- © 2007 Canadian Medical Association or its licensors
The Case: A previously healthy 27-year-old woman bearing twins during her first pregnancy arrived at our hospital's high-risk obstetrical unit when labour threatened at 33 weeks' gestation. Her antenatal care had been standard and her pregnancy, until then, unremarkable.
The results of her physical examination, including a chest exam, were normal. An initial oxygenation-by-saturation probe revealed no evidence of underlying lung disease. She was admitted to the obstetrical unit for observation and instructed to maintain strict bed rest. When routine urinalysis found leukocytes and nitrates, she was given nitrofurantoin (a 100-mg capsule, twice a day) for a suspected urinary-tract infection.
About 48 hours after admission, she developed acute dyspnea associated with cough, frothy hemoptysis and sharp retrosternal chest pain that was worse with deep inhalation. She had no purulent phlegm, fever, chills, sweats, arthralgia or rash. Her medical history was free of lung disease, deep-vein thrombosis (DVT) and pulmonary embolism, and no family members had a thromboembolic disease or thrombophilic state. She had not recently travelled nor been exposed to animals or to people with a respiratory illness. She denied smoking, drinking alcohol or using any drugs.
We consulted specialists in internal medicine and respirology. Her respiratory distress (oxygen saturation 89%, room air) improved to 95% when 50% O2 was administered via facemask. Her respiratory rate was 24 breaths/min; blood pressure, 164/100 mm Hg in both arms; heart rate, 96 beats/min and regular; and temperature, 36.4°C. Lung examination disclosed coarse expiratory breath sounds without obvious crackles. A cardiovascular exam revealed a hyperdynamic precordium with normal heart sounds and no murmurs. Her jugular venous pressure was not elevated. Results of an abdominal examination were unremarkable, whereas that of her extremities revealed mild pitting edema to both shins (which had not changed from admission). No evidence of rash was found.
Laboratory investigations revealed that breathing room air resulted in hypoxia (PO2 36 [normal 60–80] mm Hg) with mild hypercapnia (PCO2 35 [normal 28–32] mm Hg); blood gases were otherwise normal. A complete blood count revealed leukocytosis with neutrophilia, microcytic anemia and a normal platelet count. Electrolyte levels were within normal limits, unchanged from baseline. Measurements of liver enzymes were all within their normal ranges, but lactate dehyrogenase (normally 91–180 U/L) was high (239 U/L). Her serum concentrations of cardiac enzymes, creatinine kinase and troponin Ι were not elevated. Urinalysis revealed protein (1+) and microscopic blood. Sputum and blood cultures produced no infective agents.
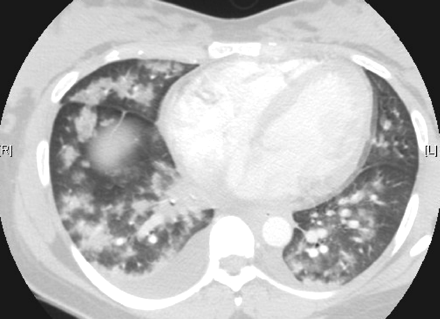
Chest radiography revealed diffuse parenchymal disease and an enlarged cardiac silhouette. Although an acute pulmonary embolism could not be excluded, a ventilation–perfusion scan showed its probability rating to be low. Doppler ultrasound studies revealed no DVT in either leg. A subsequent high-resolution CT scan of her chest and helical angiography (Fig. 1) showed no evidence of pulmonary thromboembolic disease, but did reveal bilateral, multifocal air-space disease and pleural effusions, larger on the right side.
Fig. 1: This helical CT scan of the patient's thorax reveals bilateral, multifocal air-space disease and pleural effusions that are larger on the right side.
Her left-ventricular function was normal (estimated at 55% via transthoracic echocardiography), with no structural abnormalities. The results of a vasculitis screen were negative. Her erythrocyte sediment rate (ESR) was found to be greatly elevated (73 mm; normal 0–20 mm).
The presumptive diagnosis was acute pneumonitis caused by nitrofurantoin. Nitrofurantoin was discontinued, and she began taking prednisone (50 mg/d). Within 12 hours, her breathing was noticeably easier and her oxygenation, dramatically improved (O2 saturation 96% in room air). By 24 hours, her symptoms had completely resolved. At 48 hours after her acute episode, she looked well, and noninvasive fetal monitoring showed that the twins were in no distress. Oral prednisone was discontinued and she was discharged home.
Three weeks later, she gave birth by spontaneous vaginal delivery to 2 healthy girls.
This case represents an unusual example of dyspnea and respiratory failure in a pregnant patient. Some 60%–70% of women experience dyspnea or “air hunger” during the course of a normal pregnancy, most commonly in the first or second trimesters; it is thought likely to be related to progesterone-induced hyperventilation. Cough, hemoptysis and clinically significant hypoxemia are unusual during pregnancy; any of these should prompt the physician to search for underlying lung disease (Box 1).
The antibiotic nitrofurantoin is used primarily to treat acute urinary-tract infections and to prevent chronic ones in patients with predisposing factors such as an indwelling catheter. Its side effects are usually minor, ranging from nausea and gastrointestinal discomfort to severe hypersensitivity reactions that can affect multiple organ systems.1
Nitrofurantoin-induced pneumonitis was first described in 1965 in a patient with nitrofurantoin-induced pulmonary edema.2 In 1980, 921 cases of adverse reactions to nitrofurantoin reported to the Swedish Adverse Drug Reaction Committee from 1966 through 1976 were described;3 the 2 reported most frequently (acute pulmonary reactions 43%, allergic reactions 42%) are both characteristic of an acute hypersensitivity reaction. Nitrofurantoin-induced lung toxicity appears to occur in fewer than 1 in 1000 exposures,1 with acute toxicity nearly 10 times as prevalent as chronic.1 Toxicity can occur at relatively small doses,1 and does not appear to be dose-related.
Acute changes can be imaged in cases of nitrofurantoin pneumonitis, although few such images have been published. Chronic exposure to nitrofurantoin can cause a widespread reticular pattern in lung tissue, with an appearance of irreversible fibrosis — which in some patients can nevertheless spontaneously resolve up to a year after drug discontinuation.4
In 1981, Holmberg and Boman5 first described the clinical features of acute nitrofurantoin-induced lung injury: fever, dypnea, dry cough, an elevated ESR and, less commonly, chest pain, arthralgias and diffuse rash. Eosinophils have also been found in bronchioalveolar lavage from patients with nitrofurantoin-induced lung injury. Symptoms occur 2–3 weeks after the first doses of nitrofurantoin.5 An estimated 10%–15% of patients with this condition die of it.5 The case we describe was unusual in the acuity of the patient's respiratory deterioration.
Case reports suggest that acute nitrofurantoin lung toxicity can reverse completely upon drug discontinuation.5 In acute cases, it is unclear whether steroids are of any benefit independent of discontinuation of the antibiotic.
Initial ventilation–perfusion scans, which feature a lower radiation dose than conventional CT used to diagnose pulmonary embolism (50 v. 60 mrad),6 are currently recommended for patients whose risk of pulmonary embolism or venous thromboembolism is moderate to high.7 New evidence8 suggests that CT scanning may provide even less exposure risk for the fetus and may become the preferred modality, especially when chest radiography has revealed abnormalities.
Our patient's response to nitrofurantoin discontinuation and the institution of high-dose oral corticosteroids was prompt. Although her case was relatively unusual, it highlighted steps often necessary to investigate dyspnea during pregnancy. It also illustrates well the need to investigate all pregnant women who develop notable dyspnea in association with other features such as hypoxia, cough and hemoptysis.
Footnotes
-
This article has been peer reviewed.
Competing interests: None declared.