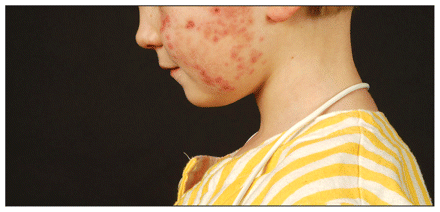
A previously healthy four-year-old girl presented with a rash of sudden onset consisting of erythematous macules on both cheeks (Figure 1) that were neither itchy nor tender and did not blanch. Over the next week, the rash spread from her cheeks to her arms, back, thighs and buttocks, and became papular and darker. The patient had no other symptoms. One week later, when the rash had not resolved, oral cephalexin was prescribed. The rash did not improve and the patient was referred to a pediatric dermatologist.
Figure 1: Erythematous macular-papular rash on the cheek of a four-year-old girl with leukemia cutis.
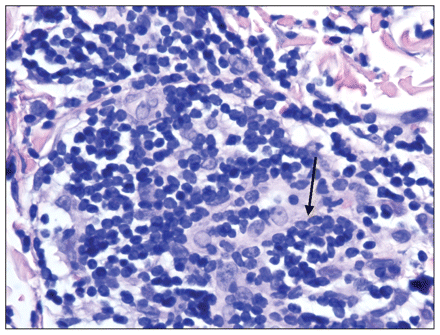
A skin biopsy performed two weeks after the onset of the rash showed leukemic blast cells (Figure 2). Bloodwork was then performed and a peripheral blood smear showed blast cells. Physical examination showed hepatosplenomegaly and mild cervical lymphadenopathy in addition to the rash.
Figure 2: Microscopic view of skin biopsy specimen (hematoxylin and eosin stain, original magnification ×600, ) showing leukemic blast cells (arrow).
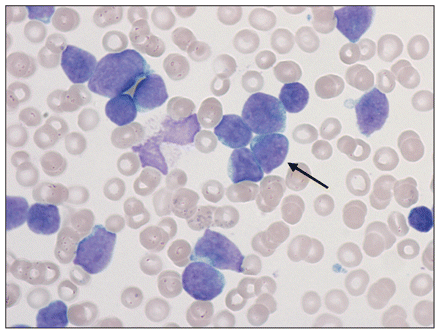
A complete blood count showed a leukocyte count of 17.7 (normal 5.0–14.5) × 109/L, a hemoglobin level of 115 (normal 115–135) g/L, a platelet count of 175 (normal 150–450) × 10 9/L, an absolute neutrophil count of 0.35 (normal 1.5–8.0) × 109/L and a blast cell count of 16.6 (normal < 0.01) × 109/L. No evidence was found of tumour lysis syndrome. A bone marrow aspirate confirmed T-cell acute lymphoblastic leukemia with 80%–85% blast cells (Figure 3). Immunophenotyping showed that the blast cells were positive for CD45, CD3, CD7, CD5 and DNA nucleotidylexotransferase. They were also weakly positive for CD79a and CD2. Cytogenetic testing showed a structural rearrangement between chromosomes 9 and 12 resulting in the deletion of their short arms (45,XX,-9,t(9;12)(q12;p1)).
Figure 3: Histologic image of bone marrow aspirate (Wright Giemsa stain, original magnification ×1000, ) showing multiple lymphoblasts (arrow).
The results of a lumbar puncture showed no evidence of leukemic cells. A radiograph of the chest was normal. Ultrasonography of the abdomen confirmed hepatosplenomegaly with extension of the edge of the liver to 3 cm below the costal margin and enlargement of the spleen to a diameter of 13.9 cm.
Induction therapy was started immediately, using protocol COG AALL 0434, which includes intrathecal methotrexate, intrathecal cytarabine, vincristine, prednisone, pegasparaginase and daunorubicin. At the end of induction, the patient’s bone marrow contained 26% blast cells. Remission was achieved during the consolidation phase using nelarabine, cyclophosphamide, cytarabine, mercaptopurine, vincristine, pegasparginase and intrathecal methotrexate. No complications occurred from the chemotherapy. The rash resolved midway through induction chemotherapy without scarring or residual pigmentation.
Discussion
Acute lymphoblastic leukemia is the most common leukemia of childhood. It commonly presents with nonspecific symptoms, such as fatigue and irritability, along with symptoms secondary to bone marrow failure. In pediatric acute lymphoblastic leukemia, 48% of patients will have petechiae or purpura on presentation. 1 Occasionally, leukemia can present directly in the skin and is referred to as leukemia cutis. Although leukemia cutis tends to present with other features of leukemia, it can occasionally precede the development of blast cells in the marrow and blood. 1 The condition is then known as aleukemic leukemia cutis. 1,2
Leukemia cutis is an infiltration of the skin by neoplastic leukocytes (myeloid or lymphoid), resulting in clinically identifiable cutaneous lesions. 3 It is seen most commonly in congenital leukemia and acute myelogenous leukemia. 3 In adults with acute myelogenous leukemia, leukemia cutis represents a higher tumour burden and tends to be predictive of a prolonged and less favourable course. 4 These tendencies do not seem to be the case in congenital leukemia, where the presence of this rash does not alter the expected course. 5 In the pediatric population, the frequency of leukemia cutis is higher in pediatric acute myelogenous leukemia (approximately 10%) than in pediatric acute lymphoblastic leukemia (1%). 1,2
The clinical appearance of leukemia cutis is variable. The most common manifestation is described as erythematous or violaceous plaques, papules or nodules involving the face, trunk and extremities. 4 Less common appearances include macules, maculopapules or plaques. 4 In the setting of acute myelogenous leukemia, leukemia cutis presents as a firm nodule with a greenish hue, known as a chloroma or granulocytic sarcoma. 1 In the neonate, leukemia cutis often presents as sites of extramedullary hematopoiesis in the skin, imparting a “blueberry muffin” appearance. 5 In our patient, the appearance of leukemia cutis was typical.
Differential diagnosis
Leukemia cutis needs to be differentiated from cutaneous manifestations of leukemia secondary to bone marrow dysfunction (such as petechiae), drug reactions and infections secondary to an immunocompromised state (Table 1).
Table 1: Differential diagnosis of leukemia cutis
Leukemia cutis must also be distinguished from the cutaneous lymphomas (i.e., mycosis fungoides and Sézary syndrome). 1,3 Children with mycosis fungoides tend to have hypopigmented lesions, especially on the buttocks. 1 Sézary syndrome is a cutaneous lymphoma that presents as diffuse erythroderma and lymphadenopathy; it involves the blood, skin and lymph nodes. 3 The syndrome is similar to mycosis fungoides but has a more aggressive course. 3 Cutaneous lymphomas are very uncommon in the pediatric population.
In our patient, leukemia cutis was the only presenting symptom of T-cell acute lymphoblastic leukemia. This instance of the disease shows that sudden rashes, if persistent and of unknown cause, warrant a full physical examination to look for other abnormalities. Complete evaluation of this type of rash should include bloodwork with a complete blood count, imaging as indicated and early involvement of a dermatologist.
-
A sudden, persistent rash of unknown cause warrants full physical examination, hematologic investigation including complete blood count, and dermatological evaluation.
-
Childhood leukemia may present directly on the skin, a condition known as leukemia cutis.
-
Leukemia cutis is seen more commonly in acute myelogenous leukemia than in acute lymphoblastic leukemia.
-
The rash generally resolves with treatment of the underlying leukemia.
Key points
Footnotes
-
This article has been peer reviewed.
Competing interests: None declared.
Contributors: All of the authors contributed to the development of the concepts in the manuscript and to the drafting and revision of the manuscript. All of them approved the final version submitted for publication.
Previously published at www.cmaj.ca
Acknowledgements: The authors would like to thank Dr. E. Leung and Dr. R. Prokopetz for providing microscopic photographs.
The section Cases presents brief case reports that convey clear, practical lessons. Preference is given to common presentations of important rare conditions, and important unusual presentations of common problems. Articles start with a case presentation (500 words maximum), and a discussion of the underlying condition follows (1000 words maximum). Generally, up to five references are permitted and visual elements (e.g., tables of the differential diagnosis, clinical features or diagnostic approach) are encouraged. Written consent from patients for publication of their story is a necessity and should accompany submissions. See information for authors at www.cmaj.ca.