Abstract
BACKGROUND: Most studies of long-term renal outcomes after acute critical illness have enrolled patients with pre-existing renal dysfunction. We assessed renal outcomes in patients who did not have pre-existing renal disease and who were admitted to hospital for acute critical illness.
METHODS: We identified adults who did not have pre-existing renal disease and who were admitted to hospital for acute critical illness between 2000 and 2011, from the Taiwan National Health Insurance Research Database. Each patient was matched 1:2 with controls without acute critical illness, according to age, sex and index date. A subset was further matched 1:1 with controls using propensity scores. Outcomes included acute kidney injury, chronic kidney disease and end-stage renal disease.
RESULTS: We evaluated 33 613 patients with acute critical illness matched to 63 148 controls, of whom 14 218 were propensity matched to 14 218 controls. Patients with acute critical illness had incidence rates per 10 000 person-years of 9.45 for acute kidney injury, 78.3 for chronic kidney disease and 21.0 for end-stage renal disease. In the propensity-matched cohort, patients with acute critical illness had significantly higher risks of acute kidney injury (adjusted hazard ratio [aHR] 2.92, 95% confidence interval [CI] 1.78–4.77), chronic kidney disease (aHR 1.81, 95% CI 1.57–2.08), and end-stage renal disease (aHR 3.60, 95% CI 2.50–5.18). Acute critical illness conferred higher mortality risk among patients who subsequently developed end-stage renal disease (aHR 3.37, 95% CI 2.07–5.49) or chronic kidney disease (aHR 2.16, 95% CI 1.67–2.80).
INTERPRETATION: Patients with acute critical illness and without pre-existing renal disease have a higher risk of adverse renal outcomes and subsequent mortality. A resolved episode of critical illness has implications for future renal function surveillance, even in patients without pre-existing renal disease.
Acute kidney injury1 is prevalent in patients with acute critical illness, with 53.2% in patients with severe sepsis, 12.9% in patients with acute myocardial infarction and 20.9% in patients with stroke.2–4 The incidence of acute kidney injury has risen substantially in the last decade, associated with the burden of acute critical illnesses, such as septicemia, respiratory failure and shock.5,6 The high prevalence of acute kidney injury in patients with acute critical illness is explained by acute physiologic abnormalities that include peripheral vasodilation, decreased cardiac output and intravascular fluid volume, which attenuate renal function reversibility even in patients with previously preserved renal function.7 Notably, about 30% of patients with acute critical illness have preadmission renal dysfunction.8 In the setting of acute critical illness, acute kidney injury requiring dialysis has been associated consistently with in-hospital mortality and morbidities, such as chronic kidney disease and end-stage renal disease.9–12
For long-term renal complications, chronic kidney disease is the most prevalent renal dysfunction.13,14 Patients with chronic kidney disease have a high rate of progression to end-stage renal disease, cardiovascular disease and mortality.14 Among patients admitted to hospital, the risk of end-stage renal disease is the highest in those with acute-on-chronic kidney disease, followed by those with acute kidney injury and those with chronic kidney disease, relative to patients without kidney diseases.15,16
Recently, acute kidney injury and chronic kidney disease have been recognized as related disease entities in that they represent a continuum of the disease process. The term “acute kidney disease” is used to define the disease course after acute kidney injury in patients with acute critical illness whose pathophysiologic processes are ongoing.17 Thus, nephrologists are recommended to keep patients who have acute critical illness and acute kidney injury or chronic kidney disease under surveillance for risk of subsequent end-stage renal disease.2,18
Despite robust evidence on acute and chronic renal risks in patients with acute critical illness, most studies have enrolled patients with pre-existing renal dysfunction.9–16 Information about whether patients with acute critical illness without pre-existing kidney disease have adverse renal outcomes is scarce. We aimed to find out whether these patients carry long-term renal risks compared with patients with nonacute critical illness.
Methods
Data source
This retrospective cohort study used data from Taiwan’s Longitudinal Health Insurance Database 2000 (LHID2000) of the National Health Insurance Research Database (NHIRD) from Jan. 1, 2000, to Dec. 31, 2011. The NHIRD, including all beneficiaries’ inpatient and outpatient medical claims, enrolled data from 23 million Taiwanese people in 2014 (99.9% of the population).19,20 The LHID2000 database comprises 1 000 000 beneficiaries randomly selected from the National Health Insurance 2000 Registry for Beneficiaries.21 Definitions of diseases in the LHID are based on the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM).
Study design and participants
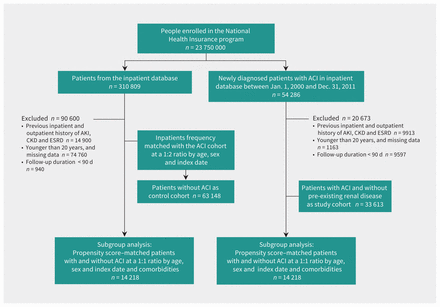
We identified inpatients with the following newly diagnosed critical illnesses: septicemia (ICD-9-CM 038), septic shock (ICD-9-CM 785.52), acute myocardial infarction (ICD-9-CM 410), stroke (ICD-9-CM 430–432) and shock (ICD-9-CM 785.59, 998.09, and 958.4) (Figure 1). We set the date of the diagnosis of critical illness as the index date. We excluded patients with previous inpatient and outpatient history of acute kidney injury (ICD-9-CM 584.5, 584.6, 584.7, 584.8, and 584.9), chronic kidney disease (stage G1–5; ICD-9-CM 585.1–581.5) and end-stage renal disease (ICD-9-CM 586), and patients younger than 20 years. We also excluded patients with incident acute kidney injury, chronic kidney disease or end-stage renal disease within 90 days of the index date. Finally, we enrolled patients with acute critical illness without pre-existing renal disease as the study cohort.
Flowchart of study design and patient selection. Note: ACI = acute critical illness, AKI = acute kidney injury, CRD = chronic renal disease, ESRD = end-stage renal disease.
The control cohort was randomly selected from the database of inpatients and outpatients without acute critical illness, using the same exclusion criteria as the control cohort. Each patient with acute critical illness was frequency matched by age (a span of every 5 years), sex and index year with control patients, at a 1:2 ratio. We also performed propensity-score matching between the acute critical illness and nonacute critical illness cohorts by gender, age, index year and underlying comorbidities at a 1:1 ratio, to reduce the risk of confounding effect.
The follow-up period commenced from the index date until the occurrence of renal complications, the date on which the patients were censored because of withdrawal from the National Health Insurance program, or the end of the study (Dec. 31, 2011), whichever occurred first.
Demographic and comorbid variables
We obtained demographic information and pre-existing comorbidities that affect renal outcomes, such as diabetes, hypertension, cirrhosis, hyperlipidemia, coronary artery disease, congestive heart failure and cancer. We also identified potential confounders of critical illness and the composite renal outcome such as Charlson Comorbidity Index score, stay in the intensive care unit (ICU) or mechanical ventilation.5,6,8,22,23
Outcomes
Primary outcomes were diagnosis of acute kidney injury, chronic kidney disease or end-stage renal disease in the follow-up period. We identified patients who fulfilled the criteria of acute kidney injury according to the clinical practice guideline from the Kidney Disease Improving Global Outcomes,1 using ICD-9-CM. Clinically recognized chronic kidney disease is defined according to the criteria in the clinical practice guideline from the Kidney Disease Outcomes Quality Initiative.24 Patients with end-stage renal disease were identified if they were registered in the catastrophic illness registry, indicating dialysis dependence for more than 3 months.
We evaluated the effect of acute critical illness status on mortality, modified by time-dependent acute kidney injury, chronic kidney disease or end-stage renal disease as the secondary end point. The accuracy of acute kidney injury, chronic kidney disease and end-stage renal disease diagnoses recorded in our claims data has been validated.25,26
Statistical analysis
We compared demographic characteristics and comorbidities in the study and control cohorts using 2 models: a frequency-matched and a propensity score–matched model. In the frequency-matched model, continuous data are reported as mean and standard deviation (SD). Categorical data are expressed as counts and percentages. χ2 and Student t tests were used for comparing dichotomous and continuous variables, respectively, between the 2 cohorts. A 2-sided p value of less than 0.05 was considered significant. In the propensity score–matched model, we used logistic regression to estimate the risk of diseases by calculating the propensity score in both cohorts in the propensity score–matching process.27 Baseline characteristics of the acute critical illness and control cohorts were compared using standardized mean differences. Values of standardized mean differences ≤ 0.10 showed a negligible difference in mean values between the 2 cohorts.
We calculated the cumulative incidence of the first diagnosis of primary renal outcome using the Kaplan–Meier method, and used the log-rank test to verify the equality of survivor functions between study groups. We used Cox proportional hazards regression models to determine the effects of acute critical illness on the risks of acute kidney injury, chronic kidney disease or end-stage renal disease and the effects of acute critical illness status on mortality in patients with acute critical illness and nonacute critical illness in the frequency- and propensity score–matched cohorts. Because death is a competing risk for renal outcomes as a function of critical illness, we analyzed a competing risk model to estimate the subhazard ratios (SHRs) and 95% confidence intervals (CIs) of the renal outcomes.28 Covariables significantly associated with outcome in the univariable competing-risks regression model were further examined by multivariable regression model. We used SAS (Version 9.3; SAS Institute, Inc., Cary, NC) for all statistical analyses.
Ethics approval
Patient records were anonymized, and this study was approved for exemption from informed consent rules by the Ethics Review Board of China Medical University and Hospital in Taiwan (CMUH104-REC2–115-CR2).
Results
Our study included 33 613 patients with acute critical illness without pre-existing renal disease, matched with 63 148 controls. Of these, 14 218 patients with acute critical illness were matched with 14 218 controls using propensity scores. The frequency-matched acute critical illness cohort predominantly included patients who were older than 65 years (53.1%), were men (57.7%) and had hypertension (67.2%) and coronary artery disease (36.9%). Sepsis and stroke were the leading causes of critical illnesses in the acute critical illness cohort, with a high prevalence of ICU stay (35.6%) and mechanical ventilation (16.9%). In the propensity score–matched model, the acute critical illness and nonacute critical illness cohorts had similar baseline characteristics (Table 1).
Baseline characteristics of patients admitted to hospital (2000–2011), by the presence of critical illness, and after matching for age, sex and index date, or for propensity score*
Primary outcome
The mean follow-up period for patients developing acute kidney injury, chronic kidney disease and end-stage renal disease in the frequency-matched acute critical illness cohort was mean ± SD 4.75 ± 3.20, 4.66 ± 3.17, and 4.75 ± 3.19 years, respectively (Table 2). Acute kidney injury was more frequent in the acute critical illness cohort than in the control cohort (9.45 v. 2.35 per 10 000 person-years, incidence rate ratio [IRR] 4.02, adjusted hazard ratio [aHR] 2.84, 95% CI 1.95–4.15). Notably, 1225 patients in the acute critical illness cohort developed chronic kidney disease, with an incidence rate of 78.3 per 10 000 person-years, also significantly higher than controls (IRR 2.08, aHR 1.81, 95% CI 1.61–2.04). Furthermore, 335 patients in the acute critical illness cohort developed end-stage renal disease, with an incidence rate of 21.0 per 10 000 person-years, significantly higher than controls (IRR 4.27, aHR 3.26, 95% CI 2.52–4.22). Results were similar in the propensity-matched subset for acute kidney injury (aHR 2.92, 95% CI 1.78–4.77), chronic kidney disease (aHR 1.81, 95% CI 1.57–2.08) and end-stage renal disease (aHR 3.60, 95% CI 2.50–5.18).
Incidence and hazard ratio of acute kidney injury, chronic kidney disease and end-stage renal disease in patients with critical illness compared with those without, by type of matching
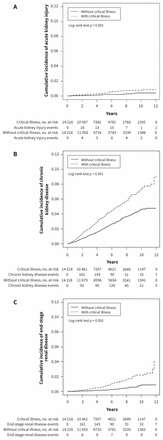
Figure 2 shows the cumulative risks of renal outcomes for the propensity score–matched cohorts followed for more than 12 years. The risks were significantly higher in the acute critical illness cohort than in the control cohort (log-rank test, p < 0.001). Notably, the incidence of chronic kidney disease prevailed over that of end-stage renal disease and acute kidney injury in the acute critical illness cohort. In the competing risk regression model, the risks of acute kidney injury, chronic kidney disease and end-stage renal disease remained higher in the propensity score–matched acute critical illness cohort (adjusted subhazard ratios [aSHRs] 1.85, 95% CI 1.23–2.79; aSHRs 1.73, 95% CI 1.51–1.98; and aSHRs 2.91, 95% CI 2.11–4.00, respectively; Table 3) than in the controls.
Cumulative incidence of acute kidney injury (A), chronic kidney disease (B) and end-stage renal disease (C) in individuals with and without critical illness by propensity-score matching.
Incidence and subhazard ratio of acute kidney injury, chronic kidney disease and end-stage renal disease in propensity score–matched cohorts, using competing-risks regression models
Table 4 shows the predictors of acute kidney injury, chronic kidney disease and end-stage renal disease in the propensity score–matched cohorts. Among these comorbidities, diabetes, hypertension and congestive heart failure were universal risk factors for acute kidney injury, chronic kidney disease and end-stage renal disease in our cohorts. Mechanical ventilation was associated with acute kidney injury (aHR 2.56, 95% CI 1.37–4.79), but not with chronic kidney disease and end-stage renal disease. Next, patients were stratified by the primary causes of critical illnesses, and renal outcome was compared for the propensity score–matched cohorts (Table 5). Among them, septicemia and septic shock were the strongest risk factor for acute kidney injury (aHR 4.93, 95% CI 2.91–8.35), chronic kidney disease (aHR 2.46, 95% CI 2.10–2.90) and end-stage renal disease (aHR 5.38, 95% CI 3.65–7.94).
Predictors of de novo acute kidney injury, chronic kidney disease and end-stage renal disease by Cox regression model among propensity score–matched patients with acute critical illness compared with those without
Comparisons of incidence and hazard ratios of acute kidney injury, chronic kidney disease and end-stage renal disease in patients with different subtypes of critical illness compared with those without critical illness in the propensity score–matched cohorts
Secondary outcome
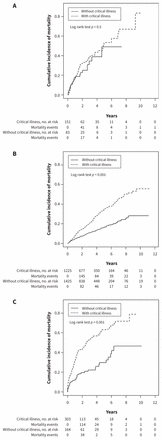
We analyzed mortality after the development of acute kidney injury, chronic kidney disease and end-stage renal disease in patients with acute critical illness and nonacute critical illness in both the frequency-matched and propensity score–matched cohorts (Table 6). Patients with acute critical illness had a higher risk of death after developing chronic kidney disease (aHR 2.16, 95% CI 1.67–2.80) and after progression to end-stage renal disease (aHR 3.37, 95% CI 2.07–5.49) than did the controls in the frequency-matched cohorts, with similar results seen in the propensity-matched cohorts. Figure 3 shows the higher cumulative risk of death after the development of chronic kidney disease and end-stage renal disease in the frequency-matched acute critical illness cohorts than that of the controls. However, we observed no significant difference in the risk of mortality after patients in the frequency-matched cohort had developed acute kidney injury.
Incidence and hazard ratios of mortality after patients developed acute kidney injury, chronic kidney disease or end-stage renal disease in the critical illness cohorts compared with those in the controls, in age- and sex-matched and propensity score–matched cohorts
Cumulative incidence of death after development of acute kidney disease (A), chronic kidney disease (B) and end-stage renal disease (C) in the age- and sex-matched cohorts.
Interpretation
We investigated long-term risks of renal sequelae and subsequent mortality in patients with acute critical illness without pre-existing renal disease. The acute critical illness cohort had an increased risk of renal complications, with the highest incidence of chronic kidney disease, followed by end-stage renal disease and acute kidney injury. Among the primary causes of critical illnesses, septicemia and septic shock were the strongest risk factors for acute kidney injury, chronic kidney disease and end-stage renal disease. These results showed that patients with acute critical illness without apparent underlying renal disease — a group traditionally considered to be at low risk for renal disease — have clinically relevant long-term renal risks. In addition, the status of acute critical illness has a negative impact on mortality, modified by subsequent development of renal complications.
Recent studies have shown that milder forms of acute kidney injury and even recovery of renal function after acute kidney injury are associated with both short- and long-term mortality. 16,29,30 Bucaloiu and colleagues advised that even a reversible acute kidney injury event is a risk factor for death and de novo chronic kidney disease (hazard ratio [HR] 1.91, compared with the nonacute kidney injury group).30 In our study, patients with acute critical illness might have renal recovery upon discharge, and showed a similar risk of chronic kidney disease (aHR: 1.81). The Kidney Disease Improving Global Outcomes guideline on acute kidney injury recommends assessing patients 2 months after acute kidney injury to evaluate the completeness of disease resolution and to detect either new-onset or worsening chronic kidney disease.2 Thus, we propose surveillance of renal function at 30–90 days and then at least yearly in patients with acute critical illness even when they do not have pre-existing renal disease.18
Among traditional risk factors for chronic kidney disease and end-stage renal disease, hypertension, diabetes and congestive heart failure were prevalent in our acute critical illness cohorts. Our results showed that acute physiologic abnormalities and mechanical ventilation are predictors of de novo acute kidney injury. To date, the Acute Physiologic and Chronic Health Evaluation (APACHE) II scoring system has been widely used for predicting survival and recovery of renal function in studies of acute kidney injury.31 Although our study did not evaluate the APACHE II score owing to database limitation, we used mechanical ventilation as a proxy for illness severity as it is an independent factor for mortality in critical illness patients.8 The US Renal Data System (2006) indicates the risk of acute kidney injury in patients who are older, have received ICU care and have multiple organ dysfunction.32 This evidence shows that acute kidney injury is a complex syndrome reflecting underlying disease burden; subsequent chronic kidney disease and end-stage renal disease may account for increased mortality in patients with acute critical illness.
Sepsis is the leading cause of acute kidney injury, accounting for 45%–70% of all acute kidney injury events in patients in the ICU.8,33 Sepsis was associated with higher aberrations in hemodynamics, illness severity and need for mechanical ventilation. 34 Circulatory shock and infection are important risk factors for acute kidney injury in patients in the ICU.35 Bellomo and colleagues proposed that even a single episode of septic acute kidney injury is associated with increased risk of developing chronic kidney disease.36 In our study, patients with sepsis had higher risks of renal complications than did those with cardiovascular diseases. We suspect that although patients may experience renal recovery from septic acute kidney injury, they may still be predisposed to subsequent chronic kidney disease and end-stage renal disease. Epidemiological studies have reported an association between pneumonia and subsequent acute kidney injury and end-stage renal disease, supporting our findings that infections may trigger not only acute but also chronic renal damage.37,38
Limitations
The study has potential limitations. First, retrospective cohort studies have various biases because of the possible unmeasured factors related to both exposure and outcomes. To limit the potential for confounding factors, we used propensity-score matching across a wide range of clinical characteristics to establish the cohorts. Second, the claim database was limited because biological information such as laboratory data (e.g., glomerular filtration rate as the overall index of renal function) were unavailable. Because physicians were reimbursed for chronic kidney disease and end-stage renal disease care after quarterly reporting of longitudinal estimated glomerular filtration rate and urinary screening data, this strengthens the validity of the renal disease diagnosis in the database we used. Finally, some patients with pre-existing unrecognized renal dysfunction could have been included in the study and control cohorts. Nevertheless, based on the consistent results obtained using various approaches, our study provides important evidence to support the increased risk of renal complications in patients with acute critical illness.
Conclusion
Our study showed higher risks of acute kidney injury, chronic kidney disease and end-stage renal disease in patients with acute critical illness without pre-existing renal disease. Developing chronic kidney disease and end-stage renal disease further increased subsequent mortality risks. Nephrological surveillance of patients with acute critical illness, particularly those with sepsis and ischemic stroke, should be an essential part of their follow-up care.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Shih-Ting Huang and Chia-Hung Kao were responsible for the conception and design of the work. Chia-Hung Kao was responsible for providing the study materials. All authors contributed to the acquisition analysis and interpretation of the data. All of the authors drafted the manuscript, revised it critically for important intellectual content, gave approval of the final version to be published and agreed to be accountable for all aspects of the work.
Funding: This work was supported by grants from the Ministry of Health and Welfare, Taiwan (MOHW107-TDU-B-212-123004), China Medical University Hospital, Academia Sinica Stroke Biosignature Project (BM10701010021), MOST Clinical Trial Consortium for Stroke (MOST 106-2321-B-039-005-).” Tseng-Lien Lin Foundation, Taichung, Taiwan, and Katsuzo and Kiyo Aoshima Memorial Funds, Japan. The funders had no role in the study design, data collection and analysis, publication decision, or manuscript drafting. No additional external funding was received for this study.
Data sharing: The data set used in this study is held by the Taiwan Ministry of Health and Welfare (MOHW). The Ministry of Health and Welfare must approve applications to access these data. Any researcher interested in accessing this data set can submit an application form to MOHW to request access. Please contact the staff of MOHW (stcarolwu{at}mohw.gov.tw) for further assistance. Taiwan Ministry of Health and Welfare Address: No.488, Sec. 6, Zhongxiao E. Rd., Nangang Dist., Taipei City 115, Taiwan (R.O.C.). Phone: +886-2-8590-6848. All relevant data are within the paper.
- Accepted July 4, 2018.