost complete opacification of the right hemithorax, air bronchograms in the middle and lower lobes, and minimal aeration to the apex. This was felt to be in keeping with whole lung consolidation and parapneumonic effusion. The left lung appeared normal.
Blood tests done on admission showed a hemoglobin level of 122 (normal 110–140) g/L, a leukocyte count of 1.5 (normal 5.5–15.5) ×109/L (neutrophils 11% [normal 47%] and bands 19% [normal 5%]) and a platelet count of 92 (normal 217–533) × 109/L. Results of blood tests were otherwise unremarkable. Venous blood gas had a pH level of 7.32 (normal 7.35–7.42), partial pressure of carbon dioxide of 43 (normal 32–43) mm Hg, a base deficit of 3.6 (normal −2 to 3) mmol/L, and a bicarbonate level of 21.8 (normal 21–26) mmol/L. The initial serum creatinine level was 43.0 (normal < 36) μmol/L and the urea level was 6.5 (normal 2.0–7.0) mmol/L, with no clinical evidence of renal dysfunction.
Given the patient’s profound increased work of breathing, she was admitted to the intensive care unit (ICU), where intubation was required because of her continued decline over the next 24 hours. Blood cultures taken on admission were negative. Nasopharyngeal aspirates were negative on rapid respiratory viral testing, but antiviral treatment for presumed pandemic (H1N1) influenza was continued given her clinical presentation, the prevalence of pandemic influenza in the community and the low sensitivity of the test in the range of only 62%.1 Viral cultures were not done. Empiric treatment with intravenous cefotaxime (200 mg/kg/d) and vancomycin (40 mg/kg/d) was started in the ICU for broad antimicrobial coverage, including possible methicillin-resistant Staphylococcus aureus or other staphylococcal infection. When Streptococcus pneumoniae infection was later confirmed, the vancomycin was discontinued and amoxicillin was started. Given the patient’s dramatic respiratory distress and life-threatening status, off-label use of intravenous zanamivir was started.
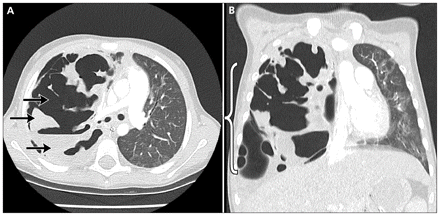
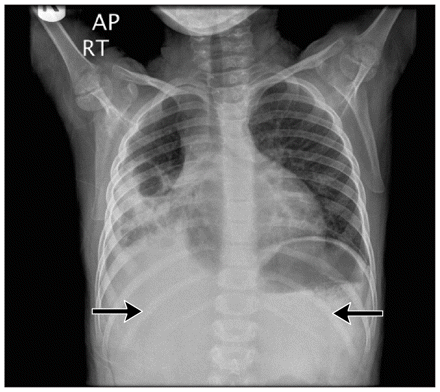
Twenty-four hours after ICU admission, our patient’s clinical status stabilized, with slow improvement over the subsequent days. Given marked clinical improvement and minimal ventilatory pressures, her endotracheal tube was successfully removed 10 days after admission. Radiographic imaging done on the same day showed continued aeration of the left lung but the development of a large right-sided pneumatocele, marked atelectasis and a mild to moderate parapneumonic effusion (Figure 1). Computed tomography (CT) done later that day showed a dramatic necrotizing process in the right middle lobe with multiple large pneumatoceles and secondary compression of the right upper and lower lobes (Figure 2). The CT scan was also suggestive of a loculated basal pneumothorax.
Portable chest radiograph of a five-year-old girl with co-infection with pandemic (H1N1) 2009 virus and Streptococcus pneumoniae, showing multiple right pneumatoceles (bracket), opacification in the right middle lobe (black arrow) and depression of the right hemidiaphragm (white arrow).
(A) Axial computed tomographic (CT) image showing necrosis of the right middle lobe with pneumatoceles and three pleural pockets (one air-filled, one fluid and one mixed; arrows). (B) Coronal CT image showing large pneumatoceles and necrosis of the right middle lobe (bracket).
Sixteen days after admission, given persistent fevers, tachypnea and oxygen requirements, a right-sided chest tube was inserted to assist with drainage of the pleural space. Cultures of the pleural fluid were negative for microbial growth, but polymerase chain reaction analysis using 16S rDNA gene sequencing was positive for S. pneumoniae. Serology for S. pneumoniae immunoglobulin G antibody was also subsequently found to be positive. The drained fluid consisted of both pus and necrotic material, in keeping with a necrotizing process. Of significant concern was the development of a bronchopleural fistula. Tachypnea and tachycardia persisted, which created issues with mobilization and nutrition. A thoracotomy was increasingly debated for likely lobar resection and debridement of devitalized tissue, but the patient began to gradually improve, leading to complete resolution of the air leak. No further surgical intervention was required, and the chest tube was removed without complication. She was discharged home on combination oral amoxicillin–clavulanate (30 mg/kg/d based on the amoxicillin component). At last follow-up months later, she continued to do well and had minimal respiratory symptoms (Figure 3). Hemagglutination inhibition and microneutralization assays comparing serum samples collected on admission and one month later showed a greater than four-fold rise in antibody titers against pandemic (H1N1) 2009 virus (hemagglutination inhibition geometric mean titer 50.4–1140.4, microneutralization titer 40–1280).
Follow-up anteroposterior chest radiograph two weeks after discharge showing significant improvements, with decreased size of the lucency in the middle lobe of the right lung, decreased consolidation in the right lower field and decreased atelectasis, but continued moderate pleural effusion (arrows).
Discussion
We present an instance of severe streptococcal pneumonia in a young girl with pandemic (H1N1) influenza that was complicated by necrosis, large pneumatoceles and development of a bronchopleural fistula. Although the incidence of streptococcal pneumonia has decreased because of vaccination, it remains a common cause of community-acquired bacterial pneumonia.2,3 In our patient, S. pneumoniae was identified on polymerase chain reaction analysis; conventional cultures may be negative following the initiation of antibiotics. Pneumococcal DNA has been identified in up to 87.5% of culture-negative pleural fluid in children with parapneumonic empyema.4 S. pneumoniae is a gram-positive, α-hemolytic coccus with 91 serotypes differentiated by their capsular polysaccharide.3 The ability of S. pneumoniae to cause disease in humans is based on its ability to adhere to human cells via surface adhesions, avoid phagocytosis by the host immune response and activate a significant inflammatory response.3 Concurrent respiratory viral infection, as well as other forms of immune incompetence and suppression, predispose the host to pneumococcal infection.3 Viral infection upregulates host cell surface receptors, which secondarily improves pneumococcal adherence; the host’s ability to clear the bacteria is further hindered by viral-induced epithelial damage.3 Clinically, children with pneumococcal pneumonia present with fever, nonproductive cough, tachypnea and fatigue.3 Radiographically, S. pneumoniae has a typical appearance of lobar consolidation and may also show complications such as pleural effusions and empyema.3 Necrotizing pneumococcal pneumonia is uncommon but has been described in previously healthy children.5 Gangrenous lung changes may result from vascular and parenchymal compromise.
Treatment of streptococcal pneumonia
In less severe cases, suspected bacterial pneumonia is often treated empirically because of the difficulty in obtaining sputum samples for confirming a causative agent.2 When available, gram-stain, culture and susceptibility tests performed on sputum samples or lung aspirates can direct antibiotic therapy.2 There is limited research regarding the most effective antibiotic treatment for community-acquired pneumonia. However, based on the research that has been done, if S. pneumoniae is suspected in uncomplicated cases, treatment should be started with amoxicillin.3,6,7 Amoxicillin–clavulanate, ampicillin, penicillin, or intravenous cephalosporins such as cefuroxime or cefotaxime are indicated for children admitted to hospital.7 Research is also limited regarding duration of antibiotic therapy; however, the British Thoracic Society suggests a course of 7–10 days of oral therapy based on “custom and practice.”6
Complications of streptococcal pneumonia
Most cases of pneumococcal pneumonia are uncomplicated; however, pleural effusion, empyema, lung abscess and necrotizing pneumonia are documented complications.8 Up to 50% of hospital-admitted children will develop a complication, but full resolution of symptoms is generally expected.9 Although the incidence of childhood bacterial pneumonias has dropped since the introduction of the 7-valent conjugate pneumococcal vaccine, complicated disease has actually increased.10 In the United States between 1997 and 2006, there was a 70% increase in the rate of hospital admission for empyema.10 The clinical course of our patient shows how, in complicated disease, large pneumatoceles may develop secondary to parenchymal necrosis. The size of these lesions may have increased in part because of positive pressure mechanical ventilation. Most pneumatoceles resolve without sequelae but occasionally require urgent surgical intervention.8
Surgical procedures for the management of pneumonia complications (pleural effusion, empyema and pneumothorax) include chest tube insertion, video-assisted thoracoscopy, and thoracotomy with possible lung resection. British Thoracic Society guidelines for the management of pleural infection in children indicate the need for adequate chest drainage (tube thoracostomy) for expanding fluid collections and compromised respiratory function.11 The timing and nature of more aggressive surgical intervention is controversial.11,12 Data to support surgical decision-making in necrotizing pneumonia are limited, but, given the clinical course of our patient, we believe that a conservative approach is prudent. A bronchopleural fistula is particularly concerning to the clinician, but patience in the management of a clinically stable patient may avoid lung surgery.
There is evidence for good long-term clinical outcomes in children with necrotizing pneumonia.13 Our patient continues to be monitored on an outpatient basis for potential complications such as bronchiectasis and recurrent respiratory infections.
Co-infection with pandemic (H1N1) 2009 virus and streptococcal pneumonia
Our patient presented in the midst of the 2009 influenza pandemic. In the course of her hospital stay, two other young patients who were otherwise healthy presented with similar signs and followed a similar clinical path. Although rapid respiratory viral testing of the nasopharyngeal swab was negative for pandemic (H1N1) influenza, we considered our patient to have a coinfection with pandemic influenza based on the following: the sensitivity of the rapid test is low; her clinical presentation was suspicious for influenza; empiric antiviral therapy had been initiated before the swab, which may have further decreased the likelihood of viral presence in her nasopharynx; and the high community prevalence of pandemic influenza. Our clinical suspicion was later validated by the demonstration of a greater than four-fold rise in antibody titers against pandemic (H1N1) 2009 virus by two different methods on specimens taken acutely and one month later.
Palacios and colleagues examined the severity of pandemic (H1N1) influenza in the presence of S. pneumoniae co-infection.14 Young (6–55 years of age) and otherwise healthy people tended to have more severe disease (defined by need for hospital admission) as a result of S. pneumoniae infection following an initial infection with pandemic (H1N1) influenza. A second study showed that pandemic (H1N1) influenza impairs both the innate and adaptive immune responses to S. pneumoniae15 through suppression of the Th1 immune cascade and through decreased production of CD4 and B lymphocytes and an upregulation of T regulator cells.
A review of 58 children admitted to a Canadian hospital with pandemic (H1N1) influenza did not find increased disease severity in comparison to seasonal influenza A.16 Nearly one-third of patients did have radiographic findings of pneumonia, but there was no report of necrotizing pneumonia within the study’s cohort. Consistent with the history of our patient, asthma seemed to increase the risk of severe disease.
Our experience with secondary pneumococcal infection suggests that conservative management consisting of antibiotics and adequate chest drainage is sufficient treatment, even if the clinical presentation and radiologic images are impressive.
-
Co-infection with the pandemic (H1N1) 2009 virus and Streptococcus pneumoniae is not uncommon and is potentially associated with more severe disease.
-
With the onset of routine pneumococcal vaccination, there has been a decrease in childhood bacterial pneumonia but, conversely, there has been an increase in severe diseases such as empyema and necrotizing infections.
-
Conservative management of complicated pneumonia consisting of antibiotics and adequate chest drainage may be sufficient treatment despite impressive radiologic images.
Key points
The section Cases presents brief case reports that convey clear, practical lessons. Preference is given to common presentations of important rare conditions, and important unusual presentations of common problems. Articles start with a case presentation (500 words maximum), and a discussion of the underlying condition follows (1000 words maximum). Generally, up to five references are permitted and visual elements (e.g., tables of the differential diagnosis, clinical features or diagnostic approach) are encouraged. Written consent from patients for publication of their story is a necessity and should accompany submissions. See information for authors at www.cmaj.ca.
Footnotes
-
See also practice article by Udupa and colleagues, page 220
-
Competing interests: Noni MacDonald is Section Editor, Population and Public Health for CMAJ and was not involved in the editorial decision-making process for this article. No competing interests declared by the other authors.
-
This article has been peer reviewed.
-
Contributors: Jo Yazer and Steven Lopushinsky were responsible for the article’s concept, drafting of the manuscript and critical revision of content. Noni MacDonald and Michael Giacomantonio provided expert opinion and critical review for intellectual content. All of the authors approved the final version submitted for publication.