- © 2007 Canadian Medical Association
Stakeholders cast it as a glittering model for future intergovernmental oversight of public health initiatives.
Skeptics, though, describe it as altogether light on concrete accomplishments. One calls it an “enduring Canadian mystery,” while another says Canada's public health community is entirely confused about what has actually been accomplished under the National Immunization Strategy, a $45 million/5-year initiative that was launched with considerable fanfare in 2003 as a national blueprint for ensuring vaccine access, supply, safety and efficacy.
All are agreed, though, that the National Immunization Strategy has successfully distributed a separate $300 million fund to provide more equitable access for 4 recently developed vaccines: acellular pertussis, meningococcal C conjugate, pneumococcal conjugate and varicella. Virtually all jurisdictions now offer the quartet as part of their panoply of public health vaccines, and it is estimated that twice as many Canadian children were protected against those childhood diseases in 2006 as compared with 2003. A similar $300 million/3-year fund was recently established by the Conservative government for an immunization drive against the human papillomavirus (CMAJ 2007;176[8]:1071).

Figure. Some 222 Canadians had contracted mumps by mid-May as a result of an outbreak that began in Nova Scotia and spread to Ontario, where 9 cases were confirmed and Toronto public health officials issued warnings after an infected person unknowingly exposed 300 others at a downtown bar. Photo by: Canapress
But progress on other elements of the National Immunization Strategy appears less evident. After nearly 4 years, organizers say rollout of program specifics is in the offing, but nowhere to be presently found are anything like a national immunization registry network, a national immunization research plan, training programs for health professionals, educational programs for the public, “national goals and recommendations for vaccine-preventable diseases” or a nationally harmonized pediatric immunization schedule. Meanwhile, immunization issues have become headline grabbers. There have been outbreaks of mumps in Nova Scotia and Ontario, as well as debates over whether a booster dose of the chicken pox vaccine is needed to limit incidence of the disease over the long term.
Progress on the National Immunization Strategy may well have occurred, but no one knows that for certain because little effort has been made to inform the community of developments, says Mary Appleton, senior manager of the Canadian Coalition for Immunization Awareness and Promotion. “Frankly, it needs more communication. What is it doing? A lot of us are asking those questions. That's the major failing of the strategy. No one is communicating what they're doing.”
Public Health Agency of Canada Immunization and Respiratory Infections Division Director, Dr. Theresa Tam, who is responsible for National Immunization Strategy oversight, counters that the initiative has been extremely successful in ensuring equitable access to vaccines, in improving “program coordination and program planning” and in doing the “development work” for future programming.
Such divergent opinion and confusion about the National Immunization Strategy's accomplishments may in part be a function of where it sits along the convoluted spectrum of immunization activities in Canada.
The National Immunization Strategy was established in 2003 by the First Ministers' Accord on Health Care Renewal and the Conference of Federal, Provincial and Territorial Deputy Ministers of Health in a bid to end the disparity in vaccination programs across the nation, while establishing a “collaborative” approach to immunization in Canada. It was to have 5 core components (“development of national goals and recommendations for immunization programs, immunization program planning, vaccine safety, vaccine supply and an immunization registry network”) and 5 “crosscutting” components (immunization research, professional and public education, vaccine-preventable disease surveillance, and “special populations” such as Aboriginals, travellers or migrants).
With 14 health care jurisdictions (10 provinces, 3 territories and the First Nations and Inuit Health Branch of Health Canada) in Canada, a collaborative approach is invariably difficult. Compounding that is the process by which vaccines are included in immunization schedules.
After a vaccine is approved as safe and effective by Health Canada, it is subject to the scrutiny of the National Advisory Committee on Immunization, a group of pediatric and adult immunization experts who essentially volunteer to study and recommend whether a vaccine should be made available for routine use among specific population groups.
In turn, the advisory committee's recommendations are scrutinized by the Canadian Immunization Committee, a 19-member committee comprised primarily of provincial public health officials. Outgoing Co-chair Dr. Greg Hammond says the Canadian Immunization Committee is responsible for developing the operational plans by which the vaccines recommended by the National Advisory Committee on Immunization might be made available to the public. It reports confidentially to the Communicable Disease Control Expert Group, which in turn reports to a 14-member federal/provincial body called The Council (typically, provincial medical officers of health). All, and various other expert and issue groups, are collectively known as the Public Health Network, which reports to the Federal/Provincial/Territorial Conference of Deputy Ministers of Health.
Therein, one immunization expert wryly notes, “lies the primary outcome of the National Immunization Strategy exercise: the creation of a massive intergovernmental vaccination bureaucracy.”
But that is no mean feat, argues Hammond, director of the Public Health Branch and Communicable Disease Control for Manitoba Health. “Many of us in public health feel that if we can get this right for immunization, then it bodes well for many other examples of collaboration around public health and even health care for the future. If we can do this in a way that is beneficial for Canadians in a sustainable way, then when we try to put in place other prevention programs, this will be a good model.”
Beyond, and some say irrespective, of the consensus achieved through that intermediary collaborative process lies the final hurdle for approval of a new vaccine. Each province or territory makes a separate determination on whether to actually publicly fund the vaccine. Traditionally, they have weighed the cost of vaccines against the cost of treating the illness and chosen the cheaper option, while, historically, it has been difficult to get provinces to add vaccines to their rosters because of the high start-up costs associated with training health care professionals, ensuring supply and conducting public awareness campaigns for new vaccines. But other factors often come into play, including delivery systems, epidemiology and the depth of a province's pockets, says BC Chief Medical Officer of Health Dr. Perry Kendall. “It often depends on how much money there is in a province, at a particular time, or whether or not that disease has picked up any political traction.”
In the case of the 4 new vaccines added in 2004, the federal government nudged the provinces along by using money as a carrot. The Conservative government opted for the same approach by creating a human papillomavirus trust fund, rather than simply establishing a large new fund for vaccines. And indeed, there are many now in the approval pipeline, including 2 rotavirus vaccines, others for measles, mumps, rubella and varicella, and a host in development as manufacturers foray into the realm of vaccines for chronic diseases.
Arguing that money is the agent that best drives change, Canadian Paediatric Society President-elect, Dr. Joanne Embree, says there's a need to create a permanent trust fund for all new and forthcoming vaccines. “The provinces could buy all their vaccines from the trust fund and that would take it out of the yearly ‚having to go to their treasury departments',” adds the pediatrician and head of the University of Manitoba's Department of Medical Microbiology. “It's a logical use of vaccines and it means that's there's sufficient money in the system for everybody to get vaccinated and universal access.”
Kendall argues such an untargeted fund is essential because most vaccination programs “fall outside the ambit of medicare,” leaving provincial public health officials to scramble for monies. “That obviously favours provinces with bigger revenue streams.”
Beyond such broad issues of funding and administrative infrastructure lie uncertainties surrounding many of the specific elements that once were lauded as future outcomes of the National Immunization Strategy, or which, from a public perspective, are the truest measure of accountability.
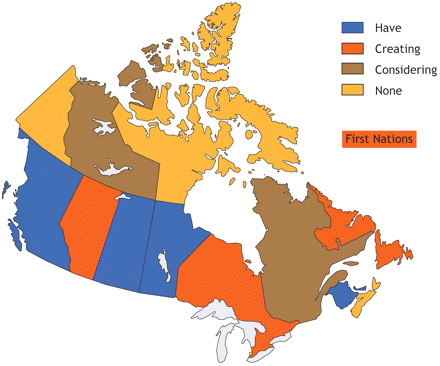
For example, National Immunization Strategy proponents originally envisioned a national immunization database, collating such information as the percentage of Canadian children who had been immunized against a specific disease. But such a national database is no longer the objective, says Tam. Rather, 14 separate registries are envisioned, (see Fig. 1) using standardized data collection and management tools.
Fig. 1: The 14 jurisdictions recognized under the National Immunization Strategy are at various stages of creating electronic systems to track immunizations.
Still, Tam is optimistic that some form of registries network will be rolled out in 2009 (particularly after $100 million was provided through the Canada Infoway initiative specifically for immunization registry technologies, under a program called Panorama), along with other specifics such as bar coding of vaccines to track use, and a standardized, nationwide immunization schedule. Later this year, the Public Health Agency of Canada also hopes to unveil a report on a consensus conference on national goals and recommendations that was held in 2005.
A quick rollout of such specifics, as well as the appointment of a national commissioner for children and youth, would make it easier to “benchmark” progress and generate broader public support and funding for immunization initiatives, argues pediatrician and former interim CMAJ Editor-in-Chief and former Dalhousie University dean of medicine Dr. Noni MacDonald.
“The formation of the CIC (Canadian Immunization Committee) and how they are trying to get more collaborative partnerships work across all the provinces and territories was a real step forward, but there's more work to do because we don't have a vaccine registry or a national immunization schedule that's followed everywhere. There are some reasons from a geography and disease prevalence point of view why you might see some variations across the country, but we have other variations in the schedule across the country for which there's no evidence that we need to have that variation. It's only confusing to parents when you're trying to do national education and it's confusing when people move.”
Embree contends there's also a need for quicker rollout of national goals and recommendations, particularly concrete action plans for specific vaccine-preventable diseases, as well as a need to develop strategies to reach those who don't have family doctors or pediatricians and thus are less likely to be reminded of the value of vaccination.
Others argue that Canada hasn't even begun to flesh out all of the requisite elements of a true national immunization strategy, including a national research plan, as well as a national strategy for insuring that there is adequate long-term, indigenous industrial capability to produce needed vaccines. There's a definite need for additional funding and coordination of a research program aimed at assessing the efficacy of vaccines, and dosages, on an ongoing basis, says Kendall. “Right now, we don't have the ongoing core funding for vaccines and we don't have the capacity nationally to do particular evaluation studies.”
“It's important that we understand the issues around safety, around new vaccines, around supply and demand,” says Dr. Bhagirath Singh, scientific director of the Canadian Institutes of Health Research's Institute of Infection and Immunity. “If there is a shortage of vaccines, what is Canada going to do? Are we going to legislate that all the vaccines cannot go out and they have to be used here? Or we going to negotiate with other partners that this a policy we're going to adopt if there is genuine need or shortage? I think those issues are real. We have to consider that, as a country, we are fortunate where we have local manufacturers who supply Canadian demands for vaccines. But if these manufacturers decide tomorrow that they'll be better off producing it in India or China or Mexico, are we going to lose capacity to meet our needs. It's a health care issue. There's a genuine need for the country to have a national vaccine strategy.”