Although Canadian guidelines for the diagnosis and management of asthma have been published over the last 15 years,1,2,3,4 there has been little focus on issues relevant to asthma in the young child or on prevention strategies for asthma. Since the last update in 2001,4 important issues and new studies focusing on asthma in early life have highlighted the need to incorporate the new information into the asthma guidelines. Reports pertaining to a number of issues published between 2000 and June 2003 were reviewed initially by small expert resource groups. The results of these reviews were discussed by stakeholders during a 2-day consensus meeting, 27–28 June 2003. A working group with a pediatric focus met under the auspices of the Canadian Network For Asthma Care and an adult asthma group met under the auspices of the Canadian Thoracic Society. On the first day, these groups met separately to discuss specific issues related to pediatric and adult asthma and, on the second day, met jointly to discuss dissemination and implementation of the asthma guidelines. Data published up to December 2004 pertaining to each of the issues considered by the consensus working group were reviewed by the individual expert resource groups, who concurred that these were insufficient to modify any of the recommendations that follow.
This summary reports the recommendations for prevention, assessment and management of asthma in children and adults. A level of evidence is assigned to each recommendation based on the strength of the supporting data5 (Table 1). Background documents supporting recommendations for children follow in a separate supplement. Background documents for adults are published in the Canadian Respiratory Journal.6
Table 1.
Definition of asthma
The definition of asthma remains descriptive and has not changed since the 1999 Canadian asthma consensus guidelines.3 Asthma is characterized by paroxysmal or persistent symptoms, such as dyspnea, chest tightness, wheezing, sputum production and cough associated with variable airflow limitation and airway hyperresponsiveness to endogenous or exogenous stimuli. Inflammation and its resultant effects on airway structure are considered the main mechanisms leading to the development and persistence of asthma.
General management of asthma
Optimal management of asthma requires adequate evaluation of the patient and his or her environment. Asthma control should be assessed using specific criteria (Table 2). Asthma severity is more difficult to assess and may only be determined after asthma control is achieved. Asthma control should be assessed at each visit.
Table 2.
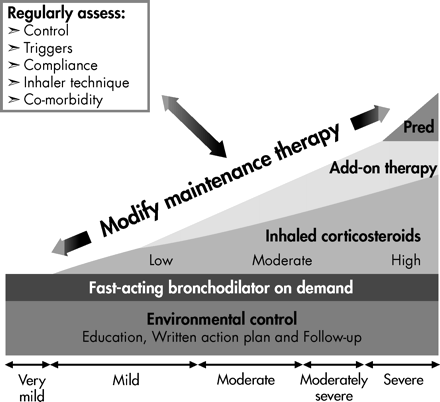
If control is inadequate, the reason or reasons should be identified and maintenance therapy should be modified (Fig. 1). Any new treatment should be considered a therapeutic trial and its effectiveness should be re-evaluated after 4–6 weeks.
Fig. 1: Continuum of treatments for asthma management. Reprinted with permission from Elsevier (J Allergy Clin Immunol 2004;113:650-6).
Inhaled corticosteroids (ICSs) should be introduced as initial maintenance treatment even when the patient reports symptoms fewer than 3 times a week. Although less effective than low-dose ICSs, leukotriene receptor antagonists (LTRAs) are an alternative for patients who cannot or will not use ICSs. If control is inadequate on low-dose ICSs, identify the reasons for poor control and, if indicated, consider additional therapy with long-acting β2-agonists or LTRAs. Severe asthma may require additional treatment with systemic steroids. Asthma control and maintenance therapy must be assessed regularly.
If good control has been sustained, consideration should be given to gradually reducing maintenance therapy, with regular reassessments to ensure that control remains adequate. This will allow determination of the minimum therapy needed to maintain acceptable asthma control.
Asthma education is an essential component of asthma care. Poor asthma control is not usually due to a lack of efficacy of the medication, but is more often related to suboptimal use of medication or aggravating factors, comorbidities, poor inhaler technique, poor environmental control or a lack of continuity of care. Suboptimal use of asthma medication may be the result of inappropriate physician recommendation, poor adherence or both, perhaps as a result of undue fear of adverse effects of therapy. In the face of poor asthma control, it is crucial to identify and address the cause (Table 3).
Table 3.
Diagnosis of asthma
Recommendations regarding the diagnosis or assessment of asthma severity in adults and older children have not changed from previous publications.3 However, the diagnosis of asthma in the preschool child was a major focus of the current discussions.
Recommendations
-
1. Physicians must obtain an appropriate patient and family history to assist them in recognizing the heterogeneity of wheezing phenotypes in preschool-aged children (level III).
-
2. In children who are unresponsive to asthma therapy, physicians must exclude other pathology that might suggest an alternative diagnosis (level IV).
-
3. The presence of atopy should be determined because it is a predictor of persistent asthma (level III).
Diagnostic tools
In children <3 years of age, neither lung function testing nor assessment of airway inflammation is clinically helpful nor commonly available for the diagnosis of asthma.4,7,8,9 Asthma diagnosis in children <6 years of age is dependent on history and physical examination. Table 4 provides some criteria to help identify a child suffering from asthma; the greater the number of criteria met, the greater the likelihood of asthma.
Table 4.
Evolution of preschool wheezing
Preschool wheezing can be classified as transient early onset wheezing (before age 3 years), which is often outgrown; persistent early onset wheezing (before age 3 years), which persists in school age; and late-onset wheezing (after age 3 years), which is less likely to resolve. Among preschool children with wheezing, 50%–60% outgrow the problem.10,11
Role of atopy
Recurrent wheezing in non-atopic preschool children is likely to resolve in childhood, but atopy is a predictor of persistent asthma.12,13,14 A clinical index may help predict which wheezing children are likely to have persistent asthma15 (Table 5). Physicians must obtain a personal and family history of atopy and look specifically for the presence of atopic dermatitis during physical examination. The presence of atopy can be established by skin-prick testing16 or measurement of specific IgE antibodies17,18 and is suggested by elevated peripheral total IgE and blood eosinophils.18,19,20
Table 5.
Prevention strategies
Recommendations
Primary prevention
-
1. With conflicting data on early life exposure to pets, no general recommendation can be made with regard to avoiding pets for primary prevention of allergy and asthma (level III). However, families with biparental atopy should avoid having cats or dogs in the home (level II).
-
2. There are conflicting and insufficient data for physicians to recommend for or against breastfeeding specifically for the prevention of asthma (level III). Due to its numerous other benefits, breastfeeding should be recommended.
Secondary prevention
-
3. Health care professionals should continue to recommend the avoidance of tobacco smoke in the environment (level IV).
-
4. For patients sensitized to house dust mites, physicians should encourage appropriate environmental control (level V).
-
5. In infants and children who are atopic, but do not have asthma, data are insufficient for physicians to recommend other specific preventive strategies (level II).
Tertiary prevention
-
6. Allergens to which a person is sensitized should be identified (level I), and a systematic program to eliminate, or at least to substantially reduce, allergen exposure in sensitized people should be undertaken (level II).
Primary prevention
Primary prevention of asthma is defined as intervention before the development of asthma or any predisposing disease such as atopic dermatitis, food allergy or allergic rhinitis. We focused on 2 specific areas of primary prevention: exposure to pets in early life and breastfeeding. Recommendations related to avoiding exposure to environmental tobacco smoke remain unchanged.
Sensitization to allergens is one of the strongest determinants of subsequent development of asthma. Several recent studies have suggested the possibility that a cat or dog in the home early in life might decrease the risk of developing allergy or asthma.21,22,23,24,25,26,27 Currently available data do not provide conclusive guidance relating to exposure to pets in early life. There is evidence that children with biparental atopy and those whose mothers have asthma should avoid exposure to pets in early life.26
There is clear evidence that breastfeeding protects against early life wheezing syndromes.28,29 However, recent studies suggest that breastfeeding may increase the risk of persistent asthma.30,31 Reasons for this are speculative, but may relate to a lower incidence of infectious diseases among breastfed children (the hygiene hypothesis) or to a higher rate of breastfeeding in atopic families (confounding by indication). However, other benefits of breastfeeding are sufficiently clear to recommend exclusive breastfeeding of infants for the first 4 months of life or more.
Secondary prevention
Secondary prevention is defined as intervention(s) for infants and children who are at high risk for the development of asthma but who have not yet developed asthma symptoms or signs.32 These patients usually have allergic conditions and a family history of allergic disease.32 There is currently insufficient evidence regarding pharmacologic treatment, control of environmental factors or allergen-specific immunotherapy to allow firm recommendations to be made. Health care personnel should continue to recommend smoke avoidance measures in all children and reduction of dust mites in the environment of sensitized people.
Tertiary prevention
Tertiary prevention implies identifying allergens to which a person is sensitized and undertaking a systematic program to eliminate, or at least substantially reduce, allergen exposure in sensitized people. This strategy is still endorsed.
Pharmacotherapy
Recommendations
First-line maintenance therapy
-
1. Physicians should recommend inhaled corticosteroids (ICSs) as the best option for anti-inflammatory monotherapy for childhood asthma (level I).
-
2. There is insufficient evidence to recommend leukotriene receptor antagonists (LTRAs) as first-line monotherapy for childhood asthma (level I). For children who cannot or will not use ICSs, LTRAs represent an alternative (level II).
Treatment of intermittent asthma with ICSs
-
3. There are insufficient data for physicians to recommend short courses of high-dose ICSs in children with mild, intermittent asthma symptoms, and the safety of this approach has not been established (level II).
-
4. Physicians must carefully monitor children with intermittent symptoms to ensure that they do not develop chronic symptoms requiring maintenance therapy (level IV).
-
5. Physicians should recommend that children with frequent symptoms, severe asthma exacerbations or both receive regular, not intermittent, treatment with ICSs (level IV).
Add-on therapies
-
6. Long-acting β2-agonists are not recommended as maintenance monotherapy in asthma (level I).
-
7. After reassessment of compliance, control of environment and diagnosis, if asthma is not optimally controlled with moderate doses of ICSs, therapy should be modified by the addition of a long-acting β2-agonist (level I). Alternatively, addition of an LTRA or increasing to a moderate dose of ICS may be considered (level I).
Relievers
Short-acting β2-agonists have been used for symptom relief for many years.33 Recently, a long- but also fast-acting agent, formoterol, has been approved for symptom relief.34 Fast-acting bronchodilators may be used to relieve acute intermittent asthma symptoms. They should be used only on demand at the minimum dose and frequency required. Need for a reliever more than 3 times a week (aside from a pre-exercise dose) suggests suboptimal asthma control and indicates the need to re-assess treatment. Inhaled ipratropium bromide is less effective, but in the emergency department, ipratropium bromide combined with fast-acting β2-agonists is effective for treatment of severe acute asthma in children and adults.35,36
First-line maintenance therapy
Early ICS treatment
The role of ICSs in the early treatment of mild-to-moderate asthma has been extensively evaluated. In a systematic review,37 treatment with beclomethasone significantly improved forced expiratory volume in 1 s (FEV1) and morning peak expiratory flow (PEF) and reduced the use of β-agonists and exacerbations compared with placebo. In a recent large prospective study of pediatric and adult patients with mild asthma, early use of moderate-dose inhaled budesonide was associated with better control of symptoms, improved FEV1 and, importantly, a marked reduction in asthma exacerbations compared with placebo.38
ICS therapy may be associated with mild reductions in linear growth in children,39 which appears to occur primarily during the first year of therapy. Prospective studies show that children treated with moderate doses of ICS for long periods of time attain their predicted adult height.40 There is no evidence to support initial treatment using combination therapy (ICS and long-acting β-agonist) in patients not previously given a trial of an ICS alone.41
An alternative to ICS is use of a leukotriene receptor antagonist (LTRA). Three well-designed trials, 1 in preschool-aged and 2 in school-aged children demonstrated the superiority of LTRA over placebo in persistent asthma.42,43,44 Montelukast was associated with fewer days of asthma symptoms and β2-agonist use, less use of rescue oral steroids and, in older children, greater improvement in lung function.
A Cochrane review of moderate-dose ICS compared with LTRA monotherapy in school-aged children with mild-to-moderate airway obstruction reported less use of β2-agonist in the ICS group but no significant difference in symptoms, spirometry or the risk of an asthma exacerbation requiring systemic steroids.45 There are currently too few trials to draw any firm conclusions. A recent systematic review comparing ICS (400 mg of beclomethasone or equivalent) to LTRAs in mild-to-moderate asthma identified 13 trials (all adult trials with 1 exception). This review found that adults treated with LTRAs were more likely to suffer an asthma exacerbation requiring a course of oral prednisone.46 Thus, ICSs remain the preferred initial treatment for asthma in children and adults.
The effectiveness of intermittent treatment
Intermittent asthma symptoms are a common pattern of asthma in infants and children, with exacerbations usually triggered by viral, upper respiratory tract infections. This form of asthma is less likely associated with atopy and may have a different natural history. Treatment is problematic, as optimal therapy has not been clearly determined. Because such children are asymptomatic between exacerbations, intermittent treatment with ICS is attractive to both physicians and families, and this management strategy is prevalent in Canada even though evidence to support the practice is scant.
Studies performed in preschool-aged children using high-dose intermittent therapy (beclomethasone 2250 μg/day, or budesonide 1600–3200 μg/day), for 5–10 days showed small reductions in asthma symptom scores and a trend toward less use of oral steroids. However, duration of symptoms, emergency visits and admissions to hospital did not appear to be affected by intermittent high-dose ICSs.47,48,49,50 Few studies evaluated the safety of intermittent high-dose ICS treatment.51
Therapy in addition to ICSs
In patients whose asthma is not adequately controlled on ICSs, available therapeutic options include add-on therapy with a long-acting β2-agonist, LTRA or theophylline or increasing the dose of ICS.
Long-acting β2-agonists
Long-acting β2-agonists are safe and effective medications for improving asthma control in older children and adults with asthma not optimally controlled despite regular maintenance therapy with ICSs,41,52,53 but they should not be used as monotherapy.54
Leukotriene receptor antagonists
In children and adults treated with moderate dose ICSs, there is some evidence suggesting that adding LTRAs is associated with improvements similar to doubling the ICS dose, but there is not yet sufficient evidence of equivalence between the 2 therapeutic strategies.55
Theophylline
In the few studies available evaluating add-on therapy in patients on ICS, theophylline was less effective than long-acting β2-agonists or LTRAs for improving asthma control.56
Comparison of long-acting β2-agonists and LTRAs as add-on therapy to ICSs
In adults, the addition of long-acting β2-agonists to 400 μg of chlorofluorocarbon-propelled beclomethasone or equivalent is more effective than LTRAs for improving lung function, reducing symptoms and use of rescue β2-agonists. However, both treatments had similar rates of asthma exacerbations and adverse events were similar in both groups.57,58,59 No similar data are yet available for children.
Inhalation devices
Only inhalation devices for childhood asthma were reviewed.
Recommendations
-
1. At each contact, health care professionals should work with patients and their families on inhaler technique (level I).
-
2. When prescribing a pressurized metered-dose inhaler (pMDI) for maintenance or acute asthma, physicians should recommend use of a valved spacer, with mouthpiece when possible, for all children (level II).
-
3. Although physicians should allow children choice of inhaler device, breath-actuated devices such as dry- powder inhalers offer a simpler option for maintenance treatment in children over 5 years of age (level IV).
-
4. Children tend to “auto-scale” their inhaled medication dose and the same dose of maintenance medication can be used at all ages for all medications (level IV).
-
5. Physicians, educators and families should be aware that jet nebulizers are rarely indicated for the treatment of chronic or acute asthma (level I). Delivery of medicinal aerosols depends on adequate inhalation technique. After repeated instruction and demonstration, more than 90% of children are able to achieve correct inhalation technique.60,61 Better knowledge of asthma, increased satisfaction with education and diminished asthma instability and attacks are associated with improved inhalation technique.62,63
One of the most difficult inhalation techniques to master is the use of a pressurized metered-dose inhaler (pMDI).64,65 Use of a spacer with pMDIs is strongly recommended for children. The pMDI with spacer can be used in place of the wet nebulizer in children of all ages in both acute and chronic care settings.66,67,68,69,70,71 Use of a mouthpiece, rather than a mask, (generally at 4 or 5 years old) maximizes lung deposition.72
Children can generally use dry-powder inhalers (DPIs), such as Turbuhaler and Diskus by the age of 5–6 years.73,74,75 Adults prefer breath-actuated DPIs over pMDIs and perform better with them.64 Using more than one inhalation device, may worsen technique with each device.76
In young children, deposition of medication in the lungs is about a tenth of the dose that would be delivered in adults. Thus, the same dose of maintenance medication can be used at all ages, because it will be “auto-scaled” down in children.77,78
Immunotherapy
The literature on immunotherapy was reviewed only for childhood asthma and the current recommendations are directed toward children.
Recommendations
-
1. Physicians should consider injection immunotherapy using appropriate allergens for the treatment of allergic asthma only when the allergic component is well documented (level I).
-
2. Physicians should not recommend the use of injection immunotherapy in place of avoidance of environmental allergens (level III).
-
3. Physicians may consider injection immunotherapy in addition to appropriate environmental control and pharmacotherapy when asthma control remains inadequate (level IV).
-
4. Immunotherapy is not recommended when asthma is unstable (level III).
Immune modulation is the only currently available therapy aimed at modifying the underlying disease process in asthma. Allergen immunotherapy is defined by the World Health Organization as therapeutic vaccine(s) for allergic diseases.79 Although debate about the value of immunotherapy continues, meta-analysis and review of immunotherapy support the potential value in childhood.80 Early immunotherapy may prevent development of asthma in children sensitized to house dust mite allergen.81 Allergen immunotherapy should be combined with allergen avoidance, pharmacotherapy and patient education. Furthermore, appropriate immunotherapy requires the use of single, well-defined allergens reaching a sufficient final dose to ensure effectiveness. As commonly undertaken, the value of immunotherapy using multiple allergens remains suspect.
Education and follow-up
Recommendations
-
1. Education is an essential component of asthma therapy and should be offered to all patients; educational interventions may be of particular benefit in patients with high asthma-related morbidity or severe asthma and at the time of emergency department visits and admissions to hospital (level I). Education programs should be evaluated (level III).
-
2. All patients should monitor their asthma using symptoms or peak expiratory flow (PEF) measurement (level I) and have written action plans for self-management that include medication adjustment in response to severity or frequency of symptoms, the need for symptom relief medication or a change in PEF (level I).
-
3. Asthma control criteria should be assessed at each visit (level IV). Measurement of pulmonary function, preferably by spirometry, should be done regularly (level III) in adults and children 6 years of age and older.
-
4. Socioeconomic and cultural factors should be taken into account in designing asthma education programs (level II).
Asthma education is an important part of asthma management and should aim primarily at changing patient behaviour, rather than simply improving knowledge.82 Patients with marked asthma-related morbidity and frequent acute care use should be targeted for asthma education. In this population, structured education with a written self-management plan, regular medical reassessment and review of key concepts reduces the number of emergency department visits.82,83,84
Recent studies, including a meta-analyses in children and adults, have confirmed that various methods of asthma education can improve symptoms, emotional state, communication with family members, school and physicians, school absenteeism, activity restriction, self-management skills, morbidity, lung function, quality of life, exacerbation rates and need for oral corticosteroids.85,86,87,88,89,90,91,92,93,94,95,96,97 Beneficial effects have been observed in a study involving adolescents, using education provided by peers.93 Long-term outcome may be improved further by reinforcement visits.91 Internet-based education may also improve adherence to the treatment plan in children. Education improves adherence to some environmental control measures, such as dust mite reduction measures, but is less helpful for animal avoidance in sensitized subjects.90
Conclusion
In Canadian children and adults with asthma, poor control remains prevalent, resulting in preventable morbidity, acute care visits, admission to hospital and even, fortunately rarely, mortality.98 In many cases, poor asthma outcomes can be avoided by ensuring that ICSs are started early and used as regular, long-term maintenance therapy, with special care taken to ensure patient compliance. Other crucial elements to achieving and maintaining good asthma control (Table 2) are environmental control measures, asthma education, treatment of comorbidity and appropriate use of add-on therapies.
It is hoped that these guidelines will improve asthma control in the many Canadians coping with this far too common disease.
Footnotes
-
*Members of the Pediatric Asthma Working Group of the Canadian Network For Asthma Care:
Mary L. Allen, MA, Allergy/Asthma Information Association, Montréal, Que.
Pierre Beaudry, MD, Canada Pediatric Society, Ottawa, Ont.
Allan Becker, MD, University of Manitoba, Winnipeg, Man.
Melva Bellafontaine, The Asthma Society of Canada, Toronto, Ont.
Denis Bérubé, MD, University of Montréal, Montréal, Que.
Andrew Cave, MD, University of Alberta, Edmonton, Alta.
Zave Chad, MD, University of Ottawa, Ottawa, Ont.
Myrna Dolovich, PEng, McMaster University, Hamilton, Ont.
Francine M. Ducharme, MD, McGill University, Montréal, Que.
Tony D'Urzo, MD, University of Toronto, Toronto, Ont.
Pierre Ernst, MD, MSc, McGill University, Montréal, Que.
Alexander Ferguson, MB, ChB, University of British Columbia, Vancouver, B.C.
Cathy Gillespie, RN, MN, CAE, Health Sciences Centre, Winnipeg, Man.
Mark Greenwald, MD, Asthma Society of Canada, Toronto, Ont.
Donna Hogg, RN, CAE, Canadian Network For Asthma Care, Edmonton, Alta.
Andrea Hudson, PhD, Canadian Pharmacists Association, Toronto, Ont.
Alan Kaplan, MD, Canadian Family Physician's Asthma Group, Richmond Hill, Ont.
Sandeep Kapur, MD, Dalhousie University, Halifax, N.S.
Cheryle Kelm, BPT, MSc, University of Calgary, Calgary, Alta.
Thomas Kovesi, MD, University of Ottawa, Ottawa, Ont.
Brian Lyttle, MD, University of Western Ontario, London, Ont.
Bruce Mazer, MD, McGill University, Montréal, Que.
Les Mery, MSc, Health Canada, Ottawa, Ont.
Mark D. Montgomery, MD, University of Calgary, Calgary, Alta.
Paul Pianosi, MD, Dalhousie University, Halifax, N.S.
Michelle Piwniuk, RRT, CAE, Canadian Society of Respiratory Therapists, Winnipeg, Man.
Amy Plint, MD, Canadian Association of Emergency Physicians, Ottawa, Ont.
John Joseph Reisman, MD, University of Ottawa, Ottawa, Ont.
Georges Rivard, MD, University of Laval, Quebec City, Que.
Malcolm Sears, MB, ChB, McMaster University, Hamilton, Ont.
F. Estelle R. Simons, MD, University of Manitoba, Winnipeg, Man.
Sheldon Spier, MD, University of Calgary, Calgary, Alta.
Robert L. Thivierge, MD, Universite de Montréal, Montréal, Que.
Wade Watson, MD, University of Manitoba, Winnipeg, Man.
Barry Zimmerman, MD, St. Michael's Hospital, Toronto, Ont.
†Members of the Adult Asthma Working Group of the Canadian Thoracic Society:
Tony Bai, MD, University of British Columbia, Vancouver, B.C.
Meyer Balter, MD, University of Toronto, Toronto, Ont.
Charles Bayliff, PharmD, London Health Sciences Centre, London, Ont.
Allan Becker, MD, University of Manitoba, Winnipeg, Man.
Louis-Philippe Boulet, MD, Université Laval, Sainte-Foy, Que.
Dennis Bowie, MD, Dalhousie University, Halifax, N.S.
André Cartier, MD, Université de Montréal, Montréal, Que.
Andrew Cave, MD, University of Alberta, Edmonton, Alta.
Kenneth Chapman, MD, University of Toronto, London, Ont.
Donald Cockcroft, MD, University of Saskatchewan, Saskatoon, Sask.
Robert Cowie, MD, University of Calgary, Calgary, Alta.
Stephen Coyle, MD, University of Manitoba, Winnipeg, Man.
Francine M. Ducharme, MD, McGill University, Montréal, Que.
Pierre Ernst, MD, McGill University, Montréal, Que.
Shelagh Finlayson, CAE, Ontario Lung Association, Toronto, Ont.
J. Mark FitzGerald, MD, University of British Columbia, Vancouver, B.C.
Frederick E Hargreave, MD, McMasterUniversity, Hamilton, Ont.
Donna Hogg, MS, RN, CAE, Dalhousie University, Halifax, N.S.
Alan Kaplan, MD, Richmond Hill, Ont.
Harold Kim, MD, Kitchener-Waterloo, Ont.
Cheryle Kelm, BPT, MSc, University of Calgary, Calgary, Alta.
Catherine Lemière, MD, Université de Montréal, Montréal, Que.
Paul O'Byrne, MD, McMaster University, Hamilton, Ont.
Malcolm Sears, MD, ChB, McMaster University, Hamilton, Ont.
Andrea White Markham, RRT, CAE, William Osler Health Centre, Brampton, Ont.
This article has been peer reviewed.
Competing interests: Allan Becker has received consultancy fees, speaker fees and/or grant support from all companies involved in asthma therapy in Canada. Catherine Lemière has received consultancy fees from ALTANA Pharma, AstraZeneca, GlaxoSmithKline and Merck Frosst; she has also received speaker fees from GlaxoSmithKline. Denis Bérubé is on the advisory boards of ALTANA Pharma and GlaxoSmithKline; he has received speaker fees from ALTANA Pharma, AstaZeneca, GlaxoSmithKline, Merck Frosst and 3M Pharmaceutical and travel assistance from GlaxoSmithKline. Louis-Philippe Boulet has been on the advisory boards of ALTANA Pharma, AstraZeneca, GlaxoSmithKline, Merck Frosst and Novartis; he has received speaker fees from AstraZeneca, GlaxoSmithKline, Merck Frosst and 3M Pharmaceutical, sponsorship for basic research from ALTANA Pharma, AstraZeneca, Merck Frosst and 3M Pharmaceutical and additional funding for participation in multicentre studies of the pharmacotherapy of asthma from AstraZeneca, ALTANA Pharma, Dynavax, Genentech, GlaxoSmithKline, Merck Frosst, Novartis, Roche, Schering and 3M Pharmaceutical. Francine Ducharme has received speaker fees from Merck. Mark FitzGerald has received honoraria, research funding and/or speaker fees from ALTANA Pharma, AstraZeneca, GlaxoSmithKline, Merck Frosst, Novartis and Roche. Thomas Kovesi has received consultancy fees and travel assistance to attend meetings from Merck Sharp and Dohme and from ALTANA Canada, and he has received speaker fees from Merck Sharp and Dohme.
References
- 1.↵
- 2.↵
- 3.↵
- 4.↵
- 5.↵
- 6.↵
- 7.↵
- 8.↵
- 9.↵
- 10.↵
- 11.↵
- 12.↵
- 13.↵
- 14.↵
- 15.↵
- 16.↵
- 17.↵
- 18.↵
- 19.↵
- 20.↵
- 21.↵
- 22.↵
- 23.↵
- 24.↵
- 25.↵
- 26.↵
- 27.↵
- 28.↵
- 29.↵
- 30.↵
- 31.↵
- 32.↵
- 33.↵
- 34.↵
- 35.↵
- 36.↵
- 37.↵
- 38.↵
- 39.↵
- 40.↵
- 41.↵
- 42.↵
- 43.↵
- 44.↵
- 45.↵
- 46.↵
- 47.↵
- 48.↵
- 49.↵
- 50.↵
- 51.↵
- 52.↵
- 53.↵
- 54.↵
- 55.↵
- 56.↵
- 57.↵
- 58.↵
- 59.↵
- 60.↵
- 61.↵
- 62.↵
- 63.↵
- 64.↵
- 65.↵
- 66.↵
- 67.↵
- 68.↵
- 69.↵
- 70.↵
- 71.↵
- 72.↵
- 73.↵
- 74.↵
- 75.↵
- 76.↵
- 77.↵
- 78.↵
- 79.↵
- 80.↵
- 81.↵
- 82.↵
- 83.↵
- 84.↵
- 85.↵
- 86.↵
- 87.↵
- 88.↵
- 89.↵
- 90.↵
- 91.↵
- 92.↵
- 93.↵
- 94.↵
- 95.↵
- 96.↵
- 97.↵
- 98.↵