Many factors influence prescribing behaviours, including scientific evidence, commercial information, physician education and patient preferences. We present an instance where prescribing was likely affected by a media report on Diane-35, a combination drug with both androgen-blocking and contraceptive properties, containing cyproterone acetate (2 mg) and ethinylestradiol (35 μg).
Diane-35 was approved for use in Canada in 1998 as a second-line treatment for severe acne with accompanying signs of androgenization in women who have not responded to oral antibiotics or other acne treatments. When Health Canada approved the drug, the restriction in use was applied because of safety concerns about genotoxic effects on liver cells (see www.cbc.ca/disclosure/archives/030114_diane/resources.html [accessed 2004 Nov 24]). From the mid-1980s to 1990s, Diane-35 had been marketed for both birth control and acne treatment in Europe, Latin America and Australasia.
In Canada, advertisements for Diane-35 portrayed young women with acne-free skin and suggested that viewers ask their doctors about the product. Although direct-to-consumer advertising of prescription drugs is illegal in Canada, Health Canada permits “reminder advertisements” stating a drug's name but not its indications. Diane-35's packaging is similar in size and shape to oral contraceptives.
Several case–control studies published in 1995–1996 on the effect of oral contraceptive drugs on the risk of venous thromboembolism (VTE) found higher risks to be associated with cyproterone. These reports were, however, based on small numbers of users. In 2001, investigators carrying out a nested case–control study reported an odds ratio of 3.9 (95% confidence interval [CI] 1.1–13.4) for VTE among women taking Diane-35 compared with those taking pills containing levonorgestrel.1 The United Kingdom, Australia and New Zealand reacted by issuing safety advisories in 2002. After Health Canada posted the UK advisory in Dec. 2002, it required Berlex to send a “Dear Health Professional” letter to Canadian physicians in April 2003 (available at www.hc-sc.gc.ca/dhp-mps/medeff/advisories-avis/prof/2003/diane-35_2_hpc-cps_e.html [accessed 2005 Oct. 20] advising them not to use Diane-35 for birth control or mild acne because of the increased risk of VTE.
On January 14, 2003 the Canadian Broadcasting Corporation (CBC) ran a television documentary on its Disclosure series about the marketing of Diane-35. (The transcript is available on the CBC's Web site, at www.cbc.ca/disclosure/archives/030114_diane/main.html [accessed 2004 Nov 24].) It featured interviews with users and prescribing physicians who believed that Diane-35 was an ordinary birth control pill. The evidence of higher risks of VTE than with commonly used oral contraceptives was discussed in the documentary.
We sought to discover what proportion of initial users of Diane-35 had medically diagnosed or treated acne. We hypothesized that the CBC documentary would affect prescribing rates to women with and without acne.
To examine this, we linked data from BC PharmaNet, a computer database of all prescriptions dispensed in British Columbia, to administrative data (BC Linked Healthcare Database) on medical consultations and diagno-ses for all women who filled prescriptions for Diane-35, Alesse (100 g levo-norgestrel and 20 g estradiol) or other estrogen– progestin combinations that were available between 1998 and 2003 for oral contraception. Alesse was chosen as the primary comparator because it was approved in 1998 for contraception and in 2002 for acne, attracting no critical media or regulatory attention during the study period (March 1998 through Dec. 2003).
We classified women as having “possible acne” or “no acne” 1 year before and 3 months after the initiation of treatment, with “possible acne” cases requiring any of the following criteria: diagnosis with an ICD-9 (International Classification of Diseases, 9th Revision) code starting with 706 (diseases of sebaceous glands); a consultation with a dermatologist; a prescription for topical anti-infectives (Anatomical Therapeutic Chemical Classification System [ATC] code D10A) or retinoids (ATC D10B); or use of tetracycline (ATC J01A) or macrolide (ATC J01F) antibiotics for 30 days or more. We recorded the monthly numbers of users of Diane-35, Alesse or other oral contraceptives, and those who had previously used Diane-35 and then switched to another oral contraceptive.
To examine the effects of the CBC documentary and Berlex's “Dear Health Professional” letter on prescribing rates, we performed an interrupted time series analysis (SAS/ETS Time Series Forecasting System). This analysis tests the slope of prescribing rates (initiation and switches) against predicted trends.
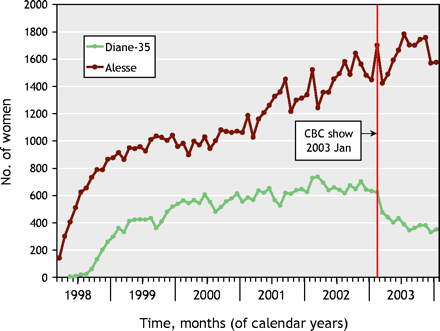
During this period, 1.11 million women aged 13–49 years lived in British Columbia; 275 956 (24.8%) received initial prescriptions for oral contraceptives or Diane-35, which included 33 095 women who received at least one prescription for Diane-35. Of these, 15 064 (45.5%) had no evidence of acne (i.e., they met no criteria for “possible acne”). Of the 83 017 women who initiated use of Alesse over the same period, 71 180 (85.7%) had no evidence of acne. The rate of treatment initiation with Diane-35 increased from the year 1998; plateaued in 2002 (mean 669/mo); declined to 476 (71% of the plateau mean) during February 2003, the month after the CBC broadcast; and continued to decline to the end of the study period (Fig. 1). In contrast, new prescriptions for Alesse increased to the end of 2003. The proportions of women with no acne among those initiating Diane-35 use before and after the CBC broadcast differed little (45.5% v. 46.8%).
Fig. 1: Number of women per month receiving their first prescription for Diane-35 or Alesse, 1998–2003.
Fig. 2 illustrates the numbers of women who switched from Diane-35 to an oral contraceptive, which rose sharply in February 2003 for both subgroups of women (possible and no acne). The difference between predicted and actual trends became significant from February 2003, within one month of the CBC show: for starting Diana-35, p = 0.009; for switching from Diana-35, p < 0.0001.
Fig. 2: Number of women who switched from Diane-35 to an oral contraceptive.
Nearly half of the initial prescriptions for Diane-35 from 1998 through 2003 were for women with no record of acne diagnosis, referral or drug therapy. This suggests that the drug was used for more than its approved indication. Our estimate of off-label use (“no acne”) is conservative as we excluded all “possible acne” cases, not just severe acne that had failed to respond to other treatments. Although women who used only over-the-counter acne treatments and had no medical diagnoses were counted as having no acne, few cases of severe, unresponsive acne would have gone undiagnosed.
Direct-to-consumer advertising may have contributed to routine use of Diane-35 to treat mild acne or as an oral contraceptive. A recent randomized controlled trial2 found that standardized patients who requested an advertised drug were equally likely to receive a prescription whether they had the indicated condition or not; when the standardized patients did not request a drug, physicians prescribed more often for the approved use. This suggests that direct-to-consumer advertising can stimulate off-label prescribing. Furthermore, the packaging similarity of Diane-35 to oral contraceptive pills is likely to have contributed to its rate of prescription for that use.
A widely broadcast news story likely had a countervailing influence. The shift in prescription rates for Diane-35 occurred just after the CBC telecast. In contrast, neither the company's letter to physicians sent in April 2003 nor the warning3 published in CMAJ in February 2003 appear to have had any similar effect on prescription rates. The CBC show conveyed 2 key messages: Diane-35's risk profile and its widespread off-label use. New prescriptions declined similarly in women with possible and with no acne, suggesting that safety concerns affected prescribing decisions to a greater extent than whether or not the drug was prescribed for its approved use. Less use overall meant fewer prescriptions to women with moderate, mild or no acne, among whom the use of Diane-35 is unnecessarily risky.
This study adds to existing evidence that media reports can influence treatment decisions.4 Although news reports are often castigated as alarmist or as raising unrealistic treatment expectations, accurate reporting can also help to build a link between research evidence and practice.
Footnotes
-
This article has been peer reviewed.
Acknowledgements: Meghan MacMahon, Morris Barer and James Wright assisted with study design and commented on successive drafts of this article. Lixiang Yan assisted in planning of data analysis and carried out the analysis.
This study is part of a larger project on the determinants of drug utilization and expenditures that is funded by the Canadian Institutes of Health Research (CIHR). Additional funding was provided by the Michael Smith Foundation (Vice-President's Research Fund, University of British Columbia [UBC]) and a CIHR postdoctoral fellowship (Barbara Mintzes). The funders had no involvement in study design, data collection, analysis or interpretation of findings. The study has received a Certificate of Expedited Ethics Approval (C02-0612) from UBC's Clinical Research Ethics Board. (Expedited approval is the process used for all administrative data analyses.)
Competing interests: None declared.
REFERENCES
- 1.
- 2.
- 3.
- 4.